Abstract
Rationale
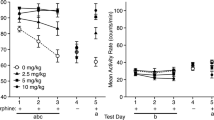
Previous studies found that naloxone (NLX) facilitated choice extinction of ethanol conditioned place preference (CPP) using long (60 min) test sessions, but there is little information on the variables determining this effect.
Objectives
These studies examined repeated exposure to NLX during extinction of ethanol- or cocaine-induced CPP using both short and long tests.
Methods
DBA/2J mice were injected with NLX (0 or 10 mg/kg) before three 10- or 60-min choice extinction tests (experiment 1). All mice received a final 60-min test without NLX. Post-test NLX was given in experiment 2. Experiment 3 tested whether NLX would affect a forced extinction procedure. Experiment 4 tested its effect on extinction of cocaine-induced CPP.
Results
Pre-test (but not post-test) injections of NLX-facilitated choice extinction of ethanol CPP at both test durations. Pre-test NLX also facilitated forced extinction. However, pre-test NLX had no effect on choice extinction of cocaine CPP.
Conclusions
Extinction test duration is not critical for engaging the opioid system during ethanol CPP extinction (experiment 1). Moreover, NLX’s effect does not depend on CPP expression during extinction, just exposure to previously conditioned cues (experiment 3). The null effect of post-test NLX eliminates a memory consolidation interpretation (experiment 2) and the failure to alter cocaine CPP extinction argues against alteration of general learning or memory processes (experiment 4). Overall, these data suggest that the endogenous opioid system mediates a conditioned motivational effect that normally maintains alcohol-induced seeking behavior, which may underlie the efficacy of opiate antagonists in the treatment of alcoholism.




Similar content being viewed by others
References
Bardo MT, Bevins RA (2000) Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology 153:31–43
Bechtholt AJ, Cunningham CL (2005) Ethanol-induced conditioned place preference is expressed through a ventral tegmental area dependent mechanism. Behav Neurosci 119:213–223. doi:10.1037/0735-7044.119.1.213
Becker A, Grecksch G, Kraus J et al (2002) Rewarding effects of ethanol and cocaine in mu opioid receptor-deficient mice. Naunyn Schmiedeberg's Arch Pharmacol 365:296–302. doi:10.1007/s00210-002-0533-2
Bormann NM, Cunningham CL (1997) The effects of naloxone on expression and acquisition of ethanol place conditioning in rats. Pharmacol Biochem Behav 58:975–982. doi:10.1016/S0091-3057(97)00304-3
Bossert JM, Marchant NJ, Calu DJ, Shaham Y (2013) The reinstatement model of drug relapse: recent neurobiological findings, emerging research topics, and translational research. Psychopharmacology 229:453–476
Bouton ME (1993) Context, time, and memory retrieval in the interference paradigms of Pavlovian learning. Psychol Bull 114:80–99. doi:10.1037/0033-2909.114.1.80
Bouton ME (2004) Context and behavioral processes in extinction. Learn Mem 11:485–494. doi:10.1101/lm.78804
Buffalari DM, See RE (2010) Amygdala mechanisms of Pavlovian psychostimulant conditioning and relapse. Curr Top Behav Neurosci 3:73–99. doi:10.1007/7854_2009_18
Burattini C, Gill TM, Aicardi G, Janak PH (2006) The ethanol self-administration context as a reinstatement cue: acute effects of naltrexone. Neuroscience 139:877–887. doi:10.1016/j.neuroscience.2006.01.009
Castellano C, Cabib S, Puglisi-Allegra S (1996) Psychopharmacology of memory modulation: evidence for multiple interaction among neurotransmitters and hormones. Behav Brain Res 77:1–21
Childress AR, Mozley PD, McElgin W et al (1999) Limbic activation during cue-induced cocaine craving. Am J Psychiatry 156:11–18. doi:10.1176/ajp.156.1.11
Ciccocioppo R, Martin-Fardon R, Weiss F (2002) Effect of selective blockade of μ1 or δ opioid receptors on reinstatement of alcohol-seeking behavior by drug-associated stimuli in rats. Neuropsychopharmacology 27:391–399. doi:10.1016/S0893-133X(02)00302-0
Ciccocioppo R, Lin D, Martin-Fardon R, Weiss F (2003) Reinstatement of ethanol-seeking behavior by drug cues following single versus multiple ethanol intoxication in the rat: effects of naltrexone. Psychopharmacology 168:208–215. doi:10.1007/s00213-002-1380-z
Crombag HS, Bossert JM, Koya E, Shaham Y (2008) Review. Context-induced relapse to drug seeking: a review. Philos Trans R Soc Lond Ser B Biol Sci 363:3233–3243. doi:10.1098/rstb.2008.0090
Cunningham CL (1995) Localization of genes influencing ethanol-induced conditioned place preference and locomotor activity in BXD recombinant inbred mice. Psychopharmacology 120:28–41. doi:10.1007/BF02246142
Cunningham CL, Prather LK (1992) Conditioning trial duration affects ethanol-induced conditioned place preference in mice. Animal Learn Behav 20:187–194. doi:10.3758/BF03200416
Cunningham CL, Dickinson SA, Okorn DM (1995) Naloxone facilitates extinction but does not affect acquisition of ethanol-induced conditioned place preference. Experimental and Clinical Psychopharmacology (Berl) 3:330–343
Cunningham CL, Henderson CM, Bormann NM (1998) Extinction of ethanol-induced conditioned place preference and conditioned place aversion: effects of naloxone. Psychopharmacology 139:62–70. doi:10.1007/s002130050690
Cunningham CL, Dickinson SD, Grahame NJ et al (1999) Genetic differences in cocaine-induced conditioned place preference in mice depend on conditioning trial duration. Psychopharmacology 146:73–80. doi:10.1007/s002130051090
Cunningham CL, Ferree NK, Howard MA (2003) Apparatus bias and place conditioning with ethanol in mice. Psychopharmacology 170:409–422. doi:10.1007/s00213-003-1559-y
Cunningham CL, Gremel CM, Groblewski PA (2006) Drug-induced conditioned place preference and aversion in mice. Nat Protoc 1:1662–1670. doi:10.1038/nprot.2006.279
Cunningham CL, Groblewski PA, Voorhees CM (2011) Place conditioning. In: Olmstead MC (ed) Animal models of drug addiction. Humana Press, Totowa, NJ, pp 167–190
Dayas CV, Liu X, Simms JA, Weiss F (2007) Distinct patterns of neural activation associated with ethanol seeking: effects of naltrexone. Biol Psychiatry 61:979–989. doi:10.1016/j.biopsych.2006.07.034
Duka T, Crombag HS, Stephens DN (2011) Experimental medicine in drug addiction: towards behavioral, cognitive and neurobiological biomarkers. J Psychopharmacol 25:1235–1255. doi:10.1177/0269881110388324
Gianoulakis C (2001) Influence of the endogenous opioid system on high alcohol consumption and genetic predisposition to alcoholism. J Psychiatry Neurosci 26:304–318
Gianoulakis C (2009) Endogenous opioids and addiction to alcohol and other drugs of abuse. Curr Top Med Chem 9:999–1015. doi:10.2174/156802609789630956
Gremel CM, Cunningham CL (2007) Role of test activity in ethanol-induced disruption of place preference expression in mice. Psychopharmacology 191:195–202. doi:10.1007/s00213-006-0651-5
Gremel CM, Young EA, Cunningham CL (2011) Blockade of opioid receptors in anterior cingulate cortex disrupts ethanol-seeking behavior in mice. Behav Brain Res 219:358–362. doi:10.1016/j.bbr.2010.12.033
Groblewski PA, Franken FH, Cunningham CL (2011) Inhibition of extracellular signal-regulated kinase (ERK) activity with SL327 does not prevent acquisition, expression, and extinction of ethanol-seeking behavior in mice. Behav Brain Res 217:399–407. doi:10.1016/j.bbr.2010.11.018
Gutstein HB, Akil H (2001) Opioid analgesics. In: Hardman JG, Limbird LE, Gilman AG (eds) Goodman and Gilmans's the pharmacological basis for therapeutics, 10th edn. McGraw-Hill, New York, pp 569–618
Hall FS, Sora I, Uhl GR (2001) Ethanol consumption and reward are decreased in μ-opiate receptor knockout mice. Psychopharmacology 154:43–49. doi:10.1007/s002130000622
Hand TH, Stinus L, Le Moal M (1989) Differential mechanisms in the acquisition and expression of heroin-induced place preference. Psychopharmacology 9:61–67
Herz A (1997) Endogenous opioid systems and alcohol addiction. Psychopharmacology 129:99–111
Introini-Collison IB, Baratti CM (1986) Opioid peptidergic systems modulate the activity of β-adrenergic mechanisms during memory consolidation processes. Behav Neural Biol 46:227–241. doi:10.1016/S0163-1047(86)90710-7
Itzhak Y, Anderson KL (2012) Changes in the magnitude of drug-unconditioned stimulus during conditioning modulate cocaine-induced place preference in mice. Addict Biol 17:706–716. doi:10.1111/j.1369-1600.2011.00334.x
Jonas DE, Amick HR, Feltner C, Bobashev G, Thomas K, Wines R et al (2014) Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis. JAMA 311:1889–1900. doi:10.1001/jama.2014.3628
Kuzmin A, Sandin J, Terenius L, Ogren SO (2003) Acquisition, expression, and reinstatement of ethanol-induced conditioned place preference in mice: effects of opioid receptor-like 1 receptor agonists and naloxone. J Pharmacol Exp Ther 304:310–318. doi:10.1124/jpet.102.041350
Lê AD, Poulos CX, Harding S et al (1999) Effects of naltrexone and fluoxetine on alcohol self-administration and reinstatement of alcohol seeking induced by priming injections of alcohol and exposure to stress. Neuropsychopharmacology 21:435–444. doi:10.1016/S0893-133X(99)00024-X
Liddie S, Itzhak Y (2016) Variations in the stimulus salience of cocaine reward influences drug-associated contextual memory. Addict Biol 21:242–254. doi:10.1111/adb.12191
Liu X, Weiss F (2002) Additive effect of stress and drug cues on reinstatement of ethanol seeking: exacerbation by history of dependence and role of concurrent activation of corticotropin-releasing factor and opioid mechanisms. J Neurosci 22:7856–7861
Mansour A, Burke S, Pavlic RJ et al (1996) Immunohistochemical localization of the cloned kappa 1 receptor in the rat CNS and pituitary. Neuroscience 71:671–690
Marinelli PW, Funk D, Harding S et al (2009) Roles of opioid receptor subtypes in mediating alcohol-seeking induced by discrete cues and context. Eur J Neurosci 30:671–679. doi:10.1111/j.1460-9568.2009.06851.x
McGaugh JL (2015) Consolidating memories. Annu Rev Psychol 66:1–24. doi:10.1146/annurev-psych-010814-014954
Mendez M, Morales-Mulia M (2008) Role of mu and delta opioid receptors in alcohol drinking behaviour. Curr Drug Abus Rev 1:239–252
Middaugh LD, Bandy ALE (2000) Naltrexone effects on ethanol consumption and response to ethanol conditioned cues in C57BL/6 mice. Psychopharmacology 151:321–327. doi:10.1007/s002130000479
Millan EZ, Marchant NJ, McNally GP (2011) Extinction of drug seeking. Behav Brain Res 217:454–462
Miranda R, Ray L, Blanchard A, Reynolds EK, Monti PM, Chun T et al (2014) Effects of naltrexone on adolescent alcohol cue reactivity and sensitivity: an initial randomized trial. Addict Biol 19:941–954. doi:10.1111/adb.12050
Monti PM, Rohsenow DJ, Hutchison KE, Swift RM, Mueller TI, Colby SM et al (1999) Naltrexone's effect on cue-elicited craving among alcoholics in treatment. Alcohol Clin Exp Res 23:1386–1394
Mucha R, Walker M (1987) Aversive property of opioid receptor blockade in drug-naive mice. Psychopharmacology 93:483–488. doi:10.1007/BF00207239
Myers KM, Carlezon WA (2010) Extinction of drug- and withdrawal-paired cues in animal models: relevance to the treatment of addiction. Neurosci Biobehav Rev 35:285–302
Neisewander JL, Pierce RC, Bardo MT (1990) Naloxone enhances the expression of morphine-induced conditioned place preference. Psychopharmacology 100:201–205. doi:10.1007/BF02244406
Norris JN, Pérez-Acosta AM, Ortega LA, Papini MR (2009) Naloxone facilitates appetitive extinction and eliminates escape from frustration. Pharmacol Biochem Behav 94:81–87. doi:10.1016/j.pbb.2009.07.012
Pastor R, Font L, Miquel M et al (2011) Involvement of the beta-endorphin neurons of the hypothalamic arcuate nucleus in ethanol-induced place preference conditioning in mice. Alcohol Clin Exp Res 35:2019–2029. doi:10.1111/j.1530-0277.2011.01553.x
Pavlov IP (1927) Conditioned reflexes: an investigation of the physiological activity of the cerebral cortex. Ann Neurosci 17:136–141
Pina MM, Cunningham CL (2014) Effects of dopamine receptor antagonists on the acquisition of ethanol-induced conditioned place preference in mice. Psychopharmacology 231:459–468. doi:10.1007/s00213-013-3252-0
Quirarte GL, Galvez R, Roozendaal B, McGaugh JL (1998) Norepinephrine release in the amygdala in response to footshock and opioid peptidergic drugs. Brain Res 808(2):134–140.
Quirk GJ, Mueller D (2008) Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 33:56–72. doi:10.1038/sj.npp.1301555
Rescorla RA (2001) Experimental extinction. In: Mowrer RR, Klein SB (eds) Handbook of contemporary learning theories. Erlbaum, Mahwah, pp 119–154
Rohsenow DJ, Monti PM, Hutchison KE, Swift RM, Colby SM, Kaplan GB (2000) Naltrexone's effects on reactivity to alcohol cues among alcoholic men. J Abnorm Psychol 109:738–742. doi:10.1037//0021-g43x.109.4.738
Roozendaal B, Lengvilas R, McGaugh JL et al (2007) Orphanin FQ/nociceptin interacts with the basolateral amygdala noradrenergic system in memory consolidation. Learn Mem 14:29–35. doi:10.1101/lm.403607
Sanchis-Segura C, Grisel JE, Olive MF et al (2005) Role of the endogenous opioid system on the neuropsychopharmacological effects of ethanol: new insights about an old question. Alcohol Clin Exp Res 29:1522–1527. doi:10.1097/01.alc.0000174913.60384.e8
Schroeder JP, Packard MG (2004) Facilitation of memory for extinction of drug-induced conditioned reward: role of amygdala and acetylcholine. Learn Mem 11:641–647. doi:10.1101/lm.78504
Shaham Y, Shalev U, Lu L et al (2003) The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology 168:3–20
Solecki W, Turek A, Kubik J, Przewlocki R (2009) Motivational effects of opiates in conditioned place preference and aversion paradigm—a study in three inbred strains of mice. Psychopharmacology 207:245–255. doi:10.1007/s00213-009-1672-7
Sudakov SK, Sotnikov SV, Chekmareva NY et al (2010) Changes in β-endorphin level in the cingulate cortex in rats after peripheral loperamide and methylnaloxone administration at rest and during emotional stress. Bull Exp Biol Med 149:167–169. doi:10.1007/s10517-010-0898-z
Taylor JR, Olausson P, Quinn JJ, Torregrossa MM (2009) Targeting extinction and reconsolidation mechanisms to combat the impact of drug cues on addiction. Neuropharmacology 56:186–195
Torregrossa MM, Taylor JR (2013) Learning to forget: manipulating extinction and reconsolidation processes to treat addiction. Psychopharmacology 226:659–672
Tseng A, Nguyen K, Hamid A et al (2013) The role of endogenous beta-endorphin and enkephalins in ethanol reward. Neuropharmacology 73:290–300. doi:10.1016/j.neuropharm.2013.06.001
Williams JT, Christie MJ, Manzoni O (2001) Cellular and synaptic adaptations mediating opioid dependence. Physiol Rev 81:299–343
Wróbel M (2011) Acquisition and expression of ethanol-induced conditioned place preference in mice is inhibited by naloxone. Pharmacol Reports 63:79–85
Young EA, Dreumont SE, Cunningham CL (2014) Role of nucleus accumbens dopamine receptor subtypes in the learning and expression of alcohol-seeking behavior. Neurobiol Learn Mem 108:28–37. doi:10.1016/j.nlm.2013.05.004
Acknowledgments
Research reported in this paper was supported by the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health under award number R01AA007702. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Font, L., Houck, C.A. & Cunningham, C.L. Naloxone effects on extinction of ethanol- and cocaine-induced conditioned place preference in mice. Psychopharmacology 234, 2747–2759 (2017). https://doi.org/10.1007/s00213-017-4672-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4672-z