Abstract
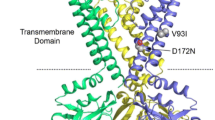
Dronedarone is a novel class III antiarrhythmic drug that is widely used in atrial fibrillation. It has been shown in native cardiomyocytes that dronedarone inhibits cardiac inwardly rectifying current IK1 at high concentrations, which may contribute both its antifibrillatory efficacy and its potential proarrhythmic side effects. However, the underlying mechanism has not been studied in further detail to date. In the mammalian heart, heterotetrameric assembly of Kir2.x channels is the molecular basis of IK1 current. Therefore, we studied the effects of dronedarone on wild-type and mutant Kir2.x channels in the Xenopus oocyte expression system. Dronedarone inhibited Kir2.1 currents but had no effect on Kir2.2 or Kir2.3 currents. Onset of block was slow but completely reversible upon washout. Blockade of Kir2.1 channels did not exhibit strong voltage dependence or frequency dependence. In a screening with different Kir2.1 mutants lacking specific binding sites within the cytoplasmic pore region, we found that residue E224 is essential for binding of dronedarone to Kir2.1 channels. In conclusion, direct block of Kir2.1 channel subunits by dronedarone through binding at E224 may underlie its inhibitory effects on cardiac IK1 current.






Similar content being viewed by others
References
Amoros I et al (2013) Propafenone blocks human cardiac Kir2.x channels by decreasing the negative electrostatic charge in the cytoplasmic pore. Biochem Pharmacol 86:267–278
Bosch RF, Li GR, Gaspo R, Nattel S (1999a) Electrophysiologic effects of chronic amiodarone therapy and hypothyroidism, alone and in combination, on guinea pig ventricular myocytes. J Pharmacol Exp Ther 289:156–165
Bosch RF, Zeng X, Grammer JB, Popovic K, Mewis C, Kühlkamp V (1999b) Ionic mechanisms of electrical remodeling in human atrial fibrillation. Cardiovasc Res 44:121–131
Connolly SJ et al (2011) Dronedarone in high-risk permanent atrial fibrillation. N Engl J Med 365:2268–2276. doi:10.1056/NEJMoa1109867
Danelich IM, Reed BN, Hollis IB, Cook AM, Rodgers JE (2013) Clinical update on the management of atrial fibrillation. Pharmacotherapy 33:422–446. doi:10.1002/phar.1217
de Boer TP et al (2010) The anti-protozoal drug pentamidine blocks KIR2.x-mediated inward rectifier current by entering the cytoplasmic pore region of the channel. Br J Pharmacol 159:1532–1541
Dhamoon AS, Jalife J (2005) The inward rectifier current (IK1) controls cardiac excitability and is involved in arrhythmogenesis. Heart Rhythm 2:316–324. doi:10.1016/j.hrthm.2004.11.012
Dobrev D, Graf E, Wettwer E, Himmel HM, Hála O, Doerfel C, Christ T, Schüler S, Ravens U (2001) Molecular basis of downregulation of G-protein-coupled inward rectifying K(+) current (I(K, ACh) in chronic human atrial fibrillation: decrease in GIRK4 mRNA correlates with reduced I(K, ACh) and muscarinic receptor-mediated shortening of action potentials. Circulation 104:2551–2557
Du X, Zhang H, Lopes C, Mirshahi T, Rohacs T, Logothetis DE (2004) Characteristic interactions with phosphatidylinositol 4,5-bisphosphate determine regulation of Kir channels by diverse modulators. J Biol Chem 279:37271–37281. doi:10.1074/jbc.M403413200
Ferrer T, Ponce-Balbuena D, Lopez-Izquierdo A, Arechiga-Figueroa IA, de Boer TP, van der Heyden MA, Sanchez-Chapula JA (2011) Carvedilol inhibits Kir2.3 channels by interference with PIP(2)–channel interaction. Eur J Pharmacol 668:72–77
Gautier P, Guillemare E, Marion A, Bertrand JP, Tourneur Y, Nisato D (2003) Electrophysiologic characterization of dronedarone in guinea pig ventricular cells. J Cardiovasc Pharmacol 41:191–202
Girmatsion Z, Biliczki P, Bonauer A, Wimmer-Greinecker G, Scherer M, Moritz A, Bukowska A, Goette A, Nattel S, Hohnloser SH, Ehrlich JR (2009) Changes in microRNA-1 expression and IK1 up-regulation in human atrial fibrillation. Heart Rhythm 6:1802–1809
Hilgemann DW (1997) Cytoplasmic ATP-dependent regulation of ion transporters and channels: mechanisms and messengers. Annu Rev Physiol 59:193–220. doi:10.1146/annurev.physiol.59.1.193
Hohnloser SH et al (2009) Effect of dronedarone on cardiovascular events in atrial fibrillation. N Engl J Med 360:668–678. doi:10.1056/NEJMoa0803778
Logothetis DE, Lupyan D, Rosenhouse-Dantsker A (2007) Diverse Kir modulators act in close proximity to residues implicated in phosphoinositide binding. J Physiol 582:953–965. doi:10.1113/jphysiol.2007.133157
Lopes CM, Zhang H, Rohacs T, Jin T, Yang J, Logothetis DE (2002) Alterations in conserved Kir channel–PIP2 interactions underlie channelopathies. Neuron 34:933–944
Lopez-Izquierdo A (2011a) Mechanisms for Kir channel inhibition by quinacrine: acute pore block of Kir2.x channels and interference in PIP2 interaction with Kir2.x and Kir6.2 channels. Pflugers Arch 462:505–517. doi:10.1007/s00424-011-0995-5
Lopez-Izquierdo A et al (2011b) The antimalarial drug mefloquine inhibits cardiac inward rectifier K + channels: evidence for interference in PIP2–channel interaction. J Cardiovasc Pharmacol 57:407–415
Madeja M, Musshoff U, Speckmann EJ (1997) Follicular tissues reduce drug effects on ion channels in oocytes of Xenopus laevis. Eur J Neurosci 9:599–604
Melnyk P, Zhang L, Shrier A, Nattel S (2002) Differential distribution of Kir2.1 and Kir2.3 subunits in canine atrium and ventricle. Am J Physiol Heart Circ Physiol 283:H1123–H1133
Noujaim SF et al (2007) Up-regulation of the inward rectifier K + current (I K1) in the mouse heart accelerates and stabilizes rotors. J Physiol 578:315–326
Noujaim SF et al (2010) Specific residues of the cytoplasmic domains of cardiac inward rectifier potassium channels are effective antifibrillatory targets. FASEB J 24:4302–4312. doi:10.1096/fj.10-163246
Noujaim SF, Stuckey JA, Ponce-Balbuena D, Ferrer-Villada T, López-Izquierdo A, Pandit SV, Sánchez-Chapula JA, Jalife J (2011) Structural bases for the different anti-fibrillatory effects of chloroquine and quinidine. Cardiovasc Res 89:862–869. doi:10.1093/cvr/cvr008
Ponce-Balbuena D, Lopez-Izquierdo A, Ferrer T, Rodriguez-Menchaca AA, Arechiga-Figueroa IA, Sanchez-Chapula JA (2009) Tamoxifen inhibits inward rectifier K + 2.x family of inward rectifier channels by interfering with phosphatidylinositol 4,5-bisphosphate–channel interactions. J Pharmacol Exp Ther 331:563–573
Preisig-Muller R (2002) Heteromerization of Kir2.x potassium channels contributes to the phenotype of Andersen’s syndrome. Proc Natl Acad Sci U S A 99:7774–7779. doi:10.1073/pnas.102609499
Rodriguez-Menchaca AA, Navarro-Polanco RA, Ferrer-Villada T, Rupp J, Sachse FB, Tristani-Firouzi M, Sanchez-Chapula JA (2008) The molecular basis of chloroquine block of the inward rectifier Kir2.1 channel. Proc Natl Acad Sci U S A 105:1364–1368. doi:10.1073/pnas.0708153105
Rohacs T, Lopes CM, Jin T, Ramdya PP, Molnar Z, Logothetis DE (2003) Specificity of activation by phosphoinositides determines lipid regulation of Kir channels. Proc Natl Acad Sci U S A 100:745–750. doi:10.1073/pnas.0236364100
Sato R, Koumi S, Singer DH, Hisatome I, Jia H, Eager S, Wasserstrom JA (1994) Amiodarone blocks the inward rectifier potassium channel in isolated guinea pig ventricular cells. J Pharmacol Exp Ther 269:1213–1219
Schimpf R, Bauersfeld U, Gaita F, Wolpert C (2005a) Short QT syndrome: successful prevention of sudden cardiac death in an adolescent by implantable cardioverter–defibrillator treatment for primary prophylaxis. Heart Rhythm 2:416–417. doi:10.1016/j.hrthm.2004.11.026
Schimpf R, Wolpert C, Gaita F, Giustetto C, Borggrefe M (2005b) Short QT syndrome. Cardiovasc Res 67:357–366. doi:10.1016/j.cardiores.2005.03.026
Schram G, Pourrier M, Wang Z, White M, Nattel S (2003) Barium block of Kir2 and human cardiac inward rectifier currents: evidence for subunit-heteromeric contribution to native currents. Cardiovasc Res 59:328–338
Singh BN et al (2007) Dronedarone for maintenance of sinus rhythm in atrial fibrillation or flutter. N England J Med 357:987–999. doi:10.1056/NEJMoa054686
Thomas D et al (2003) Acute effects of dronedarone on both components of the cardiac delayed rectifier K + current, HERG and KvLQT1/minK potassium channels. Br J Pharmacol 140:996–1002
Tristani-Firouzi M, Etheridge SP (2010) Kir2.1 channelopathies: the Andersen–Tawil syndrome. Pflugers Arch 460:289–294
Tristani-Firouzi M et al (2002) Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J Clin Investig 110:381–388
Van Wagoner DR, Pond AL, McCarthy PM, Trimmer JS, Nerbonne JM (1997) Outward K + current densities and Kv1.5 expression are reduced in chronic human atrial fibrillation. Circ Res 80:772–781
Varro A et al (2001) Electrophysiological effects of dronedarone (SR 33589), a noniodinated amiodarone derivative in the canine heart: comparison with amiodarone. Br J Pharmacol 133:625–634
Voigt N, Trausch A, Knaut M, Matschke K, Varró A, Van Wagoner DR, Nattel S, Ravens U, Dobrev D (2010) Left-to-right atrial inward rectifier potassium current gradients in patients with paroxysmal versus chronic atrial fibrillation. Circ Arrhythm Electrophysiol 3:472–480. doi:10.1161/CIRCEP.110.954636
Wang Z, Yue L, White M, Pelletier G, Nattel S (1998) Differential distribution of inward rectifier potassium channel transcripts in human atrium versus ventricle. Circulation 98:2422–2428
Xia M et al (2005) A Kir2.1 gain-of-function mutation underlies familial atrial fibrillation. Biochem Biophys Res Commun 332:1012–1019
Xie LH, John SA, Ribalet B, Weiss JN (2008) Phosphatidylinositol-4,5-bisphosphate (PIP2) regulation of strong inward rectifier Kir2.1 channels: multilevel positive cooperativity. J Physiol 586:1833–1848
Zaritsky JJ, Eckman DM, Wellman GC, Nelson MT, Schwarz TL (2000) Targeted disruption of Kir2.1 and Kir2.2 genes reveals the essential role of the inwardly rectifying K(+) current in K(+)-mediated vasodilation. Circ Res 87:160–166
Zobel C, Cho HC, Nguyen TT, Pekhletski R, Diaz RJ, Wilson GJ, Backx PH (2003) Molecular dissection of the inward rectifier potassium current (IK1) in rabbit cardiomyocytes: evidence for heteromeric co-assembly of Kir2.1 and Kir2.2. J Physiol 550:365–372
Acknowledgments
Financial support for this study was granted by the DFG (Projects ZI1177/1-1 to Dr. Zitron and SCHO1350/2-1 to Dr. Scholz) and the University of Heidelberg: Postdoc Fellowship Programme of the Faculty of Medicine to Dr. Scherer. We thank C. Jeckel for excellent technical assistance.
Ethical standards
All animal studies have been approved by the ethics committee of the University of Heidelberg and have been performed in accordance with the ethical standards laid down in the 1964 declaration of Helsinki and its later amendments (current version 2013). The manuscript does not contain clinical studies or patient data.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xynogalos, P., Seyler, C., Scherer, D. et al. Class III antiarrhythmic drug dronedarone inhibits cardiac inwardly rectifying Kir2.1 channels through binding at residue E224. Naunyn-Schmiedeberg's Arch Pharmacol 387, 1153–1161 (2014). https://doi.org/10.1007/s00210-014-1045-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-014-1045-6