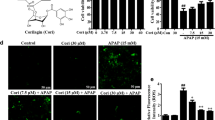
Abstract
Acetaminophen (APAP) overdose is a leading cause of drug-induced acute liver failure. Prolonged c-Jun N-terminal kinase (JNK) activation plays a central role in APAP-induced liver injury; however, growth arrest and DNA damage-inducible 45 beta (GADD45β) is known to inhibit JNK phosphorylation. The orphan nuclear receptor small heterodimer partner (SHP, NR0B2) acts as a transcriptional co-repressor of various genes. The aim of the present study was to investigate the role of SHP in APAP-evoked hepatotoxicity. We used lethal (750 mg/kg) or sublethal (300 mg/kg) doses of APAP-treated wild-type (WT), Shp knockout (Shp−/−), hepatocyte-specific Shp knockout (Shphep−/−), and Shp and Gadd45β double knockout (Shp−/−Gadd45β−/−) mice for in vivo studies. Primary mouse hepatocytes were used for a comparative in vitro study. SHP deficiency protected against APAP toxicity with an increased survival rate, decreased liver damage, and inhibition of prolonged hepatic JNK phosphorylation in mice, which was independent of APAP metabolism regulation. Furthermore, Shphep−/− mice showed diminished APAP hepatotoxicity compared with WT mice. SHP-deficient primary mouse hepatocytes also showed decreased cell death and inhibition of sustained JNK phosphorylation following toxic APAP treatment. While SHP expression declined, GADD45β expression increased after APAP treatment in WT mice. In Shp−/− mice, APAP-evoked GADD45β induction was significantly enhanced. Notably, the ameliorative effects of SHP deficiency on APAP-induced liver injury were abolished in Shp−/−Gadd45β−/− mice. The current study is the first to demonstrate that hepatocyte-specific SHP deficiency protects against APAP overdose-evoked hepatotoxicity in a JNK signaling regulation and GADD45β dependent manner. SHP is suggested to be a novel therapeutic target for APAP overdose treatment.







Similar content being viewed by others
Abbreviations
- APAP:
-
Acetaminophen
- ALT:
-
Alanine aminotransferase
- CAR:
-
Constitutive androstane receptor
- JNK:
-
c-Jun-N-terminal kinase
- CYP:
-
Cytochrome P450
- Shp flox/flox :
-
Floxed Shp exon 1 and 2
- Gadd45β −/− :
-
Gadd45β knockout
- GADD45:
-
Growth arrest and DNA damage-inducible 45
- GSH:
-
Glutathione
- H&E:
-
Hematoxylin and eosin
- MAP:
-
Mitogen-activated protein
- MKK4:
-
Kinase kinase 4
- NAQPI:
-
N-acetyl-ρ-benzoquinone imine
- NR:
-
Nuclear receptor
- ROS:
-
Reactive oxygen species
- Shp −/− Gadd45β −/− :
-
Shp and Gadd45β−/− double knockout
- Shp hep−/− :
-
Shp specifically deleted in the hepatocytes
- SHP:
-
Small heterodimer partner
- TG:
-
Transgenic
- TUNEL:
-
Terminal transferase dUTP nick end labeling
- WT:
-
Wild-type
References
Bernal W, Auzinger G, Dhawan A, Wendon J (2010) Acute liver failure. Lancet 376(9736):190–201. https://doi.org/10.1016/S0140-6736(10)60274-7
Blazka ME, Germolec DR, Simeonova P, Bruccoleri A, Pennypacker KR, Luster MI (1995) Acetaminophen-induced hepatotoxicity is associated with early changes in NF-kB and NF-IL6 DNA binding activity. J Inflamm 47(3):138–150
Chanda D, Park JH, Choi HS (2008) Molecular basis of endocrine regulation by orphan nuclear receptor Small Heterodimer Partner. Endocr J 55(2):253–268
Davidson DG, Eastham WN (1966) Acute liver necrosis following overdose of paracetamol. Br Med J 2(5512):497–499
De Smaele E, Zazzeroni F, Papa S et al (2001) Induction of gadd45beta by NF-kappaB downregulates pro-apoptotic JNK signalling. Nature 414(6861):308–313. https://doi.org/10.1038/35104560
Duan L, Davis JS, Woolbright BL et al (2016) Differential susceptibility to acetaminophen-induced liver injury in sub-strains of C57BL/6 mice: 6N versus 6J. Food Chem Toxicol 98(Pt B):107–118. https://doi.org/10.1016/j.fct.2016.10.021
Goodwin B, Jones SA, Price RR et al (2000) A regulatory cascade of the nuclear receptors FXR, SHP-1, and LRH-1 represses bile acid biosynthesis. Mol Cell 6(3):517–526
Gunawan BK, Liu ZX, Han D, Hanawa N, Gaarde WA, Kaplowitz N (2006) c-Jun N-terminal kinase plays a major role in murine acetaminophen hepatotoxicity. Gastroenterology 131(1):165–178. https://doi.org/10.1053/j.gastro.2006.03.045
Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N (2008) Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem 283(20):13565–13577. https://doi.org/10.1074/jbc.M708916200
Hinson JA, Pike SL, Pumford NR, Mayeux PR (1998) Nitrotyrosine-protein adducts in hepatic centrilobular areas following toxic doses of acetaminophen in mice. Chem Res Toxicol 11(6):604–607. https://doi.org/10.1021/tx9800349
Hinson JA, Roberts DW, James LP (2010) Mechanisms of acetaminophen-induced liver necrosis. Handb Exp Pharmacol 196:369–405. https://doi.org/10.1007/978-3-642-00663-0_12
James LP, Mayeux PR, Hinson JA (2003) Acetaminophen-induced hepatotoxicity. Drug Metab Dispos 31(12):1499–1506. https://doi.org/10.1124/dmd.31.12.1499
Jollow DJ, Mitchell JR, Potter WZ, Davis DC, Gillette JR, Brodie BB (1973) Acetaminophen-induced hepatic necrosis. II. Role of covalent binding in vivo. J Pharmacol Exp Ther 187(1):195–202
Kim YH, Hwang JH, Kim KS et al (2015) Metformin ameliorates acetaminophen hepatotoxicity via Gadd45beta-dependent regulation of JNK signaling in mice. J Hepatol 63(1):75–82. https://doi.org/10.1016/j.jhep.2015.02.008
Larson AM, Polson J, Fontana RJ et al (2005) Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology 42(6):1364–1372. https://doi.org/10.1002/hep.20948
Liebermann DA, Hoffman B (2008) Gadd45 in stress signaling. J Mol Signal 3:15. https://doi.org/10.1186/1750-2187-3-15
Locker J, Tian J, Carver R et al (2003) A common set of immediate-early response genes in liver regeneration and hyperplasia. Hepatology 38(2):314–325. https://doi.org/10.1053/jhep.2003.50299
McGill MR, Sharpe MR, Williams CD, Taha M, Curry SC, Jaeschke H (2012) The mechanism underlying acetaminophen-induced hepatotoxicity in humans and mice involves mitochondrial damage and nuclear DNA fragmentation. J Clin Invest 122(4):1574–1583. https://doi.org/10.1172/JCI59755
Mitchell JR, Jollow DJ, Potter WZ, Davis DC, Gillette JR, Brodie BB (1973a) Acetaminophen-induced hepatic necrosis. I. Role of drug metabolism. J Pharmacol Exp Ther 187(1):185–194
Mitchell JR, Jollow DJ, Potter WZ, Gillette JR, Brodie BB (1973b) Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. J Pharmacol Exp Ther 187(1):211–217
Nakagawa H, Maeda S, Hikiba Y et al (2008) Deletion of apoptosis signal-regulating kinase 1 attenuates acetaminophen-induced liver injury by inhibiting c-Jun N-terminal kinase activation. Gastroenterology 135(4):1311–1321. https://doi.org/10.1053/j.gastro.2008.07.006
Nelson SD (1990) Molecular mechanisms of the hepatotoxicity caused by acetaminophen. Semin Liver Dis 10(4):267–278. https://doi.org/10.1055/s-2008-1040482
Noh JR, Kim YH, Kim DK et al (2018) SHP deficiency increases inflammatory liver injury through CXCL2-driven neutrophil recruitment in mice. Toxicol Sci. https://doi.org/10.1093/toxsci/kfy030
Papa S, Zazzeroni F, Bubici C et al (2004) Gadd45 beta mediates the NF-kappa B suppression of JNK signalling by targeting MKK7/JNKK2. Nat Cell Biol 6(2):146–153. https://doi.org/10.1038/ncb1093
Papa S, Zazzeroni F, Fu YX et al (2008) Gadd45beta promotes hepatocyte survival during liver regeneration in mice by modulating JNK signaling. J Clin Invest 118(5):1911–1923. https://doi.org/10.1172/JCI33913
Seki E, Brenner DA, Karin M (2012) A liver full of JNK: signaling in regulation of cell function and disease pathogenesis, and clinical approaches. Gastroenterology 143(2):307–320. https://doi.org/10.1053/j.gastro.2012.06.004
Seol W, Choi HS, Moore DD (1996) An orphan nuclear hormone receptor that lacks a DNA binding domain and heterodimerizes with other receptors. Science 272(5266):1336–1339
Su AI, Guidotti LG, Pezacki JP, Chisari FV, Schultz PG (2002) Gene expression during the priming phase of liver regeneration after partial hepatectomy in mice. Proc Natl Acad Sci USA 99(17):11181–11186. https://doi.org/10.1073/pnas.122359899
Tian J, Locker J (2013) Gadd45 in the liver: signal transduction and transcriptional mechanisms. Adv Exp Med Biol 793:69–80. https://doi.org/10.1007/978-1-4614-8289-5_5
Tian J, Huang H, Hoffman B et al (2011) Gadd45beta is an inducible coactivator of transcription that facilitates rapid liver growth in mice. J Clin Invest 121(11):4491–4502. https://doi.org/10.1172/JCI38760
Wei P, Zhang J, Egan-Hafley M, Liang S, Moore DD (2000) The nuclear receptor CAR mediates specific xenobiotic induction of drug metabolism. Nature 407(6806):920–923. https://doi.org/10.1038/35038112
Win S, Than TA, Han D, Petrovic LM, Kaplowitz N (2011) c-Jun N-terminal kinase (JNK)-dependent acute liver injury from acetaminophen or tumor necrosis factor (TNF) requires mitochondrial Sab protein expression in mice. J Biol Chem 286(40):35071–35078. https://doi.org/10.1074/jbc.M111.276089
Yuk JM, Shin DM, Lee HM et al (2011) The orphan nuclear receptor SHP acts as a negative regulator in inflammatory signaling triggered by Toll-like receptors. Nat Immunol 12(8):742–751. https://doi.org/10.1038/ni.2064
Zhang J, Huang W, Chua SS, Wei P, Moore DD (2002) Modulation of acetaminophen-induced hepatotoxicity by the xenobiotic receptor CAR. Science 298(5592):422–424. https://doi.org/10.1126/science.1073502
Zhang Y, Xu P, Park K, Choi Y, Moore DD, Wang L (2008) Orphan receptor small heterodimer partner suppresses tumorigenesis by modulating cyclin D1 expression and cellular proliferation. Hepatology 48(1):289–298. https://doi.org/10.1002/hep.22342
Zhang Y, Hagedorn CH, Wang L (2011) Role of nuclear receptor SHP in metabolism and cancer. Biochim Biophys Acta 1812(8):893–908. https://doi.org/10.1016/j.bbadis.2010.10.006
Acknowledgements
We thank IB Lee, YK Choi, YJ Seo and JH Choi for technical assistance. This work was supported by a grant from the National Research Foundation of Korea (NRF) and the Korean government (MSIP) (2016R1A2A1A05004858), KRIBB Research Initiative Program of the Republic of Korea, and the Development of Platform Technology for Innovative Medical Measurements Program (No. KRISS-2018-GP2018-0018) from the Korea Research Institute of Standards and Science, and received support from French state funds through the “Agence Nationale de la Recherche” under the frame programme Investissements d’Avenir labeled ANR-10-INBS-07 PHENOMIN.
Author information
Authors and Affiliations
Contributions
YH Kim and CH Lee designed the experiments and the study, and drafted the manuscript. YH Kim, JR Noh, DH Choi, JH Kim, SJ Moon, and JH Choi performed experiments, and collected and analyzed data for the study. Yann H provided the Shpflox/flox mice model. JH Hwang, KS Kim, TG Lee, HS Choi, and CH Lee contributed to critical revisions of the text.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical standards
The manuscript does not contain clinical studies or participant data.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, YH., Noh, JR., Hwang, J.H. et al. Hepatocyte SHP deficiency protects mice from acetaminophen-evoked liver injury in a JNK-signaling regulation and GADD45β-dependent manner. Arch Toxicol 92, 2563–2572 (2018). https://doi.org/10.1007/s00204-018-2247-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2247-3