Abstract
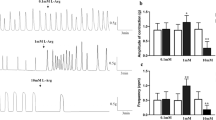
The muscular layer in the GI tract consists of an inner circular muscular layer and an outer longitudinal muscular layer. Acetylcholine (ACh) is the representative neurotransmitter that causes contractions in the gastrointestinal tracts of most animal species. There are many reports of muscarinic receptor-mediated contraction of longitudinal muscles, but few studies discuss circular muscles. The present study detailed the contractile response in the circular smooth muscles of the mouse ileum. We used small muscle strips (0.2 mm × 1 mm) and large muscle strips (4 × 4 mm) isolated from the circular and longitudinal muscle layers of the mouse ileum to compare contraction responses in circular and longitudinal smooth muscles. The time to peak contractile responses to carbamylcholine (CCh) were later in the small muscle strips (0.2 × 1 mm) of circular muscle (5.7 min) than longitudinal muscles (0.4 min). The time to peak contractile responses to CCh in the large muscle strips (4 × 4 mm) were also later in the circular muscle (3.1 min) than the longitudinal muscle (1.4 min). Furthermore, a muscarinic M2 receptor antagonist and gap junction inhibitor significantly delayed the time to peak contraction of the large muscle strips (4 × 4 mm) from the circular muscular layer. Our findings indicate that muscarinic M2 receptors in the circular muscular layer of mouse ileum exert a previously undocumented function in gut motility via the regulation of gap junctions.






Similar content being viewed by others
Abbreviations
- ACh:
-
Acetylcholine
- 4-DAMP:
-
4-Diphenylacetoxy-N-methylpiperidine methiodide
- L-NNA:
-
N-nitro-L-arginine
- TTX:
-
Tetrodotoxin
- CCh:
-
Carbamylcholine
- CBX:
-
Carbenoxolone
- MET:
-
Methoctramine
- MR:
-
Muscarinic receptors
- NO:
-
Nitric oxide
- ICCs:
-
Interstitial cells of Cajal
References
Benfenati V, Caprini M, Nicchia GP, Rossi A, Dovizio M, Cervetto C, Nobile M, Ferroni S (2009) Carbenoxolone inhibits volume-regulated anion conductance in cultured rat cortical astroglia. Channels (Austin) 3:323–336
Bramley JR, Wiles EM, Sollars PJ, Pickard GE (2011) Carbenoxolone blocks the light-evoked rise in intracellular calcium in isolated melanopsin ganglion cell photoreceptors. PLoS One 6:e22721
Callahan SM, Creed KE (1981) Electrical and mechanical activity of the isolated lower urinary tract of the guinea-pig. Br J Pharmacol 74:353–358
Caulfield MP, Birdsall NJM (1998) Classification of muscarinic acetylcholine receptors. Pharmacol Rev 50:279–290
Daniel EE, Wang YF (1999) Gap junctions in intestinal smooth muscle and interstitial cells of Cajal. Microsc Res Tech 47:309–320
Eglen RM, Hegde SS, Watson N (1996) Muscarinic receptor subtypes and smooth muscle function. Pharmacol Rev 48:531–565
Ehlert FJ (1996) The interaction of 4-DAMP mustard with subtypes of the muscarinic receptor. Life Sci 58:1971–1978
El-Yazbi AF, Schulz R, Daniel EE (2007) Differential inhibitory control of circular and longitudinal smooth muscle layers of Balb/C mouse small intestine. Auton Neurosci 131:36–44
Esqueda EE, Gerstin EH Jr, Griffin MT, Ehlert FJ (1996) Stimulation of cyclic AMP accumulation and phosphoinositide hydrolysis by M3 muscarinic receptors in the rat peripheral lung. Biochem Pharmacol 52:643–658
Frei E, Huster M, Smital P, Schlossmann J, Hofmann F, Wegener JW (2009) Calcium-dependent and calcium-independent inhibition of contraction by cGMP/cGKI in intestinal smooth muscle. Am J Physiol Gastrointest Liver Physiol 297:G834–G839
Fujita A, Okishio Y, Takeuchi T, Hata F (2004) Roles of interstitial cells of Cajal in regulation of motility of the mouse intestine. Folia Pharmacol Jpn 123:170–178
Honda K, Takano Y, Kamiya H (1993) Pharmacological profiles of muscarinic receptors in the longitudinal smooth muscle of guinea pig ileum. Jpn J Pharmacol 62:43–47
Horowitz A, Menice CB, Laporte R, Morgan KG (1996) Mechanisms of smooth muscle contraction. Physiol Rev 76:967–992
Iino S, Horiguchi S, Horiguchi K (2011) Interstitial cells of Cajal in the gastrointestinal musculature of Wjic c-kit mutant mice. J Smooth Muscle Res 47:111–121
Jacobsen JC, Aalkjaer C, Nilsson H, Matchkov VV, Freiberg J, Holstein-Rathlou NH (2007) A model of smooth muscle cell synchronization in the arterial wall. Am J Physiol Heart Circ Physiol 293:H229–H237
Janssen LJ (2002) Ionic mechanisms and Ca2+ regulation in airway smooth muscle contraction:do the data contradict dogma? Am J Physiol Lung Cell Mol Physiol 282:L1161–L1178
Kamishima T, Nelson MT, Patlak JB (1992) Carbachol modulates voltage sensitivity of calcium channels in bronchial smooth muscle of rats. Am J Physiol 263:C69–C77
Khurana S, Yamada M, Wess J, Kennedy RH, Raufman JP (2005) Deoxycholyltaurine-induced vasodilation of rodent aorta is nitric oxide- and muscarinic M3 receptor-dependent. Eur J Pharmacol 517:103–110
Kondo T, Nakajima M, Teraoka H, Unno T, Komori S, Yamada M, Kitazawa T (2011) Muscarinic receptor subtypes involved in regulation of colonic motility in mice: functional studies using muscarinic receptor-deficient mice. Eur J Pharmacol 670:236–243
Matsui M, Motomura D, Fujikawa T, Jiang J, Takahashi S, Manabe T, Taketo MM (2002) Mice lacking M2 and M3 muscarinic acetylcholine receptors are devoid of cholinergic smooth muscle contractions but still viable. J Neurosci 22:10627–10632
Mayer EA, Sun XP (1992) Contraction-coupling in colonic smooth muscle. Annu Rev Physiol 54:395–414
McConalouge K, Furness JB (1994) Gastrointestinal neurotransmitters. Baillieres Clin Endocrinol Metab 8:51–76
Missiaen L, Smedt HD, Droogmans G, Himpens B, Casteels R (1992) Calcium ion homeostasis in smooth muscle. Pharmaco Ther 56:191–231
Pacaud P, Bolton T (1991) Relation between muscarinic receptor cationic current and internal calcium in guinea-pig jejunal smooth muscle cells. J Physiol 441(477–499)
Sanders KM, Ward SM (2007) Kit mutants and gastrointestinal physiology. J Physiol 578:33–42
Sato K, Sanders KM, Gerthoffer WT, Publicover NG (1994) Sources of calcium utilized in cholinergic responses in canine colonic smooth muscle. Am J Physiol 267:C1666–C1673
Semenov I, Wang B, Herlihy JT, Brenner R (2011) BK channel β1 subunits regulate airway contraction secondary to M2 muscarinic acetylcholine receptor mediated depolarization. J Physiol 589:1803–1817
Takeuchi T, Kushida M, Hirayama N, Kitayama M, Fujita A, Hata F (2004) Mechanisms involved in carbachol-induced Ca2+ sensitization of contractile elements in rat proximal and distal colon. Br J Pharmacol 142:657–666
Takeuchi T, Nakajima H, Hata F, Azuma YT (2007a) A minor role for Ca2+ sensitization in sustained contraction through activation of muscarinic receptor in circular muscle of rat distal colon. Eur J Physiol 454:565–574
Takeuchi T, Tanaka K, Nakajima H, Matsui M, Azuma YT (2007b) M2 and M3 muscarinic receptors are involved in enteric nerve-mediated contraction of the mouse ileum: findings obtained with muscarinic-receptor knockout mouse. Am J Physiol Gastrointest Liver Physiol 292:G154–G164
Unno T, Matsuyama H, Sakamoto T, Uchiyama M, Izumi Y, Okamoto H, Yamada M, Wess J, Komori S (2005) M2 and M3 muscarinic receptor-mediated contractions in longitudinal smooth muscle of the ileum studied with receptor knockout mice. Br J Pharmacol 146:98–108
Wang GD, Wang XY, Hu HZ, Liu S, Gao N, Fang X, Xia Y, Wood JD (2007) Inhibitory neuromuscular transmission mediated by the P2Y1 purinergic receptor in guinea pig small intestine. Am J Physiol Gastrointest Liver Physiol 292:G1483–G1489
White CW, Short JL, Haynes JM, Matsui M, Ventura S (2011) Contractions of the mouse prostate elicited by acetylcholine are mediated by M(3) muscarinic receptors. J Pharmacol Exp Ther 339:870–877
Zizzo MG, Mulè F, Serio R (2007) Inhibitory purinergic transmission in mouse caecum: role for P2Y1 receptors as prejunctional modulators of ATP release. Neuroscience 150:658–664
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azuma, YT., Samezawa, N., Nishiyama, K. et al. Differences in time to peak carbachol-induced contractions between circular and longitudinal smooth muscles of mouse ileum. Naunyn-Schmiedeberg's Arch Pharmacol 389, 63–72 (2016). https://doi.org/10.1007/s00210-015-1177-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-015-1177-3