Abstract
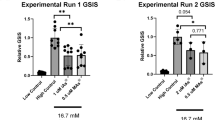
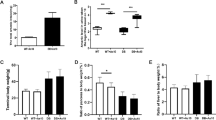
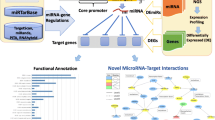
Diabetes is a metabolic disorder characterized by fasting hyperglycemia and impaired glucose tolerance. Laboratory and population studies have shown that inorganic arsenic (iAs) can impair these pathways. Other metals including cadmium (Cd) and manganese (Mn) have also been linked to diabetes phenotypes. MicroRNAs, short non-coding RNAs that regulate gene expression, have emerged as potential drivers of metabolic dysfunction. MicroRNAs responsive to metal exposures in vitro have also been reported in independent studies to regulate insulin secretion in vivo. We hypothesize that microRNA dysregulation may associate with and possibly contribute to insulin secretion impairment upon exposure to iAs, Cd, or Mn. We exposed insulin secreting rat insulinoma cells to non-cytotoxic concentrations of iAs (1 µM), Cd (5 µM), and Mn (25 µM) for 24 h followed by small RNA sequencing to identify dysregulated microRNAs. RNA sequencing was then performed to further investigate changes in gene expression caused by iAs exposure. While all three metals significantly inhibited glucose-stimulated insulin secretion, high-throughput sequencing revealed distinct microRNA profiles specific to each exposure. One of the most significantly upregulated microRNAs post-iAs treatment is miR-146a (~ + 2-fold), which is known to be activated by nuclear factor κB (NF-κB) signaling. Accordingly, we found by RNA-seq analysis that genes upregulated by iAs exposure are enriched in the NF-κB signaling pathway and genes down-regulated by iAs exposure are enriched in miR-146a binding sites and are involved in regulating beta cell function. Notably, iAs exposure caused a significant decrease in the expression of Camk2a, a calcium-dependent protein kinase that regulates insulin secretion, has been implicated in type 2 diabetes, and is a likely target of miR-146a. Further studies are needed to elucidate potential interactions among NF-kB, miR-146a, and Camk2a in the context of iAs exposure.






Similar content being viewed by others
References
Abiola M, Favier M, Christodoulou-Vafeiadou E et al (2009) Activation of Wnt/β-catenin signaling increases insulin sensitivity through a reciprocal regulation of Wnt10b and SREBP-1c in skeletal muscle cells. PLoS One 4:e8509. https://doi.org/10.1371/journal.pone.0008509
Alipoor B, Ghaedi H, Meshkani R et al (2017) Association of MiR-146a expression and type 2 diabetes mellitus: a meta-analysis. Int J Mol Cell Med 6:156–163. https://doi.org/10.22088/acadpub.BUMS.6.3.156
Bagge A, Clausen TR, Larsen S et al (2012) MicroRNA-29a is up-regulated in beta-cells by glucose and decreases glucose-stimulated insulin secretion. Biochem Biophys Res Commun 426:266–272. https://doi.org/10.1016/j.bbrc.2012.08.082
Baly DL, Curry DL, Keen CL, Hurley LS (1984) Effect of manganese deficiency on insulin secretion and carbohydrate homeostasis in rats. J Nutr 114:1438–1446. https://doi.org/10.1093/jn/114.8.1438
Beck RF, Bommarito P, Douillet C et al (2018) Circulating miRNAs associated with arsenic exposure. Environ Sci Technol. https://doi.org/10.1021/acs.est.8b06457
Belgardt B, Ahmed K, Spranger M et al (2015) The microRNA-200 family regulates pancreatic beta cell survival in type 2 diabetes. Nat Med 21:619–627. https://doi.org/10.1038/nm.3862
Bhatt K, Lanting LL, Jia Y et al (2016) Anti-inflammatory role of microRNA-146a in the pathogenesis of diabetic nephropathy. J Am Soc Nephrol 27:2277–2288. https://doi.org/10.1681/ASN.2015010111
Bloomston M, Frankel WL, Petrocca F et al (2007) MicroRNA expression patterns to differentiate pancreatic adenocarcinoma from normal pancreas and chronic pancreatitis. JAMA 297:1901. https://doi.org/10.1001/jama.297.17.1901
Bouwens L, Rooman I (2005) Regulation of pancreatic beta-cell mass. Physiol Rev 85:1255–1270. https://doi.org/10.1152/physrev.00025.2004
Chang K-C, Hsu C-C, Liu S-H et al (2013) Cadmium induces apoptosis in pancreatic β-cells through a mitochondria-dependent pathway: the role of oxidative stress-mediated c-Jun N-terminal kinase activation. PLoS One 8:e54374. https://doi.org/10.1371/journal.pone.0054374
Chen EY, Xu H, Gordonov S et al (2012) Expression2Kinases: mRNA profiling linked to multiple upstream regulatory layers. Bioinformatics 28:105–111. https://doi.org/10.1093/bioinformatics/btr625
Chen S, Feng B, Thomas AA, Chakrabarti S (2017) miR-146a regulates glucose induced upregulation of inflammatory cytokines extracellular matrix proteins in the retina and kidney in diabetes. PLoS One 12:e0173918. https://doi.org/10.1371/journal.pone.0173918
Chi SW, Zang JB, Mele A, Darnell RB (2009) Argonaute HITS-CLIP decodes microRNA? mRNA interaction maps. Nature 460:479. https://doi.org/10.1038/nature08170
Dadi PK, Vierra NC, Ustione A et al (2014) Inhibition of pancreatic β-Cell Ca2+/calmodulin-dependent protein kinase II reduces glucose-stimulated calcium influx and insulin secretion, impairing glucose tolerance. J Biol Chem 289:12435–12445. https://doi.org/10.1074/jbc.M114.562587
Del Razo LM, García-Vargas GG, Valenzuela OL et al (2011) Exposure to arsenic in drinking water is associated with increased prevalence of diabetes: a cross-sectional study in the Zimapán and Lagunera regions in Mexico. Environ Health 10:73. https://doi.org/10.1186/1476-069X-10-73
Díaz-Villaseñor A, Burns AL, Salazar AM et al (2008) Arsenite reduces insulin secretion in rat pancreatic β-cells by decreasing the calcium-dependent calpain-10 proteolysis of SNAP-25. Toxicol Appl Pharmacol 231:291–299. https://doi.org/10.1016/j.taap.2008.05.018
Dixit SS, Wang T, Manzano EJQ et al (2013) Effects of CaMKII-mediated phosphorylation of ryanodine receptor type 2 on islet calcium handling, insulin secretion, and glucose tolerance. PLoS One 8:e58655. https://doi.org/10.1371/journal.pone.0058655
Douillet C, Currier J, Saunders J et al (2013) Methylated trivalent arsenicals are potent inhibitors of glucose stimulated insulin secretion by murine pancreatic islets. Toxicol Appl Pharmacol 267:11–15. https://doi.org/10.1016/j.taap.2012.12.007
Douillet C, Huang MC, Saunders RJ et al (2017) Knockout of arsenic (+3 oxidation state) methyltransferase is associated with adverse metabolic phenotype in mice: the role of sex and arsenic exposure. Arch Toxicol 91:2617–2627. https://doi.org/10.1007/s00204-016-1890-9
Dover EN, Beck R, Huang MC et al (2017) Arsenite and methylarsonite inhibit mitochondrial metabolism and glucose-stimulated insulin secretion in INS-1 832/13 β cells. Arch Toxicol. https://doi.org/10.1007/s00204-017-2074-y
Dover EN, Patel NY, Stýblo M (2018) Impact of in vitro heavy metal exposure on pancreatic β-cell function. Toxicol Lett 299:137–144. https://doi.org/10.1016/j.toxlet.2018.09.015
Ediger BN, Du A, Liu J et al (2014) Islet-1 Is essential for pancreatic β-cell function. Diabetes 63:4206–4217. https://doi.org/10.2337/db14-0096
El Muayed M, Raja MR, Zhang X et al (2012) Accumulation of cadmium in insulin-producing β cells. Islets 4:405–416. https://doi.org/10.4161/isl.23101
Fabricio G, Malta A, Chango A, De Freitas Mathias PC (2016) Environmental contaminants and pancreatic beta-cells. J Clin Res Pediatr Endocrinol 8:257–263. https://doi.org/10.4274/jcrpe.2812
Fang L, Cai J, Chen B et al (2015) Aberrantly expressed miR-582-3p maintains lung cancer stem cell-like traits by activating Wnt/β-catenin signalling. Nat Commun 6:8640. https://doi.org/10.1038/NCOMMS9640
Gribble MO, Howard BV, Umans JG et al (2012) Arsenic exposure, diabetes prevalence, and diabetes control in the Strong Heart Study. Am J Epidemiol 176:865–874. https://doi.org/10.1093/aje/kws153
Hectors TLM, Vanparys C, van der Ven K et al (2011) Environmental pollutants and type 2 diabetes: a review of mechanisms that can disrupt beta cell function. Diabetologia 54:1273–1290. https://doi.org/10.1007/s00125-011-2109-5
Hohmeier HE, Mulder H, Chen G et al (2000) Isolation of INS-1-derived cell lines with robust ATP-sensitive K+ channel-dependent and-independent glucose-stimulated insulin secretion. Diabetes 49:424–430. https://doi.org/10.2337/diabetes.49.3.424
Huang MC, Douillet C, Su M et al (2016) Metabolomic profiles of arsenic (+3 oxidation state) methyltransferase knockout mice: effect of sex and arsenic exposure. Arch Toxicol. https://doi.org/10.1007/s00204-016-1676-0
Huang MC, Douillet C, Dover EN, Stýblo M (2018) Prenatal arsenic exposure and dietary folate and methylcobalamin supplementation alter the metabolic phenotype of C57BL/6J mice in a sex-specific manner. Arch Toxicol 92:1925–1937. https://doi.org/10.1007/s00204-018-2206-z
Huang M, Douillet C, Stýblo M (2019) Arsenite and its trivalent methylated metabolites inhibit glucose-stimulated calcium influx and insulin secretion in murine pancreatic islets. Arch Toxicol. https://doi.org/10.1007/s00204-019-02526-2
IDF (2017) Diabetes atlas, 8th edn. International Diabetes Federation, Brussels
Jo S, Chen J, Xu G et al (2018) miR-204 controls glucagon-like peptide 1 receptor expression and agonist function. Diabetes 67:256–264. https://doi.org/10.2337/db17-0506
Kitamura YI, Kitamura T, Kruse J-P et al (2005) FoxO1 protects against pancreatic β cell failure through NeuroD and MafA induction. Cell Metab 2:153–163. https://doi.org/10.1016/j.cmet.2005.08.004
Korc M (1983) Manganese action on pancreatic protein synthesis in normal and diabetic rats. Am J Physiol Liver Physiol 245:G628–G634. https://doi.org/10.1152/ajpgi.1983.245.5.G628
Kuo C-C, Howard BV, Umans JG et al (2015) Arsenic exposure, arsenic metabolism, and incident diabetes in the Strong Heart Study. Diabetes Care. https://doi.org/10.2337/dc14-1641
Lachmann A, Xu H, Krishnan J et al (2010) ChEA: transcription factor regulation inferred from integrating genome-wide ChIP-X experiments. Bioinformatics 26:2438–2444. https://doi.org/10.1093/bioinformatics/btq466
Leung AKL, Young AG, Bhutkar A et al (2011) Genome-wide identification of Ago2 binding sites from mouse embryonic stem cells with and without mature microRNAs. Nat Struct Mol Biol 18:237–244. https://doi.org/10.1038/nsmb.1991
Li XT, Yu PF, Gao Y et al (2017) Association between plasma metal levels and diabetes risk: a case-control study in China. Biomed Environ Sci 30:482–491. https://doi.org/10.3967/bes2017.064
Liu S, Guo X, Wu B et al (2014) Arsenic induces diabetic effects through beta-cell dysfunction and increased gluconeogenesis in mice. Sci Rep 4:6894. https://doi.org/10.1038/srep06894
Liu B, Feng W, Wang J et al (2016) Association of urinary metals levels with type 2 diabetes risk in coke oven workers. Environ Pollut 210:1–8. https://doi.org/10.1016/j.envpol.2015.11.046
Lorenz MA, El Azzouny MA, Kennedy RT, Burant CF (2013) Metabolome response to glucose in the β-cell line INS-1 832/13. J Biol Chem 288:10923–10935. https://doi.org/10.1074/jbc.M112.414961
Makaji E, Raha S, Wade MG, Holloway AC (2011) Effect of environmental contaminants on beta cell function. Int J Toxicol 30:410–418. https://doi.org/10.1177/1091581811405544
Maull EA, Ahsan H, Edwards J et al (2012) Evaluation of the association between arsenic and diabetes: a National Toxicology Program workshop review. Environ Health Perspect 120:1658–1670. https://doi.org/10.1289/ehp.1104579
Mendez MA, González-Horta C, Sánchez-Ramírez B et al (2016) Chronic exposure to arsenic and markers of cardiometabolic risk: a cross-sectional study in Chihuahua, Mexico. Environ Health Perspect 124:104–111. https://doi.org/10.1289/ehp.1408742
Ogata H, Goto S, Fujibuchi W, Kanehisa M (1998) Computation with the KEGG pathway database. Biosystems 47:119–128
Park K-G, Lee K-M, Seo H-Y et al (2007) Glucotoxicity in the INS-1 rat insulinoma cell line is mediated by the orphan nuclear receptor small heterodimer partner. Diabetes 56:431–437
Paul DS, Devesa V, Hernandez-Zavala A et al (2008) Environmental arsenic as a disruptor of insulin signaling. Met Ions Biol Med 10:1–7
Poy MN, Eliasson L, Krutzfeldt J et al (2004) A pancreatic islet-specific microRNA regulates insulin secretion. Nature 432:226–230. https://doi.org/10.1038/nature03076
Rhee SY, Hwang Y-C, Woo J et al (2013) Arsenic exposure and prevalence of diabetes mellitus in Korean adults. J Korean Med Sci 28:861. https://doi.org/10.3346/jkms.2013.28.6.861
Rodríguez-Comas J, Moreno-Asso A, Moreno-Vedia J et al (2017) Stress-induced MicroRNA-708 impairs β-cell function and growth. Diabetes 66:3029–3040. https://doi.org/10.2337/db16-1569
Roggli E, Britan A, Gattesco S et al (2010) Involvement of microRNAs in the cytotoxic effects exerted by proinflammatory cytokines on pancreatic β-cells. Diabetes 59:978–986. https://doi.org/10.2337/db09-0881
Ruepp A, Brauner B, Dunger-Kaltenbach I et al (2008) CORUM: the comprehensive resource of mammalian protein complexes. Nucleic Acids Res 36:D646–D650. https://doi.org/10.1093/nar/gkm936
Santos GJD, Ferreira SM, Ortis F et al (2014) Metabolic memory of β-cells controls insulin secretion and is mediated by CaMKII. Mol Metab 3:484–489. https://doi.org/10.1016/j.molmet.2014.03.011
Schug J, McKenna LB, Walton G et al (2013) Dynamic recruitment of microRNAs to their mRNA targets in the regenerating liver. BMC Genom 14:264. https://doi.org/10.1186/1471-2164-14-264
Schwartz GG, Il’yasova D, Ivanova A (2003) Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 26:468–470. https://doi.org/10.2337/DIACARE.26.2.468
Shan Z, Chen S, Sun T et al (2016) U-shaped association between plasma manganese levels and type 2 diabetes. Environ Health Perspect 124:1876–1881. https://doi.org/10.1289/EHP176
Skelin M, Rupnik M, Cencic A (2010) Pancreatic beta cell lines and their applications in diabetes mellitus research. Altex 27:105–113 (doi:20686743)
Sung T-C, Huang J-W, Guo H-R (2015) Association between arsenic exposure and diabetes: a meta-analysis. Biomed Res Int 2015:368087. https://doi.org/10.1155/2015/368087
Taganov KD, Boldin MP, Chang K-J, Baltimore D (2006) NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci USA 103:12481–12486. https://doi.org/10.1073/pnas.0605298103
Trajkovski M, Hausser J, Soutschek J et al (2011) MicroRNAs 103 and 107 regulate insulin sensitivity. Nature 474:649–653. https://doi.org/10.1038/nature10112
Treviño S, Waalkes MP, Flores Hernández JA et al (2015) Chronic cadmium exposure in rats produces pancreatic impairment and insulin resistance in multiple peripheral tissues. Arch Biochem Biophys 583:27–35. https://doi.org/10.1016/j.abb.2015.07.010
Vrijens K, Bollati V, Nawrot TS (2015) MicroRNAs as potential signatures of environmental exposure or effect: a systematic review. Environ Health Perspect. https://doi.org/10.1289/ehp.1408459
Waldsee R, Eftekhari S, Ahnstedt H et al (2014) CaMKII and MEK1/2 inhibition time-dependently modify inflammatory signaling in rat cerebral arteries during organ culture. J Neuroinflamm 11:90. https://doi.org/10.1186/1742-2094-11-90
Wang W, Xie Z, Lin Y, Zhang D (2014) Association of inorganic arsenic exposure with type 2 diabetes mellitus: a meta-analysis. J Epidemiol Community Health 68:176–184. https://doi.org/10.1136/jech-2013-203114
Wang H, Ren Y, Hu X et al (2017) Effect of Wnt signaling on the differentiation of Islet β-cells from adipose-derived stem cells. Biomed Res Int. https://doi.org/10.1155/2017/2501578
Yajima H, Komatsu M, Schermerhorn T et al (1999) cAMP enhances insulin secretion by an action on the ATP-sensitive K+ channel-independent pathway of glucose signaling in rat pancreatic islets. Diabetes 48:1006–1012. https://doi.org/10.2337/diabetes.48.5.1006
Yang W, Wang J, Chen Z et al (2017) NFE2 induces miR-423-5p to promote gluconeogenesis and hyperglycemia by repressing the hepatic FAM3A-ATP-Akt pathway. Diabetes 66:1819–1832. https://doi.org/10.2337/db16-1172
Zhang P (2017) CaMKII: the molecular villain that aggravates cardiovascular disease. Exp Ther Med 13:815–820. https://doi.org/10.3892/etm.2017.4034
Zhong P, Huang H (2017) CaMKII as a pathological mediator of inflammation, oxidative stress, ER stress, autophagy and mitochondrial dysfunction in free fatty acids/hyperlipidemia-induced cardiac remodeling both in vitro and vivo. J Mol Cell Cardiol 112:134. https://doi.org/10.1016/j.yjmcc.2017.07.016
Funding
This work was funded by NIH (Grant Number R01DK105965), NIEHS (Grant Number R01ES022697), UNC Superfund by NIEHS (Grant Number 3P42ES005948-22S2), and NIGMS (Grant Number 5T32 GM007092).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Beck, R., Chandi, M., Kanke, M. et al. Arsenic is more potent than cadmium or manganese in disrupting the INS-1 beta cell microRNA landscape. Arch Toxicol 93, 3099–3109 (2019). https://doi.org/10.1007/s00204-019-02574-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02574-8