Abstract
It has been widely documented that medicinal herbal remedies are effective, have fewer side effects than conventional medicine, and have a synergistic effect on health collaborations in the fight against complicated diseases. Traditional treatments for neurological problems in ancient times sometimes involved the use of herbal remedies and conventional methods from East Asian countries including India, Japan, China, and Korea. We collected and reviewed studies on plant-derived neuroprotective drugs and tested them in neurotoxic models. Basic research, preclinical and clinical transgene research can benefit from in silico, in vitro, and in vivo investigations. Research, summaries of the extracts, fractions, and herbal ingredients were compiled from popular scientific databases, which were then examined according to origin and bioactivity. Given the complex and varied causes of neurodegeneration, it may be beneficial to focus on multiple mechanisms of action and a neuroprotection approach. This approach aims to prevent cell death and restore function to damaged neurons, offering promising strategies for preventing and treating neurodegenerative diseases. Neurodegenerative illnesses can potentially be treated with natural compounds that have been identified as neuroprotective agents. To gain deeper insights into the neuropharmacological mechanisms underlying the neuroprotective and therapeutic properties of naturally occurring antioxidant phytochemical compounds in diverse neurodegenerative diseases, this study aims to comprehensively review such compounds, focusing on their modulation of apoptotic markers such as caspase, Bax, Bcl-2, and proinflammatory markers. In addition, we delve into a range of efficacies of antioxidant phytochemical compounds as neuroprotective agents in animal models. They reduce the oxidative stress of the brain and have been shown to have anti-apoptotic effects. Many researches have demonstrated that plant extracts or bioactive compounds can fight neurodegenerative disorders. Herbal medications may offer neurodegenerative disease patients’ new treatments. This may be a cheaper and more culturally appropriate alternative to standard drugs for millions of people with age-related NDDs.




Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- PCD:
-
Programmed cell death
- Fas:
-
Fatty acid synthetase
- mFas:
-
Membrane fatty acid synthetase
- sFas:
-
Soluble fatty acid synthetase
- FasL:
-
Fatty acid synthetase ligand
- FasR:
-
Fatty acid synthetase receptor
- TNF:
-
Tumor necrosis factor
- TNFα:
-
Tumor necrosis factor-alpha
- TNFR1:
-
Tumor necrosis factor receptor 1
- TNFR2:
-
Tumor necrosis factor receptor 2
- APO-1:
-
Apoptosis antigen 1
- DISC:
-
Death-inducing signaling complex
- CD95:
-
Cluster of differentiation 95
- DD:
-
Death domain
- CD4:
-
Cluster of differentiation 4
- Bcl-2:
-
B cell lymphoma protein 2
- TRADD:
-
TNF receptor–associated death domain
- NF-kB:
-
NF-kappa B
- JNK:
-
C-Jun N-terminal kinase
- MOMP:
-
Mitochondrial outer membrane permeabilization
- SMAC:
-
Second mitochondrial activator of caspases
- DIABLO:
-
Direct IAP–binding protein with low PI
- HtrA2:
-
High-temperature requirement
- AIF:
-
Apoptosis-inducing factor
- CAD:
-
Caspase-activated DNase
- Bcl-10:
-
B cell lymphoma protein 10
- BAX:
-
BCL2-associated X protein
- BAK:
-
BCL2 antagonist killer 1
- BID:
-
BH3-interacting domain death agonist
- BAD:
-
BCL2 antagonist of cell death
- BIM:
-
BCL2-interacting protein BIM
- BIK:
-
BCL2-interacting killer
- BLK:
-
Bik-like killer protein
- Bcl-X:
-
BCL2 like 1
- Bcl-XL:
-
BCL2-related protein, long isoform
- Bcl-XS:
-
BCL2-related protein, short isoform
- Bcl-W:
-
BCL2 like 2 proteins
- BAG:
-
BCL2-associated athanogene
- Puma:
-
BCL2-binding component 3
- Noxa:
-
Phorbol-12-myristate-13-acetate–induced protein 1
- Caspase-9:
-
Cysteinyl aspartic acid-protease-9
- UPR:
-
Unfolded protein response
- ER:
-
Endoplasmic reticulum
- IP3R:
-
1,4,5-Triphosphate receptor
- RyR:
-
Ryanodine receptor
- LMP:
-
Lysosomal membrane permeabilization
- AD:
-
Alzheimer’s disease
- PD:
-
Parkinson’s disease
- HD:
-
Huntington’s disease
- ALS:
-
Amyotrophic lateral sclerosis
- Cyto-C:
-
Cytochrome C
- LPS:
-
Lipopolysaccharide
- COX-2:
-
Cycloxygenase-2
- IL-1:
-
Interluekin-1
- IL-6:
-
Interluekin-6
- MAPK:
-
Mitogen-activated protein kinase
- AA:
-
Asiatic acid
- MMP:
-
Mitochondrial matrix metalloproteinase
- NLRP3:
-
NLR family pyrin domain containing 3
- GSH:
-
Glutathione
- SOD:
-
Superoxide dismutase
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- CA1:
-
Cornu ammonis 1
- CA3:
-
Cornu ammonis 3
- DA:
-
Dopamine
- FA:
-
Ferulic acid
- CAT:
-
Catalase
- GPx:
-
Glutathione peroxidase
- 6-OHDA:
-
6-Hydroxydopamine hydrobromide
- DRP-1:
-
Dynamin-related protein 1
- PGC1:
-
Peroxisome proliferator–activated receptor-gamma coactivator
- MFN-2:
-
Mutofusin-2
- HSP-70:
-
70 Kilodalton heat shock proteins
- TH:
-
Thyroxine
- TQ:
-
Thymoquinone
- IFN:
-
Interferon
- EA:
-
Ellagic acid
- MDA:
-
Malondialdehyde
- NRF-2:
-
Nuclear factor erythroid 2–related factor 2
- CA:
-
Caffeic acid
- CAPE:
-
Caffeine phenethyl ester
- RNS:
-
Reactive nitrogen species
- AKT:
-
Protein kinase B
- AIF:
-
Apoptosis-inducing factor
- MAO-B:
-
Monoamine oxidase B
- EGCG:
-
Epigallocatechin-3-gallate
- PQ:
-
Paraquat
- Caspase-3:
-
Cysteinyl aspartic acid-protease-3
- NDD:
-
Neurodegenerative disorders
- PERK:
-
Protein kinase RNA-like endoplasmic reticulum kinase
- CHOP:
-
C/EBP homologous protein
- TF:
-
Theaflavin
- GBE:
-
Gingko biloba leaf extract
- ERK1/2:
-
Extracellular signal–regulated protein kinase
- SF:
-
Sodium ferulate
- Aβ:
-
Amyloid beta
- SIRT1:
-
Sirtuin 1
- STZ:
-
Streptozocin
- TLR4:
-
Toll-like receptor 4
- BIM:
-
Bcl-2-like protein 11
- GADD45:
-
Growth arrest and DNA damage–inducible 45 proteins
- CA:
-
Centella asiatica
- CB:
-
Cannabinoid
- 9-THC:
-
(9)-Tetrahydrocannabinol
- CBD:
-
Cannabidiol
- GABA:
-
Gamma-aminobutyric acid
References
Abdulwanis Mohamed Z et al (2019) Neuroprotective potential of secondary metabolites from Melicope lunu-ankenda (Rutaceae). Molecules 24(17):3109
Ajayi SA et al (2011) The microstructural effects of aqueous extract of Garcinia kola (Linn) on the hippocampus and cerebellum of malnourished mice. Asian Pac J Trop Biomed 1(4):261–265
Akbar M et al (2016) Mitochondrial dysfunction and cell death in neurodegenerative diseases through nitroxidative stress. Brain Res 1637:34–55
Al-Amri JS et al (2013) Effect of epigallocatechin-3-gallate on inflammatory mediators release in LPS-induced Parkinson’s disease in rats. Indian J Exp Biol 51(5):357–62
Allen RT et al (1997) Morphological and biochemical characterization and analysis of apoptosis. J Pharmacol Toxicol Methods 37(4):215–228
Ameisen JC (2002) On the origin, evolution, and nature of programmed cell death: a timeline of four billion years. Cell Death Differ 9(4):367–393
Anand A et al (2019) Vanillin: a comprehensive review of pharmacological activities. Plant Arch 19(2):1000–1004
Anandhan A et al (2013a) Therapeutic attenuation of neuroinflammation and apoptosis by black tea theaflavin in chronic MPTP/probenecid model of Parkinson’s disease. Neurotox Res 23(2):166–173
Ardah MT et al (2020) Ellagic acid prevents dopamine neuron degeneration from oxidative stress and neuroinflammation in MPTP model of Parkinson’s disease. Biomolecules 10(11):1519
Asadian G et al (2011) Study of variation of biochemical components inhypericum perforatum l. grown in north of Iran. J Med Plants Res 6(3)
Association A (2018) 2018 Alzheimer’s disease facts and figures. Alzheimer’s & Dementia 14(3):367–429
Babbar R et al (2021) A comprehensive review on therapeutic applications of ferulic acid and its novel analogues: a brief literature. Mini Rev Med Chem 21(12):1578–1593
Balakrishnan R et al (2021) Natural phytochemicals as novel therapeutic strategies to prevent and treat Parkinson’s disease: current knowledge and future perspectives. Oxid Med Cell Longev 25:2021:6680935
Baluchnejadmojarad T et al (2017) Ellagic acid exerts protective effect in intrastriatal 6-hydroxydopamine rat model of Parkinson’s disease: possible involvement of ERβ/Nrf2/HO-1 signaling. Brain Res 1662:23–30
Barone E et al (2009) Ferulic acid and its therapeutic potential as a hormetin for age-related diseases. Biogerontology 10(2):97–108
Bian D et al (2012) Madecassoside, a triterpenoid saponin isolated from Centella asiatica herbs, protects endothelial cells against oxidative stress. J Biochem Mol Toxicol 26(10):399–406
Binawade Y, Jagtap A (2013) Neuroprotective effect of lutein against 3-nitropropionic acid–induced Huntington’s disease–like symptoms: possible behavioral, biochemical, and cellular alterations. J Med Food 16(10):934–943
Blamire A et al (2000) Interleukin-1β-induced changes in blood–brain barrier permeability, apparent diffusion coefficient, and cerebral blood volume in the rat brain: a magnetic resonance study. J Neurosci 20(21):8153–8159
Boatright KM, Salvesen GS (2003) Mechanisms of caspase activation. Curr Opin Cell Biol 15(6):725–731
Borase HP et al (2022) Design and evaluation of natural deep eutectic solvents system for chrysin to elicit its solubility, stability, and bioactivity. J Mol Liq 345:118205
Broderick TL et al (2020) Neuroprotective effects of chronic resveratrol treatment and exercise training in the 3xTg-AD mouse model of Alzheimer’s disease. Int J Mol Sci 21(19)
Buba CI et al (2016) Garcinia kola: the phytochemistry, pharmacology and therapeutic applications. Int J Pharmacognosy 3(2):67–81
Burz C et al (2009) Apoptosis in cancer: key molecular signaling pathways and therapy targets. Acta Oncol 48(6):811–821
Carson MJ et al (2006) CNS immune privilege: hiding in plain sight. Immunol Rev 213(1):48–65
Chandran R, Abrahamse H (2020) Identifying plant-based natural medicine against oxidative stress and neurodegenerative disorders. Oxid Med Cell Longev 2020:8648742
Chaudhary P et al (2011) Lipoic acid decreases inflammation and confers neuroprotection in experimental autoimmune optic neuritis. J Neuroimmunol 233(1–2):90–96
Chaudhary A et al (2019) Ferulic acid: a promising therapeutic phytochemical and recent patents advances. Recent Pat Inflamm Allergy Drug Discov 13(2):115–123
Chen S et al (2014) Celastrol prevents cadmium-induced neuronal cell death via targeting JNK and PTEN-Akt/mTOR network. J Neurochem 128(2):256–266
Chen D et al (2019) Asiatic acid protects dopaminergic neurons from neuroinflammation by suppressing mitochondrial ROS production. Biomol Ther 27(5):442
Chongtham A, Agrawal N (2016) Curcumin modulates cell death and is protective in Huntington’s disease model. Sci Rep 6(1):1–10
Choudhury B (2015) Approach to neurological disorder in Ayurveda. Ind J Med Res and Pharm Sci 2:69–73
Clifford MN (1999) Chlorogenic acids and other cinnamates–nature, occurrence and dietary burden. J Sci Food Agric 79(3):362–372
Colín-González AL et al (2012) The antioxidant mechanisms underlying the aged garlic extract-and S-allylcysteine-induced protection. Oxid Med Cell Longev 2012:907162
Cowan KJ, Storey KB (2003) Mitogen-activated protein kinases: new signaling pathways functioning in cellular responses to environmental stress. J Exp Biol 206(7):1107–1115
Crespo-Bujosa HB, Gonzalez MJ (2018) Phytochemicals for the treatment of multiple sclerosis? A review of scientific evidence. J Orthomol Med 33(1):1–8
Cui J et al (2018) Neuroprotective effect of naringin, a flavone glycoside in quinolinic acid-induced neurotoxicity: possible role of PPAR-γ, Bax/Bcl-2, and caspase-3. Food Chem Toxicol 121:95–108
D’Arcy MS (2019) Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int 43(6):582–592
David AVA et al (2016) Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev 10(20):84
DeMaagd G, Philip A (2015) Parkinson’s disease and its management: part 1: disease entity, risk factors, pathophysiology, clinical presentation, and diagnosis. Pharmacy and Therapeutics 40(8):504
Diamant G (1829) Dikstein R (2013) Transcriptional control by NF-κB: elongation in focus. Biochim Biophys Acta 9:937–945
Dinc E et al (2017) Protective effect of combined caffeic acid phenethyl ester and bevacizumab against hydrogen peroxide-induced oxidative stress in human RPE cells. Curr Eye Res 42(12):1659–1666
Doonan F, Cotter TG (2008) Morphological assessment of apoptosis. Methods 44(3):200–204
Dyment DA et al (2004) Genetics of multiple sclerosis. The Lancet Neurology 3(2):104–110
Ebrahimi SS et al (2017) Thymoquinone exerts neuroprotective effect in animal model of Parkinson’s disease. Toxicol Lett 276:108–114
Ekowati J et al (2020) Ferulic acid prevents angiogenesis through cyclooxygenase-2 and vascular endothelial growth factor in the chick embryo chorioallantoic membrane model. Turk J Pharm Sci 17(4):424–431
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35(4):495–516
Ernanin Dyah W et al (2021) Virtual prediction of purple rice ferulic acid as anti-inflammatory of TNF-α signaling. Berkala Penelitian Hayati 27(2):59–66
Farkhondeh T et al (2019) Chrysin attenuates inflammatory and metabolic disorder indices in aged male rat. Biomed Pharmacother 109:1120–1125
Farquhar MG, Palade GE (1965) Cell junctions in amphibian skin. J Cell Biol 26(1):263–291
Fei C et al (2013) Ginkgo biloba extract protects substantia nigral neurons from apoptosis in PD rat model. J Neurol Disord 1(118):2
Ficarra S et al (2016) Insights into the properties of the two enantiomers of trans-δ-viniferin, a resveratrol derivative: antioxidant activity, biochemical and molecular modeling studies of its interactions with hemoglobin. Mol BioSyst 12(4):1276–1286
Finucane DM et al (1999) Bax-induced caspase activation and apoptosis via cytochromec release from mitochondria is inhibitable by Bcl-xL. J Biol Chem 274(4):2225–2233
Firdaus F et al (2018) Ellagic acid attenuates arsenic induced neuro-inflammation and mitochondrial dysfunction associated apoptosis. Toxicol Rep 5:411–417
Fisher M et al (1987) Linoleic acid levels in white blood cells, platelets, and serum of multiple sclerosis patients. Acta Neurol Scand 76(4):241–245
Fleischer A et al (2006) Modulating apoptosis as a target for effective therapy. Mol Immunol 43(8):1065–1079
Fulda S et al (2010) Targeting mitochondria for cancer therapy. Nat Rev Drug Discovery 9(6):447–464
Fusar-Poli P et al (2009) Distinct effects of Δ9-tetrahydrocannabinol and cannabidiol on neural activation during emotional processing. Arch Gen Psychiatry 66(1):95–105
Galende AV et al (2021) Report by the Spanish Foundation of the Brain on the social impact of Alzheimer disease and other types of dementia. Neurología (english Edition) 36(1):39–49
Ghadially FN (2013) Ultrastructural pathology of the cell and matrix: a text and atlas of physiological and pathological alterations in the fine structure of cellular and extracellular components. Butterworth-Heinemann
Giatti S et al (2012) Neuroactive steroids, their metabolites, and neuroinflammation. J Mol Endocrinol 49(3):R125–R134
Gomes BAQ et al (2018) Neuroprotective mechanisms of resveratrol in Alzheimer’s disease: role of SIRT1. Oxid Med Cell Longev 2018:8152373
Gomez DR et al (2013) Neuroprotective properties of standardized extracts of Hypericum perforatum on rotenone model of Parkinson’s disease. CNS Neurol Disord Drug Targets 12(5):665–679
Granja A et al (2017) Therapeutic potential of epigallocatechin gallate nanodelivery systems. Biomed Res Int 2017:5813793
Gu X et al (2016) Protective effect of paeoniflorin on inflammation and apoptosis in the cerebral cortex of a transgenic mouse model of Alzheimer’s disease. Mol Med Rep 13(3):2247–2252
Guha M, Mackman N (2001) LPS induction of gene expression in human monocytes. Cell Signal 13(2):85–94
Gul S et al (1970) Fatty acid composition of phospholipids from platelets and erythrocytes in multiple sclerosis. J Neurol Neurosurg Psychiatry 33(4):506–510
Guo B et al (2016) Multifunction of chrysin in Parkinson’s model: anti-neuronal apoptosis, neuroprotection via activation of MEF2D, and inhibition of monoamine oxidase-B. J Agric Food Chem 64(26):5324–5333
Gupta A et al (2021) Ferulic acid-mediated modulation of apoptotic signaling pathways in cancer. Adv Protein Chem Struct Biol 125:215–257
Hanisch UK (2002) Microglia as a source and target of cytokines. Glia 40(2):140–155
Hanna DM et al (2015) ADIOL protects against 3-NP-induced neurotoxicity in rats: possible impact of its anti-oxidant, anti-inflammatory and anti-apoptotic actions. Prog Neuropsychopharmacol Biol Psychiatry 60:36–51
Harry GJ, Kraft AD (2012) Microglia in the developing brain: a potential target with lifetime effects. Neurotoxicology 33(2):191–206
Hashim P (2011) Centella asiatica in food and beverage applications and its potential antioxidant and neuroprotective effect. Int Food Res J 18(4):44056033
Hashimoto M, Hossain S (2011) Neuroprotective and ameliorative actions of polyunsaturated fatty acids against neuronal diseases: beneficial effect of docosahexaenoic acid on cognitive decline in Alzheimer’s disease. J Pharmacol Sci 116(2):150–162
He Z et al (2023) Protective effects of luteolin against amyloid beta-induced oxidative stress and mitochondrial impairments through peroxisome proliferator-activated receptor γ-dependent mechanism in Alzheimer’s disease. Redox Biol 66:102848
Hengartner MO (1997) Genetic control of programmed cell death and aging in the nematode Caenorhabditis elegans. Exp Gerontol 32(4–5):363–374
Horvitz HR (1994) Genetic control of programmed cell death in the nematode Caenorhabditis elegans. Springer, Apoptosis, pp 1–13
Hou RR et al (2008) Neuroprotective effects of (−)-epigallocatechin-3-gallate (EGCG) on paraquat-induced apoptosis in PC12 cells. Cell Biol Int 32(1):22–30
Huang W-C, Hung M-C (2013) Beyond NF-κB activation: nuclear functions of IκB kinase α. J Biomed Sci 20:1–13
Huang C-L et al (2011) Gastrodia elata prevents huntingtin aggregations through activation of the adenosine A2A receptor and ubiquitin proteasome system. J Ethnopharmacol 138(1):162–168
Imai T et al (2014) Protective effect of S-allyl-L-cysteine against endoplasmic reticulum stress-induced neuronal death is mediated by inhibition of calpain. Amino Acids 46:385–393
Janhom P, Dharmasaroja P (2015) Neuroprotective effects of alpha-mangostin on MPP+-induced apoptotic cell death in neuroblastoma SH-SY5Y cells. J Toxicol 2015:919058
Jena AB et al (2022) An in silico investigation on the interactions of curcumin and epigallocatechin-3-gallate with NLRP3 inflammasome complex. Biomed Pharmacother 156:113890
Jin Y et al (2008) Neuroprotective effect of sodium ferulate and signal transduction mechanisms in the aged rat hippocampus 1. Acta Pharmacol Sin 29(12):1399–1408
Johnson TV et al (2010) Neuroprotective effects of intravitreal mesenchymal stem cell transplantation in experimental glaucoma. Invest Ophthalmol vis Sci 51(4):2051–2059
Joseph J et al (2009) Nutrition, brain aging, and neurodegeneration. J Neurosci 29(41):12795–12801
Jung KK et al (2006) Inhibitory effect of curcumin on nitric oxide production from lipopolysaccharide-activated primary microglia. Life Sci 79(21):2022–2031
Kang C-H et al (2013) Quercetin inhibits lipopolysaccharide-induced nitric oxide production in BV2 microglial cells by suppressing the NF-κB pathway and activating the Nrf2-dependent HO-1 pathway. Int Immunopharmacol 17(3):808–813
Karimi A et al (2015) Herbal versus synthetic drugs; beliefs and facts. J Nephropharmacol 4(1):27
Kaushal V, Schlichter LC (2008) Mechanisms of microglia-mediated neurotoxicity in a new model of the stroke penumbra. J Neurosci 28(9):2221–2230
Khurana N, Gajbhiye A (2013) Ameliorative effect of Sida cordifolia in rotenone induced oxidative stress model of Parkinson’s disease. Neurotoxicology 39:57–64
Kim S-J et al (2007) Epigallocatechin-3-gallate suppresses NF-κB activation and phosphorylation of p38 MAPK and JNK in human astrocytoma U373MG cells. J Nutr Biochem 18(9):587–596
Kim IS et al (2011) Neuroprotective effects of vanillyl alcohol in Gastrodia elata Blume through suppression of oxidative stress and anti-apoptotic activity in toxin-induced dopaminergic MN9D cells. Molecules 16(7):5349–5361
Kirimlioglu V et al (2008) Effect of resveratrol on oxidative stress enzymes in rats subjected to 70% partial hepatectomy. Elsevier, Transplantation proceedings
Koch M et al (2006) Erythrocyte membrane fatty acids in benign and progressive forms of multiple sclerosis. J Neurol Sci 244(1–2):123–126
Koh S-H et al (2006) The effect of epigallocatechin gallate on suppressing disease progression of ALS model mice. Neurosci Lett 395(2):103–107
Kou X et al (2012) Ampelopsin inhibits H2O2-induced apoptosis by ERK and Akt signaling pathways and up-regulation of heme oxygenase-1. Phytother Res 26(7):988–994
Kumar P et al (2009a) Lycopene modulates nitric oxide pathways against 3-nitropropionic acid-induced neurotoxicity. Life Sci 85(19–20):711–718
Kumar P et al (2009b) Sesamol attenuate 3-nitropropionic acid-induced Huntington-like behavioral, biochemical, and cellular alterations in rats. J Asian Nat Prod Res 11(5):439–450
Kumar M et al (2021) Tomato (Solanum lycopersicum L.) seed: a review on bioactives and biomedical activities. Biomed Pharmacother 142:112018
Kyriakis JM, Avruch J (2001) Mammalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev 81(2):807–869
Lam PY, Ko KM (2012) Beneficial effect of (–) Schisandrin B against 3-nitropropionic acid-induced cell death in PC12 cells. BioFactors 38(3):219–225
Lambert JD, Elias RJ (2010) The antioxidant and pro-oxidant activities of green tea polyphenols: a role in cancer prevention. Arch Biochem Biophys 501(1):65–72
Legiawati L et al (2018) In silico study of Centella asiatica active compounds as anti-inflammatory agent by decreasing Il-1 and Il-6 activity, promoting Il-4 activity. J Pharm Sci Res 10(9):2142–2147
Li Y-H et al (2017) Antioxidant effects of celastrol against hydrogen peroxide-induced oxidative stress in the cell model of amyotrophic lateral sclerosis. Sheng Li Xue Bao 69(6):751–758
Li C et al (2018) Pinostrobin exerts neuroprotective actions in neurotoxin-induced Parkinson’s disease models through Nrf2 induction. J Agric Food Chem 66(31):8307–8318
Lv J et al (2018) Pharmacological review on asiatic acid and its derivatives: a potential compound. SLAS Technology 23(2):111–127
Lyman M et al (2014) Neuroinflammation: the role and consequences. Neurosci Res 79:1–12
Maianski NA et al (2003) Tumor necrosis factor α induces a caspase-independent death pathway in human neutrophils. Blood, J Am Soc Hematol 101(5):1987–1995
Majno G, Joris I (1995) Apoptosis, oncosis, and necrosis. An overview of cell death. Am J Pathol 146(1):3
Manavalan A et al (2012) Gastrodia elata Blume (Tianma) mobilizes neuro-protective capacities. Int J Biochem Mol Biol 3(2):219
Mandel S et al (2004) Cell signaling pathways in the neuroprotective actions of the green tea polyphenol (-)-epigallocatechin-3-gallate: implications for neurodegenerative diseases. J Neurochem 88(6):1555–1569
Marracci GH et al (2002) Alpha lipoic acid inhibits T cell migration into the spinal cord and suppresses and treats experimental autoimmune encephalomyelitis. J Neuroimmunol 131(1–2):104–114
Meesarapee B et al (2014) Curcumin I mediates neuroprotective effect through attenuation of quinoprotein formation, p-p38 MAPK expression, and caspase-3 activation in 6-hydroxydopamine treated SH-SY5Y cells. Phytother Res 28(4):611–616
Millington C et al (2014) Chronic neuroinflammation in Alzheimer’s disease: new perspectives on animal models and promising candidate drugs. Biomed Res Int 2014:309129
Ming LC (2016) Use of herbal products in Southeast Asian countries. Arch Pharm Pract 7(5):S1
Mir RH et al (2022) Plant-derived natural compounds for the treatment of amyotrophic lateral sclerosis: an update. Curr Neuropharmacol 20(1):179
Mir SM et al (2016) Omega-3 fatty acids in inflammatory diseases. Omega-3 Fatty Acids: Keys to Nutritional Health 141–155. https://doi.org/10.1007/978-3-319-40458-5
Moon D-O et al (2010) Rosmarinic acid sensitizes cell death through suppression of TNF-α-induced NF-κB activation and ROS generation in human leukemia U937 cells. Cancer Lett 288(2):183–191
Muñoz-Espín D, Serrano M (2014) Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 15(7):482–496
Musfiroh I et al (2023) Stability analysis of the asiatic acid-COX-2 complex using 100 ns molecular dynamic simulations and its selectivity against COX-2 as a potential anti-inflammatory candidate. Molecules 28(9):3762
Nabavi SF et al (2015) Luteolin as an anti-inflammatory and neuroprotective agent: a brief review. Brain Res Bull 119:1–11
Nagoor Meeran MF et al (2018) Pharmacological properties, molecular mechanisms, and pharmaceutical development of asiatic acid: a pentacyclic triterpenoid of therapeutic promise. Front Pharmacol 9:892
Naik E, Dixit VM (2011) Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J Exp Med 208(3):417–420
Nataraj J et al (2016) Lutein protects dopaminergic neurons against MPTP-induced apoptotic death and motor dysfunction by ameliorating mitochondrial disruption and oxidative stress. Nutr Neurosci 19(6):237–246
Naziroglu M et al (2014) Modulation of oxidative stress, apoptosis, and calcium entry in leukocytes of patients with multiple sclerosis by Hypericum perforatum. Nutr Neurosci 17(5):214–221
Nicholas R, Rashid W (2013) Multiple sclerosis. Am Fam Physician 87(10):712
Noursadeghi M et al (2008) Quantitative imaging assay for NF-κB nuclear translocation in primary human macrophages. J Immunol Methods 329(1–2):194–200
Orhan IE (2012) Centella asiatica (L.) Urban: from traditional medicine to modern medicine with neuroprotective potential. Evid Based Complement Alternat Med 2012:946259
Ossola B et al (2009) The multiple faces of quercetin in neuroprotection. Expert Opin Drug Saf 8(4):397–409
Otuechere CA et al (2023) Action of vanillin-spiked zinc ferrite nanoparticles against cadmium-induced liver damage: computational insights with AKT 1, BCl-2 and TLR 8 proteins. J Drug Deliv Sci Technol 80:104139
Packer L et al (1997) Neuroprotection by the metabolic antioxidant α-lipoic acid. Free Radical Biol Med 22(1–2):359–378
Pal SK, Shukla Y (2003) Herbal medicine: current status and the future. Asian Pac J Cancer Prev 4(4):281–288
Park S et al (2011) Kaempferol acts through mitogen-activated protein kinases and protein kinase B/AKT to elicit protection in a model of neuroinflammation in BV2 microglial cells. Br J Pharmacol 164(3):1008–1025
Parodi V, Jacchetti E, Osellame R, Cerullo G, Polli D, Raimondi MT (2020) Nonlinear Optical Microscopy: From Fundamentals to Applications in Live Bioimaging. Front Bioeng Biotechnol 8:585363
Patra SK (2008) Dissecting lipid raft facilitated cell signaling pathways in cancer. Biochim Biophys Acta 1785(2):182–206
Pedraza-Chaverrí J et al (2009) ROS scavenging capacity and neuroprotective effect of α-mangostin against 3-nitropropionic acid in cerebellar granule neurons. Exp Toxicol Pathol 61(5):491–501
Peng W et al (2022) Paeoniflorin is a promising natural monomer for neurodegenerative diseases via modulation of Ca2+ and ROS homeostasis. Curr Opin Pharmacol 62:97–102
Pérez-De La Cruz V et al (2006) Protective effect of S-allylcysteine on 3-nitropropionic acid-induced lipid peroxidation and mitochondrial dysfunction in rat brain synaptosomes. Brain Res Bull 68(5):379–383
Psotova J et al (2006) Photoprotective properties of Prunella vulgaris and rosmarinic acid on human keratinocytes. J Photochem Photobiol, B 84(3):167–174
Qu M et al (2011) Protective effects of lycopene against amyloid β-induced neurotoxicity in cultured rat cortical neurons. Neurosci Lett 505(3):286–290
Qureshi M et al (2018) Therapeutic potential of curcumin for multiple sclerosis. Neurol Sci 39:207–214
Radad K et al (2009) Thymoquinone protects dopaminergic neurons against MPP+ and rotenone. Phytother Res 23(5):696–700
Rando TA (2006) Stem cells, ageing and the quest for immortality. Nature 441(7097):1080–1086
Rani L et al (2022) Evaluation of potential neuroprotective effects of Vanillin against MPP+/MPTP-induced dysregulation of dopaminergic regulatory mechanisms in SH-SY5Y cells and a mouse model of Parkinson’s disease. Mol Neurobiol 60(8):4693–4715
Ravishankar D et al (2016) Thioflavones as novel neuroprotective agents. Bioorg Med Chem 24(21):5513–5520
Ren Y, Savill J (1998) Apoptosis: the importance of being eaten. Cell Death Differ 5(7):563–568
Renaud J et al (2015) Epigallocatechin-3-gallate, a promising molecule for Parkinson’s disease? Rejuvenation Res 18(3):257–269
Roach H et al (2004) Chondroptosis: a variant of apoptotic cell death in chondrocytes? Apoptosis 9(3):265–277
Rowland LP, Shneider NA (2001) Amyotrophic lateral sclerosis. N Engl J Med 344(22):1688–1700
Sagredo O et al (2007) Cannabidiol reduced the striatal atrophy caused 3-nitropropionic acid in vivo by mechanisms independent of the activation of cannabinoid, vanilloid TRPV1 and adenosine A2A receptors. Eur J Neurosci 26(4):843–851
Sagredo O et al (2012) Cannabinoids: novel medicines for the treatment of Huntington’s disease. Recent Pat CNS Drug Discov 7(1):41–48
Salehi B et al (2019) The therapeutic potential of apigenin. Int J Mol Sci 20(6):1305
Salinthone S et al (2008) Lipoic acid: a novel therapeutic approach for multiple sclerosis and other chronic inflammatory diseases of the CNS. Endocr Metab Immune Disord Drug Targets 8(2):132–142
Salinthone S et al (2010) Lipoic acid attenuates inflammation via cAMP and protein kinase A signaling. PLoS ONE 5(9):e13058
Samarghandian S et al (2018) A review on possible therapeutic effect of Nigella sativa and thymoquinone in neurodegenerative diseases. CNS Neurol Disord Drug Targets 17(6):412–420
Sandhir R, Mehrotra A (2013) Quercetin supplementation is effective in improving mitochondrial dysfunctions induced by 3-nitropropionic acid: implications in Huntington’s disease. Biochim Biophys Acta 1832(3):421–430
Sandhir R et al (2014) Curcumin nanoparticles attenuate neurochemical and neurobehavioral deficits in experimental model of Huntington’s disease. NeuroMol Med 16(1):106–118
Sani MM et al (2016) Phytopharmacology and phytotherapy of regulatory T cells: a new approach to treat multiple sclerosis. Pharm Lett 8(3):215–220
Saravanan V et al (2021) Molecular docking of active compounds from Phoenix pusilla root extract against antidiabetic and anti-inflammatory drug targets. J Appl Biol Biotechnol 9(1):26–30
Sarkaki A et al (2019) Chrysin prevents cognitive and hippocampal long-term potentiation deficits and inflammation in rat with cerebral hypoperfusion and reperfusion injury. Life Sci 226:202–209
Saudou F, Humbert S (2016) The biology of huntingtin. Neuron 89(5):910–926
Scalbert A, Williamson G (2000) Dietary intake and bioavailability of polyphenols. J Nutr 130(8):2073S-2085S
Schillace R et al (2007) Lipoic acid stimulates cAMP production in T lymphocytes and NK cells. Biochem Biophys Res Commun 354(1):259–264
Schuh C, Schieberle P (2006) Characterization of the key aroma compounds in the beverage prepared from Darjeeling black tea: quantitative differences between tea leaves and infusion. J Agric Food Chem 54(3):916–924
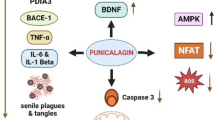
Seeram NP et al (2005) In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J Nutr Biochem 16(6):360–367
Sethi G et al (2007) Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-κB–regulated gene products and TAK1-mediated NF-κB activation. Blood 109(7):2727–2735
Sgarbossa A et al (2015) Ferulic acid: a hope for Alzheimer’s disease therapy from plants. Nutrients 7(7):5764–5782
Shabab T et al (2017) Neuroinflammation pathways: a general review. Int J Neurosci 127(7):624–633
Sharma J et al (2013) Ethnomedicinal plants used for treating epilepsy by indigenous communities of sub-Himalayan region of Uttarakhand, India. J Ethnopharmacol 150(1):353–370
Sharma D et al (2016) Quercetin attenuates neuronal death against aluminum-induced neurodegeneration in the rat hippocampus. Neuroscience 324:163–176
Shi C et al (2010) Ginkgo biloba extract in Alzheimer’s disease: from action mechanisms to medical practice. Int J Mol Sci 11(1):107–123
Shi X et al (2015) Curcumin inhibits Aβ-induced microglial inflammatory responses in vitro: involvement of ERK1/2 and p38 signaling pathways. Neurosci Lett 594:105–110
Shinomol GK (2008) Effect of Centella asiatica leaf powder on oxidative markers in brain regions of prepubertal mice in vivo and its in vitro efficacy to ameliorate 3-NPA-induced oxidative stress in mitochondria. Phytomedicine 15(11):971–984
Silva JM et al (2020) Secondary metabolites with antioxidant activities for the putative treatment of amyotrophic lateral sclerosis (ALS): “Experimental evidences”. Oxid Med Cell Longev 2020:5642029
Singh R et al (2019) Regulation of apoptosis in health and disease: the balancing act of BCL-2 family proteins. Nat Rev Mol Cell Biol 20(3):175–193
Smith KJ (2006) Pathophysiology of multiple sclerosis. Rev Prat 56(12):1299–1303
Srinivasan M et al (2007) Ferulic acid: therapeutic potential through its antioxidant property. Journal of Clinical Biochemistry and Nutrition 40(2):92–100
Suk K (2005) Regulation of neuroinflammation by herbal medicine and its implications for neurodegenerative diseases. Neurosignals 14(1–2):23–33
Swaroop S et al (2016) HSP60 plays a regulatory role in IL-1β-induced microglial inflammation via TLR4-p38 MAPK axis. J Neuroinflammation 13:1–19
Tancheva LP et al (2020) Neuroprotective mechanisms of three natural antioxidants on a rat model of parkinson’s disease: a comparative study. Antioxidants 9(1):49
Tang D et al (2019) The molecular machinery of regulated cell death. Cell Res 29(5):347–364
Taupin P (2008) Adult neurogenesis, neuroinflammation and therapeutic potential of adult neural stem cells. Int J Med Sci 5(3):127
Thornberry NA, Lazebnik Y (1998) Caspases: enemies within. Science 281(5381):1312–1316
Tie L et al (2008) Down-regulation of brain-pancreas relative protein in diabetic rats and by high glucose in PC12 cells: prevention by calpain inhibitors. J Pharmacol Sci 106(1):28–37
Tronche F et al (1997) Analysis of the distribution of binding sites for a tissue-specific transcription factor in the vertebrate genome. J Mol Biol 266(2):231–245
Truong VL et al (2018) Role of resveratrol in regulation of cellular defense systems against oxidative stress. BioFactors 44(1):36–49
Turan D et al (2020) Evaluation of the neuroprotective potential of caffeic acid phenethyl ester in a cellular model of Parkinson’s disease. Eur J Pharmacol 883:173342
Van Den Bossche K et al (2006) The quest for the mechanism of melanin transfer. Traffic 7(7):769–778
Vince JE et al (2018) The mitochondrial apoptotic effectors BAX/BAK activate caspase-3 and-7 to trigger NLRP3 inflammasome and caspase-8 driven IL-1β activation. Cell Rep 25(9):2339-2353. e2334
Vradenburg G (2015) A pivotal moment in Alzheimer’s disease and dementia: how global unity of purpose and action can beat the disease by 2025. Expert Rev Neurother 15(1):73–82
Walczak H (2011) TNF and ubiquitin at the crossroads of gene activation, cell death, inflammation, and cancer. Immunol Rev 244(1):9–28
Walle T et al (2001) Disposition and metabolism of the flavonoid chrysin in normal volunteers. Br J Clin Pharmacol 51(2):143–146
Wang L et al (2012) Arsenic modulates heme oxygenase-1, interleukin-6, and vascular endothelial growth factor expression in endothelial cells: roles of ROS, NF-κB, and MAPK pathways. Arch Toxicol 86:879–896
Wang K et al (2014) Protective effect of paeoniflorin on Aβ25–35-induced SH-SY5Y cell injury by preventing mitochondrial dysfunction. Cell Mol Neurobiol 34(2):227–234
Wang J et al (2016) Neuroprotective effect of several phytochemicals and its potential application in the prevention of neurodegenerative diseases. Geriatrics 1(4):29
Wang Y et al (2017) Toxicity of inhaled particulate matter on the central nervous system: neuroinflammation, neuropsychological effects and neurodegenerative disease. J Appl Toxicol 37(6):644–667
Wang T-F et al (2022) Inhibition of nigral microglial activation reduces age-related loss of dopaminergic neurons and motor deficits. Cells 11(3):481
Welbat JU et al (2018) Neuroprotective properties of asiatic acid against 5-fluorouracil chemotherapy in the hippocampus in an adult rat model. Nutrients 10(8):1053
Wilson R, Tocher DR (1991) Lipid and fatty acid composition is altered in plaque tissue from multiple sclerosis brain compared with normal brain white matter. Lipids 26(1):9–15
Wright JS (2002) Predicting the antioxidant activity of curcumin and curcuminoids. J Mol Struct (thoechem) 591(1–3):207–217
Yang Y et al (2015) Ginsenoside Rg1 suppressed inflammation and neuron apoptosis by activating PPARγ/HO-1 in hippocampus in rat model of cerebral ischemia-reperfusion injury. Int J Clin Exp Pathol 8(3):2484
Yang X et al (2021) Ginsenoside Rg1 exerts neuroprotective effects in 3-nitropronpionic acid-induced mouse model of Huntington’s disease via suppressing MAPKs and NF-κB pathways in the striatum. Acta Pharmacol Sin 42(9):1409–1421
Yuan C-X et al (2008) Effects of traditional Chinese herbal medicine on the neurobehavioral manifestations and the activity of dopamine D2 receptor in corpora striatum of rats with levodopa-induced dyskinesias. Zhong Xi Yi Jie He Xue Bao 6(10):1024–1028
Zeng K-W et al (2012) Schisandrin B exerts anti-neuroinflammatory activity by inhibiting the Toll-like receptor 4-dependent MyD88/IKK/NF-κB signaling pathway in lipopolysaccharide-induced microglia. Eur J Pharmacol 692(1–3):29–37
Zhang X et al (2014) A review of experimental research on herbal compounds in amyotrophic lateral sclerosis. Phytother Res 28(1):9–21
Zhang J et al (2016) Neuroprotective effects of theaflavins against oxidative stress-induced apoptosis in PC12 cells. Neurochem Res 41(12):3364–3372
Acknowledgements
The authors are thankful to the Institute for providing excellent support to complete this work.
Author information
Authors and Affiliations
Contributions
BS: writing original draft and preparation of figures; NR—helped in writing manuscript and referencing; CS—critically review and editing; AS: conceptualization, supervision, critical review, and editing. The authors confirm that no paper mill and artificial intelligence was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors give full consent to publish.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarkar, B., Rana, N., Singh, C. et al. Medicinal herbal remedies in neurodegenerative diseases: an update on antioxidant potential. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-03027-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-03027-5