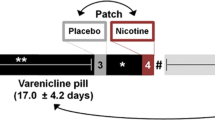
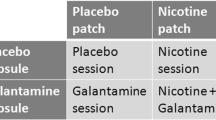
Abstract
Nicotine is a cholinergic agonist with known pro-cognitive effects in the domains of alerting and orienting attention. However, its effects on attentional top-down functions such as response inhibition and interference control are less well characterised. Here, we investigated the effects of 7 mg transdermal nicotine on performance on a battery of response inhibition and interference control tasks. A sample of N = 44 healthy adult non-smokers performed antisaccade, stop signal, Stroop, go/no-go, flanker, shape matching and Simon tasks, as well as the attentional network test (ANT) and a continuous performance task (CPT). Nicotine was administered in a within-subjects, double-blind, placebo-controlled design, with order of drug administration counterbalanced. Relative to placebo, nicotine led to significantly shorter reaction times on a prosaccade task and on CPT hits but did not significantly improve inhibitory or interference control performance on any task. Instead, nicotine had a negative influence in increasing the interference effect on the Simon task. Nicotine did not alter inter-individual associations between reaction times on congruent trials and error rates on incongruent trials on any task. Finally, there were effects involving order of drug administration, suggesting practice effects but also beneficial nicotine effects when the compound was administered first. Overall, our findings support previous studies showing positive effects of nicotine on basic attentional functions but do not provide direct evidence for an improvement of top-down cognitive control through acute administration of nicotine at this dose in healthy non-smokers.






Similar content being viewed by others
Notes
Further support comes from a re-analysis of our previously published data (Polner et al. 2015) from a sample of N = 440: There were significant correlations between congruent RT and incongruent/no-go error rate for the saccade (r = −.25, p < .001), flanker (r = −.18, p < .001) and go/no-go (r = −.46, p < .001) but not the Simon task (r = .05, p = .22). Stroop data from that study were not available on an individual trial level and were thus not included in this re-analysis.
References
AhnAllen CG, Nestor PG, Shenton ME et al (2008) Early nicotine withdrawal and transdermal nicotine effects on neurocognitive performance in schizophrenia. Schizophr Res 100:261–269. doi:10.1016/j.schres.2007.07.030
Barch DM, Braver TS, Carter CS et al (2009) CNTRICS final task selection: executive control. Schizophr Bull 35:115–135. doi:10.1093/schbul/sbn154
Bari A, Robbins TW (2013) Inhibition and impulsivity: behavioral and neural basis of response control. Prog Neurobiol 108:44–79
Barkley R (1997) Behavioral inhibition, sustained attention, and executive functions: constructing a unifying theory of ADHD. Psychol Bull 121:65–94. doi:10.1037/0033-2909.121.1.65
Barr RS, Culhane MA, Jubelt LE et al (2008) The effects of transdermal nicotine on cognition in nonsmokers with schizophrenia and nonpsychiatric controls. Neuropsychopharmacology 33:480–490. doi:10.1038/sj.npp.1301423
Beer AL, Vartak D, Greenlee MW (2013) Nicotine facilitates memory consolidation in perceptual learning. Neuropharmacology 64:443–451. doi:10.1016/j.neuropharm.2012.06.019
Beglinger LJ, Gaydos B, Tangphao-Daniels O et al (2005) Practice effects and the use of alternate forms in serial neuropsychological testing. Arch Clin Neuropsychol 20:517–529. doi:10.1016/j.acn.2004.12.003
Bekker EM, Böcker KBE, Van Hunsel F et al (2005) Acute effects of nicotine on attention and response inhibition. Pharmacol Biochem Behav 82:539–548. doi:10.1016/j.pbb.2005.10.009
Bogacz R, Wagenmakers EJ, Forstmann BU, Nieuwenhuis S (2010) The neural basis of the speed-accuracy tradeoff. Trends Neurosci 33:10–16
Bundesen C, Vangkilde S, Petersen A (2014) Recent developments in a computational theory of visual attention (TVA). Vis Res 116:1–9. doi:10.1016/j.visres.2014.11.005
Cornblatt BA, Risch NJ, Faris G et al (1988) The continuous performance test, identical pairs version (CPT-IP): I. New findings about sustained attention in normal families. Psychiatry Res 26:223–238. doi:10.1016/0165-1781(88)90076-5
Davidson DJ, Zacks RT, Williams CC (2003) Stroop interference, practice, and aging. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn 10:85–98. doi:10.1076/anec.10.2.85.14463
DeSchepper B, Treisman A (1996) Visual memory for novel shapes: implicit coding without attention. J Exp Psychol Learn Mem Cogn 22:27–47. doi:10.1037/0278-7393.22.1.27
Dinur-Klein L, Kertzman S, Rosenberg O et al (2014) Response inhibition and sustained and attention in heavy smokers versus non-smokers. Isr J Psychiatry Relat Sci 51:240–246
Dunbar K, MacLeod CM (1984) A horse race of a different color: Stroop interference patterns with transformed words. J Exp Psychol Hum Percept Perform 10:622–639. doi:10.1037/0096-1523.10.5.622
Easterbrook JA (1959) The effect of emotion on cue utilization and the organization of behavior. Psychol Rev 66:183–201. doi:10.1037/h0047707
Elliott R, Sahakian BJ, Matthews K et al (1997) Effects of methylphenidate on spatial working memory and planning in healthy young adults. Psychopharmacology 131:196–206. doi:10.1007/s002130050284
Ettinger U, Kumari V (2003) Pharmacological studies of smooth pursuit and antisaccade eye movements in schizophrenia: current status and directions for future research. Curr Neuropharmacol 1:285–300
Ettinger U, Kumari V, Crawford TJ et al (2003) Reliability of smooth pursuit, fixation, and saccadic eye movements. Psychophysiology 40:620–628
Ettinger U, Kumari V, Crawford TJ et al (2005) Saccadic eye movements, schizotypy, and the role of neuroticism. Biol Psychol 68:61–78. doi:10.1016/j.biopsycho.2004.03.014
Ettinger U, Williams SCRR, Patel D et al (2009) Effects of acute nicotine on brain function in healthy smokers and non-smokers: estimation of inter-individual response heterogeneity. NeuroImage 45:549–561. doi:10.1016/j.neuroimage.2008.12.029
Fan J, McCandliss BD, Sommer T et al (2002) Testing the efficiency and independence of attentional networks. J Cogn Neurosci 14:340–347. doi:10.1162/089892902317361886
Fan J, McCandliss BD, Fossella J et al (2005) The activation of attentional networks. NeuroImage 26:471–479. doi:10.1016/j.neuroimage.2005.02.004
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191. doi:10.3758/BF03193146
Foulds J, Stapleton J, Swettenham J et al (1996) Cognitive performance effects of subcutaneous nicotine in smokers and never-smokers. Psychopharmacology 127:31–38
Foulds J, Stapleton JA, Bell N et al (1997) Mood and physiological effects of subcutaneous nicotine in smokers and never-smokers. Drug Alcohol Depend 44:105–115. doi:10.1016/S0376-8716(96)01327-0
Franken IHA, van Strien JW, Kuijpers I (2010) Evidence for a deficit in the salience attribution to errors in smokers. Drug Alcohol Depend 106:181–185. doi:10.1016/j.drugalcdep.2009.08.014
Freedman R (2014) α7-nicotinic acetylcholine receptor agonists for cognitive enhancement in schizophrenia. Annu Rev Med 65:245–261. doi:10.1146/annurev-med-092112-142937
Friedman NP, Miyake A (2004) The relations among inhibition and interference control functions: a latent-variable analysis. J Exp Psychol Gen 133:101–135. doi:10.1037/0096-3445.133.1.101
Giessing C, Thiel CM, Alexander-Bloch AF et al (2013) Human brain functional network changes associated with enhanced and impaired attentional task performance. J Neurosci 33:5903–5914. doi:10.1523/JNEUROSCI.4854-12.2013
Goodman R (2014) Humility pills: building an ethics of cognitive enhancement. J Med Philos (United Kingdom) 39:258–278. doi:10.1093/jmp/jhu017
Gorsline J, Gupta SK, Dye D, Rolf CN (1993) Steady-state pharmacokinetics and dose relationship of nicotine delivered from Nicoderm (nicotine transdermal system). J Clin Pharmacol 33:161–168. doi:10.1002/j.1552-4604.1993.tb03938.x
Gorsline J, Okerholm RA, Rolf CN et al (1992) Comparison of plasma nicotine concentrations after application of nicoderm (nicotine transdermal system) to different skin sites. J Clin Pharmacol 32:576–581. doi:10.1177/009127009203200615
Hahn B (2015) Nicotinic receptors and attention. In: Current topics in behavioral neurosciences. pp 103–135
Heishman SJ, Kleykamp BA, Singleton EG (2010) Meta-analysis of the acute effects of nicotine and smoking on human performance. Psychopharmacology 210:453–469
Hommel B (2011) The Simon effect as tool and heuristic. Acta Psychol 136:189–202. doi:10.1016/j.actpsy.2010.04.011
Husain M, Mehta MA (2011) Cognitive enhancement by drugs in health and disease. Trends Cogn Sci 15:28–36
Hutton SB, Ettinger U (2006) The antisaccade task as a research tool in psychopathology: a critical review. Psychophysiology 43:302–313. doi:10.1111/j.1469-8986.2006.00403.x
Ishigami Y, Klein RM (2010) Repeated measurement of the components of attention using two versions of the Attention Network Test (ANT): stability, isolability, robustness, and reliability. J Neurosci Methods 190:117–128. doi:10.1016/j.jneumeth.2010.04.019
Javitt DC, Freedman R (2015) Sensory processing dysfunction in the personal experience and neuronal machinery of schizophrenia. Am J Psychiatry 172:17–31. doi:10.1176/appi.ajp.2014.13121691
Kambeitz J, la Fougère C, Werner N, Pogarell O, Riedel M, Falkai P, Ettinger U (2016) Nicotine-dopamine-transporter interactions during reward-based decision making. Eur Neuropsychopharmacol 26(6):938–947. doi:10.1016/j.euroneuro.2016.03.011
Kaufman LD, Pratt J, Levine B, Black SE (2010) Antisaccades: a probe into the dorsolateral prefrontal cortex in Alzheimer’s disease. A critical review J Alzheimers Dis 19:781–793. doi:10.3233/JAD-2010-1275
Kleykamp BA, Jennings JM, Blank MD, Eissenberg T (2005) The effects of nicotine on attention and working memory in never-smokers. Psychol Addict Behav 19:433–438. doi:10.1037/0893-164X.19.4.433
Kornblum S, Hasbroucq T, Osman A (1990) Dimensional overlap: cognitive basis for stimulus-response compatibility—a model and taxonomy. Psychol Rev 97:253–270. doi:10.1037/0033-295X.97.2.253
Kumari V, Postma P (2005) Nicotine use in schizophrenia: the self medication hypotheses. Neurosci Biobehav Rev 29:1021–1034. doi:10.1016/j.neubiorev.2005.02.006
Lecrubier Y, Sheehan DV, Weiller E et al (1997) The Mini International Neuropsychiatric Interview (MINI). A short diagnostic structured interview: reliability and validity according to the CIDI. Eur Psychiatry 12:224–231. doi:10.1016/S0924-9338(97)83296-8
Lehrl S, Triebig G, Fischer B (1995) Multiple choice vocabulary test MWT as a valid and short test to estimate premorbid intelligence. Acta Neurol Scand 91:335–345. doi:10.1111/j.1600-0404.1995.tb07018.x
Lindgren M, Stenberg G, Rosén I (1996) Effects of nicotine in visual attention tasks. Hum Psychopharmacol Clin Exp 11:47–51. doi:10.1002/(SICI)1099-1077(199601)11:1<47::AID-HUP743>3.0.CO;2-1
Luijten M, van Meel CS, Franken IHA (2011) Diminished error processing in smokers during smoking cue exposure. Pharmacol Biochem Behav 97:514–520. doi:10.1016/j.pbb.2010.10.012
Mancuso G, Warburton DM, Mélen M et al (1999) Selective effects of nicotine on attentional processes. Psychopharmacology 146:199–204
Massen C (2004) Parallel programming of exogenous and endogenous components in the antisaccade task. Q J Exp Psychol A 57:475–498. doi:10.1080/02724980343000341
Mehta M (2002) Where do we go from here? The importance of initial values. Neuropsychopharmacology 27:879–880. doi:10.1016/S0893-133X(02)00359-7
Meyhöfer I, Bertsch K, Esser M, Ettinger U (2015) Variance in saccadic eye movements reflects stable traits. Psychophysiology. doi:10.1111/psyp.12592
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24:167–202
Miyake A, Friedman NP (2012) The nature and organization of individual differences in executive functions: four general conclusions. Curr Dir Psychol Sci 21:8–14. doi:10.1177/0963721411429458
Miyake A, Friedman NP, Emerson MJ et al (2000) The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: a latent variable analysis. Cogn Psychol 41:49–100. doi:10.1006/cogp.1999.0734
Myers CS, Taylor RC, Salmeron BJ et al (2013) Nicotine enhances alerting, but not executive, attention in smokers and nonsmokers. Nicotine Tob Res 15:277–281. doi:10.1093/ntr/nts108
Newhouse PA, Potter A, Singh A (2004) Effects of nicotinic stimulation on cognitive performance
Newhouse PA, Potter AS, Dumas JA, Thiel CM (2011) Functional brain imaging of nicotinic effects on higher cognitive processes. In: Biochemical pharmacology. pp 943–951
Noorani I, Carpenter RHS (2013) Antisaccades as decisions: LATER model predicts latency distributions and error responses. Eur J Neurosci 37:330–338. doi:10.1111/ejn.12025
Norris H (1971) The action of sedatives on brain stem oculomotor systems in man. Neuropharmacology 10:181–191. doi:10.1016/0028-3908(71)90039-6
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113. doi:10.1016/0028-3932(71)90067-4
Perkins KA, Jetton C, Keenan J (2003) Common factors across acute subjective effects of nicotine. Nicotine Tob Res 5:869–875. doi:10.1080/14622200310001614629
Petrovsky N, Ettinger U, Quednow BB et al (2012) Nicotine differentially modulates antisaccade performance in healthy male non-smoking volunteers stratified for low and high accuracy. Psychopharmacology 221:27–38. doi:10.1007/s00213-011-2540-9
Petrovsky N, Ettinger U, Kessler H et al (2013a) The effect of nicotine on sensorimotor gating is modulated by a CHRNA3 polymorphism. Psychopharmacology 229:31–40. doi:10.1007/s00213-013-3081-1
Petrovsky N, Ettinger U, Quednow BB et al (2013b) Nicotine enhances antisaccade performance in schizophrenia patients and healthy controls. Int J Neuropsychopharmacol 16:1473–1481. doi:10.1017/S1461145713000011
Pierce JE, McDowell JE (2016) Modulation of cognitive control levels via manipulation of saccade trial-type probability assessed with event-related BOLD fMRI. J Neurophysiol 115:763–772. doi:10.1152/jn.00776.2015
Polner B, Aichert DS, Macare C et al (2015) Gently restless: association of ADHD-like traits with response inhibition and interference control. Eur Arch Psychiatry Clin Neurosci 265:689–699. doi:10.1007/s00406-014-0531-7
Portaccio E, Goretti B, Zipoli V et al (2010) Reliability, practice effects, and change indices for Rao’s Brief Repeatable Battery. Mult Scler 16:611–617. doi:10.1177/1352458510362818
Posner MI, Petersen SE (1990) The attention system of the human brain. Annu Rev Neurosci 13:25–42. doi:10.1146/annurev.ne.13.030190.000325
Potter AS, Newhouse PA (2004) Effects of acute nicotine administration on behavioral inhibition in adolescents with attention-deficit/hyperactivity disorder. Psychopharmacology 176:182–194. doi:10.1007/s00213-004-1874-y
Potter AS, Newhouse PA, Bucci DJ (2006) Central nicotinic cholinergic systems: a role in the cognitive dysfunction in attention-deficit/hyperactivity disorder? Behav Brain Res 175:201–211
Potter AS, Newhouse PA (2008) Acute nicotine improves cognitive deficits in young adults with attention-deficit/hyperactivity disorder. Pharmacol Biochem Behav 88:407–417. doi:10.1016/j.pbb.2007.09.014
Potter AS, Bucci DJ, Newhouse PA (2012) Manipulation of nicotinic acetylcholine receptors differentially affects behavioral inhibition in human subjects with and without disordered baseline impulsivity. Psychopharmacology 220:331–340. doi:10.1007/s00213-011-2476-0
Provost SC, Woodward R (1991) Effects of nicotine gum on repeated administration of the Stroop test. Psychopharmacology 104:536–540. doi:10.1007/BF02245662
Raghunathan TE, Rosenthal R, Rubin DB (1996) Comparing correlated but nonoverlapping correlations. Psychol Methods 1:178–183. doi:10.1037/1082-989X.1.2.178
Reilly JL, Lencer R, Bishop JR et al (2008) Pharmacological treatment effects on eye movement control. Brain Cogn 68:415–435. doi:10.1016/j.bandc.2008.08.026
Ridderinkhof KR, Scheres A, Oosterlaan J, Sergeant JA (2005) Delta plots in the study of individual differences: new tools reveal response inhibition deficits in AD/Hd that are eliminated by methylphenidate treatment. J Abnorm Psychol 114:197–215. doi:10.1037/0021-843X.114.2.197
Rubia K, Smith A, Taylor E (2007) Performance of children with attention deficit hyperactivity disorder (ADHD) on a test battery of impulsiveness. Child Neuropsychol 13:276–304. doi:10.1080/09297040600770761
Schaefer J, Giangrande E, Weinberger DR, Dickinson D (2013) The global cognitive impairment in schizophrenia: consistent over decades and around the world. Schizophr Res 150:42–50
Schlam TR, Japuntich SJ, Piper ME et al (2011) Cognitive conflict following appetitive versus negative cues and smoking cessation failure. Psychopharmacology 214:603–616. doi:10.1007/s00213-010-2063-9
Schmechtig A, Lees J, Grayson L et al (2013) Effects of risperidone, amisulpride and nicotine on eye movement control and their modulation by schizotypy. Psychopharmacology 227:331–345. doi:10.1007/s00213-013-2973-4
Simon JR, Small AM (1969) Processing auditory information: interference from an irrelevant cue. J Appl Psychol 53:433–435. doi:10.1037/h0028034
Smucny J, Olincy A, Eichman LS, Tregellas JR (2015) Neuronal effects of nicotine during auditory selective attention. Psychopharmacology 232:2017–2028. doi:10.1007/s00213-014-3832-7
Suter TW, Buzzi R, Woodson PP, Bättig K (1983) Psychophysiological correlates of conflict solving and cigarette smoking. Act Nerv Super (Praha) 25:261–272
Talanow T, Kasparbauer A-M, Steffens M et al (2016) Facing competition: neural mechanisms underlying parallel programming of antisaccades and prosaccades. Brain Cogn 107:37–47. doi:10.1016/j.bandc.2016.05.006
Thienel R, Voss B, Kellermann T et al (2009) Nicotinic antagonist effects on functional attention networks. Int J Neuropsychopharmacol 12:1295–1305. doi:10.1017/S1461145709990551
van den Wildenberg WPM, Wylie SA, Forstmann BU et al (2010) To head or to heed? Beyond the surface of selective action inhibition: a review. Front Hum Neurosci 4:222. doi:10.3389/fnhum.2010.00222
Vangkilde S, Bundesen C, Coull JT (2011) Prompt but inefficient: nicotine differentially modulates discrete components of attention. Psychopharmacology 218:667–680. doi:10.1007/s00213-011-2361-x
Wesnes K, Revell A (1984) The separate and combined effects of scopolamine and nicotine on human information processing. Psychopharmacology 84:5–11
Wignall ND, De Wit H (2011) Effects of nicotine on attention and inhibitory control in healthy nonsmokers. Exp Clin Psychopharmacol 19:183–191. doi:10.1037/a0023292
Wöstmann NM, Aichert DS, Costa A et al (2013) Reliability and plasticity of response inhibition and interference control. Brain Cogn 81:82–94. doi:10.1016/j.bandc.2012.09.010
Yerkes RM, Dodson JD (1908) The relation of strength of stimulus to rapidity of habit-formation. J Comp Neurol Psychol 18:459–482. doi:10.1037/h0073415
Yin J, Yuan K, Feng D et al (2015) Inhibition control impairments in adolescent smokers: electrophysiological evidence from a Go/NoGo study. Brain Imaging Behav. doi:10.1007/s11682-015-9418-0
Acknowledgements
We would like to thank Isabel Dietrich, Hannah-Christine Faber, Alena Grimm and Moritz Esser for assistance with data collection. Nadine Petrovsky was supported by a Gielen-Leyendecker Fellowship. Veena Kumari gratefully acknowledges a Humboldt Research Award from the Humboldt Foundation. The study received no further grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ulrich Ettinger, Eliana Faiola, Anna Kasparbauer, Nadine Petrovsky, Raymond Chan, Roman Liepelt and Veena Kumari declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 310 kb)
Rights and permissions
About this article
Cite this article
Ettinger, U., Faiola, E., Kasparbauer, AM. et al. Effects of nicotine on response inhibition and interference control. Psychopharmacology 234, 1093–1111 (2017). https://doi.org/10.1007/s00213-017-4542-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4542-8