Abstract
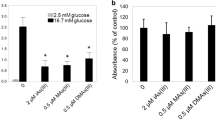
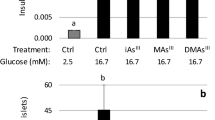
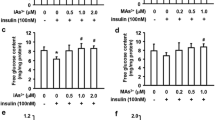
Growing evidence suggests that exposure to environmental contaminants contributes to the current diabetes epidemic. Inorganic arsenic (iAs), a drinking water and food contaminant, is one of the most widespread environmental diabetogens according to epidemiological studies. Several schemes have been proposed to explain the diabetogenic effects of iAs exposure; however, the exact mechanism remains unknown. We have shown that in vitro exposure to low concentrations of arsenite (iAsIII) or its trivalent methylated metabolites, methylarsonite (MAsIII) and dimethylarsinite (DMAsIII), inhibits glucose-stimulated insulin secretion (GSIS) from isolated pancreatic islets, with little effect on insulin transcription or total insulin content. The goal of this study was to determine if exposure to trivalent arsenicals impairs mitochondrial metabolism, which plays a key role in the regulation of GSIS in β cells. We used a Seahorse extracellular flux analyzer to measure oxygen consumption rate (OCR), a proxy for mitochondrial metabolism, in cultured INS-1 832/13 β cells exposed to iAsIII, MAsIII, or DMAsIII and stimulated with either glucose or pyruvate, a final product of glycolysis and a substrate for the Krebs cycle. We found that 24-h exposure to 2 μM iAsIII or 0.375–0.5 μM MAsIII inhibited OCR in both glucose- and pyruvate-stimulated β cells in a manner that closely paralleled GSIS inhibition. In contrast, 24-h exposure to DMAsIII (up to 2 µM) had no effects on either OCR or GSIS. These results suggest that iAsIII and MAsIII may impair GSIS in β cells by inhibiting mitochondrial metabolism, and that at least one target of these arsenicals is pyruvate decarboxylation or downstream reactions.







Similar content being viewed by others
References
Bergquist ER, Fischer RJ, Sugden KD, Martin BD (2009) Inhibition by methylated organo-arsenicals of the respiratory 2-oxo-acid dehydrogenases. J Organomet Chem 694:973–980. doi:10.1016/j.jorganchem.2008.12.028
Caicedo A (2013) Paracrine and autocrine interactions in the human islet: more than meets the eye. Semin Cell Dev Biol 24:11–21. doi:10.1016/j.semcdb.2012.09.007
Del Razo LM, García-Vargas GG, Valenzuela OL et al (2011) Exposure to arsenic in drinking water is associated with increased prevalence of diabetes: a cross-sectional study in the Zimapán and Lagunera regions in Mexico. Environ Health Glob Access Sci Source 10:73. doi:10.1186/1476-069X-10-73
Díaz-Villaseñor A, Cruz L, Cebrián A et al (2013) Arsenic exposure and calpain-10 polymorphisms impair the function of pancreatic beta-cells in humans: a pilot study of risk factors for T2DM. PLoS One 8:e51642. doi:10.1371/journal.pone.0051642
Douillet C, Currier J, Saunders J et al (2013) Methylated trivalent arsenicals are potent inhibitors of glucose stimulated insulin secretion by murine pancreatic islets. Toxicol Appl Pharmacol 267:11–15. doi:10.1016/j.taap.2012.12.007
Fu J, Woods CG, Yehuda-Shnaidman E et al (2010) Low-level arsenic impairs glucose-stimulated insulin secretion in pancreatic beta cells: involvement of cellular adaptive response to oxidative stress. Environ Health Perspect 118:864–870. doi:10.1289/ehp.0901608
Fu Z, Gilbert ER, Liu D (2013) Regulation of insulin synthesis and secretion and pancreatic beta-cell dysfunction in diabetes. Curr Diabetes Rev 9:25–53
Gong Z, Lu X, Cullen WR, Le XC (2001) Unstable trivalent arsenic metabolites, monomethylarsonous acid and dimethylarsinous acid. J Anal At Spectrom 16:1409–1413. doi:10.1039/B105834G
Green PS, Leeuwenburgh C (2002) Mitochondrial dysfunction is an early indicator of doxorubicin-induced apoptosis. Biochim Biophys Acta BBA—Mol Basis Dis 1588:94–101. doi:10.1016/S0925-4439(02)00144-8
Gribble MO, Howard BV, Umans JG et al (2012) Arsenic exposure, diabetes prevalence, and diabetes control in the strong heart study. Am J Epidemiol 176:865–874. doi:10.1093/aje/kws153
Hernández-Zavala A, Matoušek T, Drobná Z et al (2008) Speciation analysis of arsenic in biological matrices by automated hydride generation-cryotrapping-atomic absorption spectrometry with multiple microflame quartz tube atomizer (multiatomizer). J Anal At Spectrom 23:342–351. doi:10.1039/b706144g
Hohmeier HE, Mulder H, Chen G et al (2000) Isolation of INS-1-derived cell lines with robust ATP-sensitive K+ channel-dependent and -independent glucose-stimulated insulin secretion. Diabetes 49:424–430. doi:10.2337/diabetes.49.3.424
Hosseini M-J, Shaki F, Ghazi-Khansari M, Pourahmad J (2013) Toxicity of arsenic (III) on isolated liver mitochondria: a new mechanistic approach. Iran J Pharm Res IJPR 12:121–138
Janjic D, Wollheim CB (1992) Islet cell metabolism is reflected by the MTT (tetrazolium) colorimetric assay. Diabetologia 35:482–485. doi:10.1007/BF02342448
Koen YM, Liu K, Shinogle H et al (2016) Comparative toxicity and metabolism of N-acyl homologues of acetaminophen and its isomer 3′-hydroxyacetanilide. Chem Res Toxicol 29:1857–1864. doi:10.1021/acs.chemrestox.6b00270
Maechler P (2013) Mitochondrial function and insulin secretion. Mol Cell Endocrinol 379:12–18. doi:10.1016/j.mce.2013.06.019
Martinez VD, Vucic EA, Becker-Santos DD et al (2011) Arsenic exposure and the induction of human cancers. J Toxicol 2011:e431287. doi:10.1155/2011/431287
Maull EA, Ahsan H, Edwards J et al (2012) Evaluation of the association between arsenic and diabetes: a National Toxicology Program workshop review. Environ Health Perspect 120:1658–1670. doi:10.1289/ehp.1104579
Moon K, Guallar E, Navas-Acien A (2012) Arsenic exposure and cardiovascular disease: an updated systematic review. Curr Atheroscler Rep 14:542–555. doi:10.1007/s11883-012-0280-x
Naranmandura H, Xu S, Sawata T et al (2011) Mitochondria are the main target organelle for trivalent monomethylarsonous acid (MMA(III))-induced cytotoxicity. Chem Res Toxicol 24:1094–1103. doi:10.1021/tx200156k
Pace C, Banerjee TD, Welch B et al (2016) Monomethylarsonous acid, but not inorganic arsenic, is a mitochondria-specific toxicant in vascular smooth muscle cells. Toxicol In Vitro 35:188–201. doi:10.1016/j.tiv.2016.06.006
Partridge MA, Huang SXL, Hernandez-Rosa E et al (2007) Arsenic induced mitochondrial DNA damage and altered mitochondrial oxidative function: implications for genotoxic mechanisms in mammalian cells. Cancer Res 67:5239–5247. doi:10.1158/0008-5472.CAN-07-0074
Paul DS, Walton FS, Saunders RJ, Stýblo M (2011) Characterization of the impaired glucose homeostasis produced in C57BL/6 mice by chronic exposure to arsenic and high-fat diet. Environ Health Perspect 119:1104–1109. doi:10.1289/ehp.1003324
Peng Q, Harlow SD, Park SK (2015) Urinary arsenic and insulin resistance in US adolescents. Int J Hyg Environ Health 218:407–413. doi:10.1016/j.ijheh.2015.03.006
Petrick JS, Jagadish B, Mash EA, Aposhian HV (2001) Monomethylarsonous acid (MMAIII) and arsenite: LD50 in hamsters and in vitro inhibition of pyruvate dehydrogenase. Chem Res Toxicol 14:651–656. doi:10.1021/tx000264z
Prakash C, Soni M, Kumar V (2015) Biochemical and molecular alterations following arsenic-induced oxidative stress and mitochondrial dysfunction in rat brain. Biol Trace Elem Res 167:121–129. doi:10.1007/s12011-015-0284-9
Rhee SY, Hwang Y-C, Woo J et al (2013) Arsenic exposure and prevalence of diabetes mellitus in Korean adults. J Korean Med Sci 28:861–868. doi:10.3346/jkms.2013.28.6.861
Shen S, Li X-F, Cullen WR et al (2013) Arsenic binding to proteins. Chem Rev 113:7769–7792. doi:10.1021/cr300015c
Styblo M, Razo LMD, Vega L et al (2000) Comparative toxicity of trivalent and pentavalent inorganic and methylated arsenicals in rat and human cells. Arch Toxicol 74:289–299. doi:10.1007/s002040000134
Sumedha NC, Miltonprabu S (2015) Cardiac mitochondrial oxidative stress and dysfunction induced by arsenic and its amelioration by diallyl trisulphide. Toxicol Res 4:291–301. doi:10.1039/C4TX00097H
Tyler CR, Allan AM (2014) The effects of arsenic exposure on neurological and cognitive dysfunction in human and rodent studies: a review. Curr Environ Health Rep 1:132–147. doi:10.1007/s40572-014-0012-1
Wang S-L, Chang F-H, Liou S-H et al (2007) Inorganic arsenic exposure and its relation to metabolic syndrome in an industrial area of Taiwan. Environ Int 33:805–811. doi:10.1016/j.envint.2007.03.004
Yang YHC, Szabat M, Bragagnini C et al (2011) Paracrine signalling loops in adult human and mouse pancreatic islets: netrins modulate beta cell apoptosis signalling via dependence receptors. Diabetologia 54:828–842. doi:10.1007/s00125-010-2012-5
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by a grant from the National Institutes of Health [R01ES022697], a National Research Service Award from the National Institute of Environmental Health Sciences, NIH [T32 ES007126], the UNC Nutrition Obesity Research Center funded by the National Institute of Diabetes and Digestive and Kidney Diseases [DK056350], and by a Grant from the National Institutes of Health [R01DK107481 to ELK].
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dover, E.N., Beck, R., Huang, M.C. et al. Arsenite and methylarsonite inhibit mitochondrial metabolism and glucose-stimulated insulin secretion in INS-1 832/13 β cells. Arch Toxicol 92, 693–704 (2018). https://doi.org/10.1007/s00204-017-2074-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-017-2074-y