Abstract
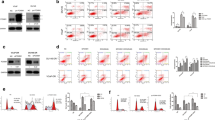
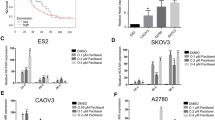
Extra spindle-polar body like 1 (ESPL1) is associated with the development of a variety of cancers, including bladder cancer, and is closely related to chemoresistance. In this study, we aimed to reveal the role of ESPL1 in bladder cancer progression and cisplatin (DDP) resistance. First, ESPL1 was found to be highly expressed in tumor tissues and cells of bladder cancer, and more highly expressed in cisplatin resistant tumor tissues or cells. The binding of PAX2 in ESPL1 promoter region was predicted by Jaspar database and verified by Ch-IP analysis and the luciferase reporter gene assay. Next, cisplatin-resistant T24 cells (T24/DDP) were established and transfected with ESPL1 siRNA (si-ESPL1) or overexpression vector (pcDNA-ESPL1) or co-transfected with PAX2 siRNA (si-PAX2) or overexpression vector (pcDNA-PAX2), and then treated with DDP or AG490, an inhibitor of JAK2. The results showed that silencing ESPL1 significantly reduced T24/DDP cell viability, colony formation and invasion, enhanced sensitivity to DDP, and induced cell apoptosis. Silencing PAX2 decreased ESPL1 expression, enhanced sensitivity to DDP, and induced apoptosis of T24/DDP cells, and inhibited activation of JAK2/STAT3 pathway. Overexpressing ESPL1 reversed the effect of PAX2 silencing on T24/DDP cells, while AG490 counteracted the reversal effect of overexpressing ESPL1. Finally, a xenograft tumor model was established and found that silencing ESPL1 or DDP treatment inhibited tumor growth, while silencing ESPL1 combined with DDP treatment had the best effect. In summary, this study suggested that PAX2-mediated ESPL1 transcriptional activation enhanced cisplatin resistance in bladder cancer by activating JAK2/STAT3 pathway.







Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Aier I, Semwal R, Dhara A, Sen N, Varadwaj PK (2019) An integrated epigenome and transcriptome analysis identifies PAX2 as a master regulator of drug resistance in high grade pancreatic ductal adenocarcinoma. PLoS One 14(10):e0223554
Aier I, Semwal R, Raj U, Varadwaj PK (2021) Comparative modeling and structure based drug repurposing of PAX2 transcription factor for targeting acquired chemoresistance in pancreatic ductal adenocarcinoma. J Biomol Struct Dyn 39(6):2071–2078
Bagratuni T, Mavrianou N, Gavalas NG, Tzannis K, Arapinis C, Liontos M, Christodoulou MI, Thomakos N, Haidopoulos D, Rodolakis A, Kastritis E, Scorilas A, Dimopoulos MA, Bamias A (2020) JQ1 inhibits tumour growth in combination with cisplatin and suppresses JAK/STAT signalling pathway in ovarian cancer. Eur J Cancer (Oxford, England: 1990) 126:125–135
Chen Q, Hu J, Deng J, Fu B, Guo J (2019) Bioinformatics analysis identified key molecular changes in bladder cancer development and recurrence. Biomed Res Int 2019:3917982
Chestukhin A, Pfeffer C, Milligan S, DeCaprio JA, Pellman D (2003) Processing, localization, and requirement of human separase for normal anaphase progression. Proc Natl Acad Sci USA 100(8):4574–4579
Colina JA, Varughese P, Karthikeyan S, Salvi A, Modi DA, Burdette JE (2020) Reduced PAX2 expression in murine fallopian tube cells enhances estrogen receptor signaling. Carcinogenesis 41(5):646–655
Deutsch U, Dressler GR, Gruss P (1988) Pax 1, a member of a paired box homologous murine gene family, is expressed in segmented structures during development. Cell 53(4):617–625
Gui T, Yao C, Jia B, Shen K (2021) Identification and analysis of genes associated with epithelial ovarian cancer by integrated bioinformatics methods. PLoS One 16(6):e0253136
Hu B, Wei LU, Liang H, Su M, Wang R, Su T, Li Q, Yin Q, Feng Y, Jiang J (2023) Correlation between serum ESPL1 and hepatitis B virus-related hepatocellular carcinoma histological grade: a Chinese single-center case-control study. Anticancer Res 43(9):3997–4005
Hueber PA, Waters P, Clark P, Eccles M, Goodyer P (2006) PAX2 inactivation enhances cisplatin-induced apoptosis in renal carcinoma cells. Kidney Int 69(7):1139–1145
Jiang L, Siu MK, Wong OG, Tam KF, Lu X, Lam EW, Ngan HY, Le XF, Wong ES, Monteiro LJ, Chan HY, Cheung AN (2011) iASPP and chemoresistance in ovarian cancers: effects on paclitaxel-mediated mitotic catastrophe. Clin Cancer Res 17(21):6924–6933
Kumar R (2017) Separase: Function beyond cohesion cleavage and an emerging oncogene. J Cell Biochem 118(6):1283–1299
Lee SB, Doberstein K, Baumgarten P, Wieland A, Ungerer C, Bürger C, Hardt K, Boehncke WH, Pfeilschifter J, Mihic-Probst D, Mittelbronn M, Gutwein P (2011) PAX2 regulates ADAM10 expression and mediates anchorage-independent cell growth of melanoma cells. PLoS One 6(8):e22312
Liu P, Gao Y, Huan J, Ge X, Tang Y, Shen W, Tian Y, Shen W, Zou S, Zhou J, Zhang S (2015) Upregulation of PAX2 promotes the metastasis of esophageal cancer through interleukin-5. Cell Physiol Biochem 35(2):740–754
Liu Z, Lian X, Zhang X, Zhu Y, Zhang W, Wang J, Wang H, Liu B, Ren Z, Zhang M, Liu M, Gao Y (2021) ESPL1 Is a novel prognostic biomarker associated with the malignant features of glioma. Front Genet 12:666106
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods (San Diego, Calif.) 25(4):402–8
Mo ZT, Li WN, Zhai YR, Gao SY (2017) The effects of icariin on the expression of HIF-1α, HSP-60 and HSP-70 in PC12 cells suffered from oxygen-glucose deprivation-induced injury. Pharm Biol 55(1):848–852
Mukherjee M, Ge G, Zhang N, Edwards DG, Sumazin P, Sharan SK, Rao PH, Medina D, Pati D (2014) MMTV-Espl1 transgenic mice develop aneuploid, estrogen receptor alpha (ERα)-positive mammary adenocarcinomas. Oncogene 33(48):5511–5522
Ojha R, Singh SK, Bhattacharyya S (2016) JAK-mediated autophagy regulates stemness and cell survival in cisplatin resistant bladder cancer cells. Biochim Biophys Acta 1860(11 Pt A):2484–2497
Richters A, Aben KKH, Kiemeney L (2020) The global burden of urinary bladder cancer: an update. World J Urol 38(8):1895–1904
Schöckel L, Möckel M, Mayer B, Boos D, Stemmann O (2011) Cleavage of cohesin rings coordinates the separation of centrioles and chromatids. Nat Cell Biol 13(8):966–972
Shah JB, McConkey DJ, Dinney CP (2011) New strategies in muscle-invasive bladder cancer: on the road to personalized medicine. Clin Cancer Res 17(9):2608–2612
Shen M, Xu Z, Xu W, Jiang K, Zhang F, Ding Q, Xu Z, Chen Y (2019) Inhibition of ATM reverses EMT and decreases metastatic potential of cisplatin-resistant lung cancer cells through JAK/STAT3/PD-L1 pathway. J Exp Clin Cancer Res: CR 38(1):149
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA A Cancer J Clin 70(1):7–30
Song R, Huang J, Yang C, Li Y, Zhan G, Xiang B (2022) ESPL1 is elevated in hepatocellular carcinoma and predicts prognosis. Int J Gen Med 15:8381–8398
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin 71(3):209–249
Ueda T, Ito S, Shiraishi T, Taniguchi H, Kayukawa N, Nakanishi H, Nakamura T, Naya Y, Hongo F, Kamoi K, Okihara K, Kawauchi A, Miki T (2015) PAX2 promoted prostate cancer cell invasion through transcriptional regulation of HGF in an in vitro model. Biochem Biophys Acta 1852(11):2467–2473
Wang G, Zhao W, Wang H, Qiu G, Jiang Z, Wei G, Li X (2019) Exosomal MiR-744 inhibits proliferation and sorafenib chemoresistance in hepatocellular carcinoma by targeting PAX2. Med Sci Monit 25:7209–7217
Wang R, Zang W, Hu B, Deng D, Ling X, Zhou H, Su M, Jiang J (2020) Serum ESPL1 can be used as a biomarker for patients with hepatitis B virus-related liver cancer: a Chinese case-control study. Technol Cancer Res Treat 19:1533033820980785
Witjes JA, Bruins HM, Cathomas R, Compérat EM, Cowan NC, Gakis G, Hernández V, Linares Espinós E, Lorch A, Neuzillet Y, Rouanne M, Thalmann GN, Veskimäe E, Ribal MJ, van der Heijden AG (2021) European Association of Urology guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2020 guidelines. Eur Urol 79(1):82–104
Zhang Y, Tseng JT, Lien IC, Li F, Wu W, Li H (2020) mRNAsi index: machine learning in mining lung adenocarcinoma stem cell biomarkers. Genes 11(3):257–286
Zhang N, Pati D (2017) Biology and insights into the role of cohesin protease separase in human malignancies. Biol Rev Camb Philos Soc 92(4):2070–2083
Zhu QC, Sun JH, Liang MJ, Zhang ZY, Xia Y (2022) PHF20L1 mediates PAX2 expression to promote angiogenesis and liver metastasis in colorectal cancer through regulating HIC1. Biol Chem 403(10):917–928
Acknowledgements
We appreciate all the members involved in this study.
Funding
This study was supported by grants from the Hospital National Natural Science Foundation Booster Fund (No. 2021ZTXM027) and National Natural Science Foundation of China (No. 82002687).
Author information
Authors and Affiliations
Contributions
All authors contributed to data analysis, drafting, and revising the article; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All authors contributed to the article and approved the submitted version. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethics approval
This study had gained the informed consent from each subject and been approved by the Medical Ethics Committee of Tangdu hospital.
Consent to participate
Each participant signed written informed consent before the study.
Consent for publication
All authors consent to publish this work.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Wang, Y., Tang, Q. et al. PAX2 mediated upregulation of ESPL1 contributes to cisplatin resistance in bladder cancer through activating the JAK2/STAT3 pathway. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-03061-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-03061-3