Abstract
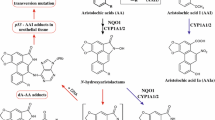
Aristolochic acid (AA-I) induces upper urothelial tract cancer (UUTC) and bladder cancer (BC) in humans. AA-I forms the 7-(2ʹ-deoxyadenosin-N6-yl)aristolactam I (dA-AL-I) adduct, which induces multiple A:T-to-T:A transversion mutations in TP53 of AA-I exposed UTUC patients. This mutation is rarely reported in TP53 of other transitional cell carcinomas and thus recognized as an AA-I mutational signature. A:T-to-T:A transversion mutations were recently detected in bladder tumors of patients in Asia with known AA-I-exposure, implying that AA-I contributes to BC. Mechanistic studies on AA-I genotoxicity have not been reported in human bladder. In this study, we examined AA-I DNA adduct formation and mechanisms of toxicity in the human RT4 bladder cell line. The biological potencies of AA-I were compared to 4-aminobiphenyl, a recognized human bladder carcinogen, and several structurally related carcinogenic heterocyclic aromatic amines (HAA), which are present in urine of smokers and omnivores. AA-I (0.05–10 µM) induced a concentration- and time-dependent cytotoxicity. AA-I (100 nM) DNA adduct formation occurred at over a thousand higher levels than the principal DNA adducts formed with 4-ABP or HAAs (1 µM). dA-AL-I adduct formation was detected down to a 1 nM concentration. Studies with selective chemical inhibitors provided evidence that NQO1 is the major enzyme involved in AA-I bio-activation in RT4 cells, whereas CYP1A1, another enzyme implicated in AA-I toxicity, had a lesser role in bio-activation or detoxification of AA-I. AA-I DNA damage also induced genotoxic stress leading to p53-dependent apoptosis. These biochemical data support the human mutation data and a role for AA-I in BC.




Similar content being viewed by others
References
Arlt VM, Ferluga D, Stiborova M et al (2002a) Isaristolochic acid a risk factor for Balkan endemic nephropathy-associated urothelial cancer? Int J Cancer 101(5):500–502. https://doi.org/10.1002/ijc.10602
Arlt VM, Stiborova M, Schmeiser HH (2002b) Aristolochic acid as a probable human cancer hazard in herbal remedies: a review. Mutagenesis 17(4):265–277. https://doi.org/10.1093/mutage/17.4.265
Arlt VM, Stiborova M, vom Brocke J et al (2007) Aristolochic acid mutagenesis: molecular clues to the aetiology of Balkan endemic nephropathy-associated urothelial cancer. Carcinogenesis 28(11):2253–2261. https://doi.org/10.1093/carcin/bgm082
Aubrey BJ, Kelly GL, Janic A, Herold MJ, Strasser A (2018) How does p53 induce apoptosis and how does this relate to p53-mediated tumour suppression? Cell Death Differ 25(1):104-113. https://doi.org/10.1038/cdd.2017.169
Bellamri M, Xiao S, Murugan P, Weight CJ, Turesky RJ (2018) Metabolic activation of the cooked meat carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine in human prostate. ToxicolSci 163(2):543–556. https://doi.org/10.1093/toxsci/kfy060
Bellamri M, Yao L, Bonala R, Johnson F, Von Weymarn LB, Turesky RJ (2019) Bioactivation of the tobacco carcinogens 4-aminobiphenyl (4-ABP) and 2-amino-9H-pyrido[2,3-b]indole (AalphaC) in human bladder RT4 cells. Arch Toxicol 93(7):1893–1902. https://doi.org/10.1007/s00204-019-02486-7
Bessette EE, Goodenough AK, Langouet S et al (2009) Screening for DNA adducts by data-dependent constant neutral loss-triple stage mass spectrometry with a linear quadrupole ion trap mass spectrometer. Anal Chem 81(2):809–819. https://doi.org/10.1021/ac802096p
Bieler CA, Stiborova M, Wiessler M, Cosyns JP, van Ypersele de Strihou C, Schmeiser HH (1997) 32P-post-labelling analysis of DNA adducts formed by aristolochic acid in tissues from patients with Chinese herbs nephropathy. Carcinogenesis 18(5):1063–1067. https://doi.org/10.1093/carcin/18.5.1063
Burke MD, Mayer RT (1983) Differential effects of phenobarbitone and 3-methylcholanthrene induction on the hepatic microsomal metabolism and cytochrome P-450-binding of phenoxazone and a homologous series of its n-alkyl ethers (alkoxyresorufins). ChemBiol Interact 45(2):243–258. https://doi.org/10.1016/0009-2797(83)90072-8
Chen M, Gong L, Qi X et al (2011) Inhibition of renal NQO1 activity by dicoumarol suppresses nitroreduction of aristolochic acid I and attenuates its nephrotoxicity. ToxicolSci 122(2):288–296. https://doi.org/10.1093/toxsci/kfr138
Cheng ST, Hu JL, Ren JH et al (2021) Dicoumarol, an NQO1 inhibitor, blocks cccDNA transcription by promoting degradation of HBx. J Hepatol 74(3):522–534. https://doi.org/10.1016/j.jhep.2020.09.019
Choudry GA, Stewart PA, Double JA et al (2001) A novel strategy for NQO1 (NAD(P)H:quinone oxidoreductase, EC 1.6.99.2) mediated therapy of bladder cancer based on the pharmacological properties of EO9. Br J Cancer 85(8):1137–1146. https://doi.org/10.1054/bjoc.2001.2056
Cooper MJ, Haluschak JJ, Johnson D et al (1994) p53 mutations in bladder carcinoma cell lines. Oncol Res 6(12):569–579
Debelle FD, Vanherweghem JL, Nortier JL (2008) Aristolochic acid nephropathy: a worldwide problem. Kidney Int 74(2):158–169. https://doi.org/10.1038/ki.2008.129
Dehn DL, Siegel D, Swann E, Moody CJ, Ross D (2003) Biochemical, cytotoxic, and genotoxic effects of ES936, a mechanism-based inhibitor of NAD(P)H:quinone oxidoreductase 1, in cellular systems. MolPharmacol 64(3):714–720. https://doi.org/10.1124/mol.64.3.714
Dong H, Suzuki N, Torres MC et al (2006) Quantitative determination of aristolochic acid-derived DNA adducts in rats using 32P-postlabeling/polyacrylamide gel electrophoresis analysis. Drug MetabDispos 34(7):1122–1127. https://doi.org/10.1124/dmd.105.008706
Dracinska H, Barta F, Levova K et al (2016) Induction of cytochromes P450 1A1 and 1A2 suppresses formation of DNA adducts by carcinogenic aristolochic acid I in rats in vivo. Toxicology 344–346:7–18. https://doi.org/10.1016/j.tox.2016.01.011
Ferrucci LM, Sinha R, Ward MH et al (2010) Meat and components of meat and the risk of bladder cancer in the NIH-AARP Diet and Health Study. Cancer 116(18):4345–4353. https://doi.org/10.1002/cncr.25463
Grollman AP (2013) Aristolochic acid nephropathy: Harbinger of a global iatrogenic disease. Environ Mol Mutagen 54(1):1–7. https://doi.org/10.1002/em.21756
Grollman AP, Shibutani S, Moriya M et al (2007) Aristolochic acid and the etiology of endemic (Balkan) nephropathy. Proc Natl AcadSci USA 104(29):12129–12134. https://doi.org/10.1073/pnas.0701248104
Hoang ML, Chen CH, Sidorenko VS et al (2013) Mutational signature of aristolochic acid exposure as revealed by whole-exome sequencing. Sci Transl Med 5(197):197ra102. https://doi.org/10.1126/scitranslmed.3006200
IARC (2004) Iarc Working Group on the Evaluation of Carcinogenic Risks to Humans: Tobacco smoke and involuntary smoking. IARC MonogrEvalCarcinog Risks Hum 83:1–1438
IARC (2012) Working Group on the Evaluation of Carcinogenic Risks to Humans Pharmaceuticals. A review of human carcinogens. IARC MonogrEvalCarcinog Risks Hum 100(Pt A):1–401
Jelakovic B, Dika Z, Arlt VM et al (2019) Balkan endemic nephropathy and the causative role of aristolochic acid. SeminNephrol 39(3):284–296. https://doi.org/10.1016/j.semnephrol.2019.02.007
Kirlin WG, Trinidad A, Yerokun T et al (1989) Polymorphic expression of acetyl coenzyme A-dependent arylamine N-acetyltransferase and acetyl coenzyme A-dependent O-acetyltransferase-mediated activation of N-hydroxyarylamines by human bladder cytosol. Cancer Res 49(9):2448–2454
Krais AM, Speksnijder EN, Melis JP et al (2016) The impact of p53 on DNA damage and metabolic activation of the environmental carcinogen benzo[a]pyrene: effects in Trp53(+/+), Trp53(+/-) and Trp53(-/-) mice. Arch Toxicol 90(4):839–851. https://doi.org/10.1007/s00204-015-1531-8
Nakajima T, Aoyama T (2000) Polymorphism of drug-metabolizing enzymes in relation to individual susceptibility to industrial chemicals. Ind Health 38(2):143–152. https://doi.org/10.2486/indhealth.38.143
Ng AWT, Poon SL, Huang MN et al (2017) Aristolochic acids and their derivatives are widely implicated in liver cancers in Taiwan and throughout Asia. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aan6446
Okuno Y, Bonala R, Attaluri S et al (2019) Bioactivation mechanisms of N-hydroxyaristolactams: nitroreduction metabolites of aristolochic acids. Environ Mol Mutagen 60(9):792–806. https://doi.org/10.1002/em.22321
O’Toole CM, Povey S, Hepburn P, Franks LM (1983) Identity of some human bladder cancer cell lines. Nature 301(5899):429–430. https://doi.org/10.1038/301429a0
Pacifici GM, Franchi M, Colizzi C, Giuliani L, Rane A (1988) Sulfotransferase in humans: development and tissue distribution. Pharmacology 36(6):411–419. https://doi.org/10.1159/000138330
Pathak KV, Chiu TL, Amin EA, Turesky RJ (2016) Methemoglobin formation and characterization of hemoglobin adducts of carcinogenic aromatic amines and heterocyclic aromatic amines. Chem Res Toxicol 29(3):255–269. https://doi.org/10.1021/acs.chemrestox.5b00418
Poon SL, Huang MN, Choo Y et al (2015) Mutation signatures implicate aristolochic acid in bladder cancer development. Genome Med 7(1):38. https://doi.org/10.1186/s13073-015-0161-3
Reshetnikova G, Sidorenko VS, Whyard T et al (2016) Genotoxic and cytotoxic effects of the environmental pollutant 3-nitrobenzanthrone on bladder cancer cells. Exp Cell Res 349(1):101–108. https://doi.org/10.1016/j.yexcr.2016.10.003
Romanov V, Whyard TC, Waltzer WC, Grollman AP, Rosenquist T (2015) Aristolochic acid-induced apoptosis and G2 cell cycle arrest depends on ROS generation and MAP kinases activation. Arch Toxicol 89(1):47–56. https://doi.org/10.1007/s00204-014-1249-z
Sborchia M, De Prez EG, Antoine MH et al (2019) The impact of p53 on aristolochic acid I-induced nephrotoxicity and DNA damage in vivo and in vitro. Arch Toxicol 93(11):3345–3366. https://doi.org/10.1007/s00204-019-02578-4
Shibutani S, Dong H, Suzuki N, Ueda S, Miller F, Grollman AP (2007) Selective toxicity of aristolochic acids I and II. Drug MetabDispos 35(7):1217–1222. https://doi.org/10.1124/dmd.107.014688
Stiborova M, Mares J, Frei E, Arlt VM, Martinek V, Schmeiser HH (2011) The human carcinogen aristolochic acid i is activated to form DNA adducts by human NAD(P)H:quinone oxidoreductase without the contribution of acetyltransferases or sulfotransferases. Environ Mol Mutagen 52(6):448–459. https://doi.org/10.1002/em.20642
Stiborova M, Levova K, Barta F et al (2012) Bioactivation versus detoxication of the urothelial carcinogen aristolochic acid I by human cytochrome P450 1A1 and 1A2. ToxicolSci 125(2):345–358. https://doi.org/10.1093/toxsci/kfr306
Stiborova M, Levova K, Barta F et al (2014) The influence of dicoumarol on the bioactivation of the carcinogen aristolochic acid I in rats. Mutagenesis 29(3):189–200. https://doi.org/10.1093/mutage/geu004
Stiborova M, Barta F, Levova K et al (2015) A mechanism of O-demethylation of aristolochic acid I by cytochromes P450 and their contributions to this reaction in human and rat livers: experimental and theoretical approaches. Int J MolSci 16(11):27561–27575. https://doi.org/10.3390/ijms161126047
Stiborova M, Arlt VM, Schmeiser HH (2016) Balkan endemic nephropathy: an update on its aetiology. Arch Toxicol 90(11):2595–2615. https://doi.org/10.1007/s00204-016-1819-3
Stiborova M, Arlt VM, Schmeiser HH (2017) DNA adducts formed by aristolochic acid are unique biomarkers of exposure and explain the initiation phase of upper urothelial cancer. Int J Mol Sci. https://doi.org/10.3390/ijms18102144
Sugimura T, Wakabayashi K, Nakagama H, Nagao M (2004) Heterocyclic amines: mutagens/carcinogens produced during cooking of meat and fish. Cancer Sci 95(4):290–299. https://doi.org/10.1111/j.1349-7006.2004.tb03205.x
Tassaneeyakul W, Birkett DJ, Veronese ME et al (1993) Specificity of substrate and inhibitor probes for human cytochromes P450 1A1 and 1A2. J PharmacolExpTher 265(1):401–407
Turesky RJ, Le Marchand L (2011) Metabolism and biomarkers of heterocyclic aromatic amines in molecular epidemiology studies: lessons learned from aromatic amines. Chem Res Toxicol 24(8):1169–1214. https://doi.org/10.1021/tx200135s
Van Noorden CJ (2001) The history of Z-VAD-FMK, a tool for understanding the significance of caspase inhibition. ActaHistochem 103(3):241–251. https://doi.org/10.1078/0065-1281-00601
Vanherweghem JL, Depierreux M, Tielemans C et al (1993) Rapidly progressive interstitial renal fibrosis in young women: association with slimming regimen including Chinese herbs. Lancet 341(8842):387–391. https://doi.org/10.1016/0140-6736(93)92984-2
Vousden KH, Lane DP (2007) p53 in health and disease. Nat Rev Mol Cell Biol 8(4):275–283. https://doi.org/10.1038/nrm2147
Walton MI, Wilson SC, Hardcastle IR, Mirza AR, Workman P (2005) An evaluation of the ability of pifithrin-alpha and -beta to inhibit p53 function in two wild-type p53 human tumor cell lines. Mol Cancer Ther 4(9):1369–1377. https://doi.org/10.1158/1535-7163.MCT-04-0341
Wohak LE, Baranski AC, Krais AM, Schmeiser HH, Phillips DH, Arlt VM (2018) The impact of p53 function on the metabolic activation of the carcinogenic air pollutant 3-nitrobenzanthrone and its metabolites 3-aminobenzanthrone and N-hydroxy-3-aminobenzanthrone in human cells. Mutagenesis 33(4):311–321. https://doi.org/10.1093/mutage/gey025
Wohak LE, Monien B, Phillips DH, Arlt VM (2019) Impact of p53 function on the sulfotransferase-mediated bioactivation of the alkylated polycyclic aromatic hydrocarbon 1-hydroxymethylpyrene in vitro. Environ Mol Mutagen 60(8):752–758. https://doi.org/10.1002/em.22299
Yun BH, Rosenquist TA, Sidorenko V et al (2012) Biomonitoring of aristolactam-DNA adducts in human tissues using ultra-performance liquid chromatography/ion-trap mass spectrometry. Chem Res Toxicol 25(5):1119–1131. https://doi.org/10.1021/tx3000889
Yun BH, Yao L, Jelakovic B et al (2014) Formalin-fixed paraffin-embedded tissue as a source for quantitation of carcinogen DNA adducts: aristolochic acid as a prototype carcinogen. Carcinogenesis 35(9):2055–2061. https://doi.org/10.1093/carcin/bgu101
Zhou L, Fu P, Huang XR, Liu F, Lai KN, Lan HY (2010) Activation of p53 promotes renal injury in acute aristolochic acid nephropathy. J Am SocNephrol 21(1):31–41. https://doi.org/10.1681/ASN.2008111133
Acknowledgements
The Turesky laboratory gratefully acknowledges the support of the Masonic Chair in Cancer Causation.
Funding
This research was supported by R01ES030559 (RJT) from the National Institute of Environmental Health Sciences and by R01CA220367 (RJT) from the National Cancer Institute, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bellamri, M., Brandt, K., Brown, C.V. et al. Cytotoxicity and genotoxicity of the carcinogen aristolochic acid I (AA-I) in human bladder RT4 cells. Arch Toxicol 95, 2189–2199 (2021). https://doi.org/10.1007/s00204-021-03059-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-03059-3