Abstract
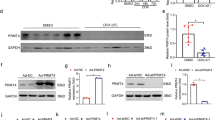
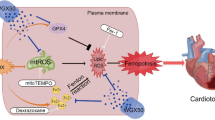
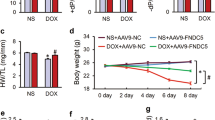
Doxorubicin (DOX) is one of the most frequently used chemotherapeutic drugs belonging to the class of anthracyclines. However, the cardiotoxic effects of anthracyclines limit their clinical use. Recent studies have suggested that ferroptosis is the main underlying pathogenetic mechanism of DOX-induced cardiomyopathy (DIC). BTB-and-CNC homology 1 (Bach1) acts as a key role in the regulation of ferroptosis. However, the mechanistic role of Bach1 in DIC remains unclear. Therefore, this study aimed to investigate the underlying mechanistic role of Bach1 in DOX-induced cardiotoxicity using the DIC mice in vivo (DOX at cumulative dose of 20 mg/kg) and the DOX-treated H9c2 cardiomyocytes in vitro (1 μM). Our results show a marked upregulation in the expression of Bach1 in the cardiac tissues of the DOX-treated mice and the DOX-treated cardiomyocytes. However, Bach1−/− mice exhibited reduced lipid peroxidation and less severe cardiomyopathy after DOX treatment. Bach1 knockdown protected against DOX-induced ferroptosis in both in vivo and in vitro models. Ferrostatin-1 (Fer-1), a potent inhibitor of ferroptosis, significantly alleviated DOX-induced cardiac damage. However, the cardioprotective effects of Bach1 knockdown were reversed by pre-treatment with Zinc Protoporphyrin (ZnPP), a selective inhibitor of heme oxygenase-1(HO-1). Taken together, these findings demonstrated that Bach1 promoted oxidative stress and ferroptosis through suppressing the expression of HO-1. Therefore, Bach1 may present as a promising new therapeutic target for the prevention and early intervention of DOX-induced cardiotoxicity.
Highlight
-
Ferroptosis is responsible for doxorubicin-induced cardiotoxicity.
-
Bach1 expression is prominently increased by doxorubicin.
-
Bach1 knockdown alleviates doxorubicin-induced cardiotoxicity.
-
Bach1 promotes ferroptosis of cardiomyocytes through downregulation of HO-1.







Similar content being viewed by others
Data availability
Data will be made available from the corresponding author on reasonable request.
References
Ahuja M, Kaidery NA, Dutta D et al (2022) Harnessing the therapeutic potential of the Nrf2/Bach1 signaling pathway in Parkinson’s disease. Antioxidants (basel). https://doi.org/10.3390/antiox11091780
Binaschi M, Bigioni M, Cipollone A et al (2001) Anthracyclines: selected new developments. Curr Med Chem Anticancer Agents 1(2):113–130. https://doi.org/10.2174/1568011013354723
Brigelius-Flohe R, Maiorino M (2013) Glutathione peroxidases. Biochim Biophys Acta 1830(5):3289–3303. https://doi.org/10.1016/j.bbagen.2012.11.020
Cai L, Arbab AS, Lee TJ et al (2022) BACH1-Hemoxygenase-1 axis regulates cellular energetics and survival following sepsis. Free Radic Biol Med 188:134–145. https://doi.org/10.1016/j.freeradbiomed.2022.06.005
Fang X, Wang H, Han D et al (2019) Ferroptosis as a target for protection against cardiomyopathy. Proc Natl Acad Sci U S A 116(7):2672–2680. https://doi.org/10.1073/pnas.1821022116
Gatti RA, Shaked R, Mohandas TK, Salser W (1987) Human ferritin genes: chromosomal assignments and polymorphisms. Am J Hum Genet 41(4):654–667
Johmura Y, Harris AS, Ohta T, Nakanishi M (2020) FBXO22, an epigenetic multiplayer coordinating senescence, hormone signaling, and metastasis. Cancer Sci 111(8):2718–2725. https://doi.org/10.1111/cas.14534
Kumagai T, Matsukawa N, Kaneko Y, Kusumi Y, Mitsumata M, Uchida K (2004) A lipid peroxidation-derived inflammatory mediator: identification of 4-hydroxy-2-nonenal as a potential inducer of cyclooxygenase-2 in macrophages. J Biol Chem 279(46):48389–48396. https://doi.org/10.1074/jbc.M409935200
Li X, Lin Y, Wang S et al (2020) Extracellular superoxide dismutase is associated with left ventricular geometry and heart failure in patients with cardiovascular disease. J Am Heart Assoc 9(15):e016862. https://doi.org/10.1161/JAHA.120.016862
Li D, Song C, Zhang J, Zhao X (2023) Resveratrol alleviated 5-FU-induced cardiotoxicity by attenuating GPX4 dependent ferroptosis. J Nutr Biochem 112:109241. https://doi.org/10.1016/j.jnutbio.2022.109241
Liu N, Zou L, Hu M, Zhang M (2019) Heme as a target for protection against doxorubicin-induced apoptosis in H9c2 cardiomyocytes. Cell Stress Chaperones 24(6):1211–1217. https://doi.org/10.1007/s12192-019-01045-4
Liu JE, Barac A, Thavendiranathan P, Scherrer-Crosbie M (2020) Strain imaging in cardio-oncology. JACC CardioOncol 2(5):677–689. https://doi.org/10.1016/j.jaccao.2020.10.011
Lunning MA, Kutty S, Rome ET et al (2015) Cardiac magnetic resonance imaging for the assessment of the myocardium after doxorubicin-based chemotherapy. Am J Clin Oncol 38(4):377–381. https://doi.org/10.1097/COC.0b013e31829e19be
Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53:401–426. https://doi.org/10.1146/annurev-pharmtox-011112-140320
Mito S, Ozono R, Oshima T et al (2008) Myocardial protection against pressure overload in mice lacking Bach1, a transcriptional repressor of heme oxygenase-1. Hypertension 51(6):1570–1577. https://doi.org/10.1161/HYPERTENSIONAHA.107.102566
Nishizawa H, Matsumoto M, Shindo T et al (2020) Ferroptosis is controlled by the coordinated transcriptional regulation of glutathione and labile iron metabolism by the transcription factor BACH1. J Biol Chem 295(1):69–82. https://doi.org/10.1074/jbc.RA119.009548
Nishizawa H, Yamanaka M, Igarashi K (2022) Ferroptosis: regulation by competition between NRF2 and BACH1 and propagation of the death signal. FEBS J. https://doi.org/10.1111/febs.16382
Park HS, Hong YJ, Han K et al (2021) Ultrahigh-field cardiovascular magnetic resonance T1 and T2 mapping for the assessment of anthracycline-induced cardiotoxicity in rat models: validation against histopathologic changes. J Cardiovasc Magn Reson 23(1):76. https://doi.org/10.1186/s12968-021-00767-8
Raschi E, Vasina V, Ursino MG, Boriani G, Martoni A, De Ponti F (2010) Anticancer drugs and cardiotoxicity: insights and perspectives in the era of targeted therapy. Pharmacol Ther 125(2):196–218. https://doi.org/10.1016/j.pharmthera.2009.10.002
Siegel D, Bersie S, Harris P et al (2021) A redox-mediated conformational change in NQO1 controls binding to microtubules and alpha-tubulin acetylation. Redox Biol 39:101840. https://doi.org/10.1016/j.redox.2020.101840
Sun J, Hoshino H, Takaku K et al (2002) Hemoprotein Bach1 regulates enhancer availability of heme oxygenase-1 gene. EMBO J 21(19):5216–5224. https://doi.org/10.1093/emboj/cdf516
Swain SM, Whaley FS, Ewer MS (2003) Congestive heart failure in patients treated with doxorubicin: a retrospective analysis of three trials. Cancer 97(11):2869–2879. https://doi.org/10.1002/cncr.11407
Tadokoro T, Ikeda M, Ide T et al (2020) Mitochondria-dependent ferroptosis plays a pivotal role in doxorubicin cardiotoxicity. JCI Insight. https://doi.org/10.1172/jci.insight.132747
Tian X, Cong F, Guo H, Fan J, Chao G, Song T (2019) Downregulation of Bach1 protects osteoblasts against hydrogen peroxide-induced oxidative damage in vitro by enhancing the activation of Nrf2/ARE signaling. Chemico-Biol Interact. https://doi.org/10.1016/j.cbi.2019.06.019
Wang H, Liu C, Zhao Y, Gao G (2020) Mitochondria regulation in ferroptosis. Eur J Cell Biol 99(1):151058. https://doi.org/10.1016/j.ejcb.2019.151058
Wiel C, Le Gal K, Ibrahim MX et al (2019) BACH1 stabilization by antioxidants stimulates lung cancer metastasis. Cell 178(2):330–345. https://doi.org/10.1016/j.cell.2019.06.005. (e22)
Xu J, Zhu K, Wang Y, Chen J (2023) The dual role and mutual dependence of heme/HO-1/Bach1 axis in the carcinogenic and anti-carcinogenic intersection. J Cancer Res Clin Oncol 149(1):483–501. https://doi.org/10.1007/s00432-022-04447-7
Yang WS, SriRamaratnam R, Welsch ME et al (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 156(1–2):317–331. https://doi.org/10.1016/j.cell.2013.12.010
Yu W, Chen C, Xu C et al (2022) Activation of p62-NRF2 axis protects against doxorubicin-induced ferroptosis in cardiomyocytes: a novel role and molecular mechanism of resveratrol. Am J Chin Med 50(8):2103–2123. https://doi.org/10.1142/S0192415X22500902
Yusoff FM, Maruhashi T, Kawano KI et al (2021) Bach1 plays an important role in angiogenesis through regulation of oxidative stress. Microvasc Res 134:104126. https://doi.org/10.1016/j.mvr.2020.104126
Zenke-Kawasaki Y, Dohi Y, Katoh Y et al (2007) Heme induces ubiquitination and degradation of the transcription factor Bach1. Mol Cell Biol 27(19):6962–6971. https://doi.org/10.1128/MCB.02415-06
Zhang X, Guo J, Wei X et al (2018) Bach1: function, regulation, and involvement in disease. Oxid Med Cell Longev 2018:1347969. https://doi.org/10.1155/2018/1347969
Zhao L, Qi Y, Xu L et al (2018) MicroRNA-140-5p aggravates doxorubicin-induced cardiotoxicity by promoting myocardial oxidative stress via targeting Nrf2 and Sirt2. Redox Biol 15:284–296. https://doi.org/10.1016/j.redox.2017.12.013
Funding
This work was supported by grants from Joint Fund of Shandong Natural Science Foundation (No. ZR2022LSW025), Shandong Natural Science Foundation (No. ZR2021MH400) and Shandong Natural Science Foundation (No. ZR2019MH125).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Haiyan Lou planned experiments and interpreted data. Yanwei Wang performed most of the experiments and analyzed data. Jingru Qiu and Nan Zhang participated in the animal experiment. Hua Yan and Shixuan Gao participated in the cell experiment. Ning Xu participated in the experiment design and data interpretation. Cuiyan Wang participated in the experiment design and manuscript revision. Yanwei Wang wrote the first draft of the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Qiu, J., Yan, H. et al. The Bach1/HO-1 pathway regulates oxidative stress and contributes to ferroptosis in doxorubicin-induced cardiomyopathy in H9c2 cells and mice. Arch Toxicol (2024). https://doi.org/10.1007/s00204-024-03697-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00204-024-03697-3