Abstract
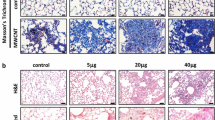
Pulmonary exposure to certain forms of carbon nanotubes (CNT) induces fibrosing lesions in the lungs that manifest an acute inflammation followed by chronic interstitial fibrosis. The mechanism of CNT-induced fibrogenesis is largely unknown. The biphasic development with drastically distinct pathologic manifestations suggests a junction of acute-to-chronic transition. Here we analyzed the molecular pathways and regulators underlying the pathologic development of CNT-induced lung fibrosis. Mice were exposed to multi-walled CNT (MWCNT; XNRI MWNT-7, Mitsui; 40 μg) by pharyngeal aspiration for 7 days along with vehicle and carbonaceous controls. Genome-wide microarray analyses of the lungs identified a range of differentially expressed genes that potentially function in the acute-to-chronic transition through pathways involving immune and inflammatory regulation, responses to stress and extracellular stimuli, and cell migration and adhesion. In particular, a T helper 2 (Th2)-driven innate immune response was significantly enriched. We then demonstrated that MWCNT induced the expression of Th2 cytokines interleukin (IL)-4 and IL-13, and a panel of signature downstream genes, such as Il4i1, Chia, and Ccl11/Eotaxin, time dependently. Induction of Th2 cytokines took place in CD4+ T lymphocytes indicating activation of Th2 cells. Furthermore, induction involved activation of a Th2 cell-specific signaling pathway through phosphorylation of STAT6 and up-regulation of GATA-3 to mediate the transcription of Th2 target genes. Our study uncovers activation of a Th2-driven immune/inflammatory response during pulmonary fibrosis development induced by MWCNT. The findings provide novel insights into the molecular events that control the transition from an acute inflammatory response to chronic fibrosis through Th2 functions in CNT-exposed lungs.









Similar content being viewed by others
Abbreviations
- BAL:
-
Bronchoalveolar lavage
- Ccl11:
-
Chemokine (C–C motif) ligand 11, or eotaxin
- Chia:
-
Chitinase, acidic, or AMCase
- CNT:
-
Carbon nanotubes
- DM:
-
Dispersion medium
- ELISA:
-
Enzyme-linked immunosorbent assay
- FDR:
-
False discovery rate
- FN1:
-
Fibronectin
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- GATA-3:
-
GATA-binding protein 3
- GO:
-
Gene ontology
- IL:
-
Interleukin
- Il4i1:
-
Interleukin 4 induced 1, or Fig. 1
- IL-4Rα:
-
IL-4 receptor α
- MWCNT:
-
Multi-walled carbon nanotubes
- STAT6:
-
Signal transducer and activator of transcription 6
- SWCNT:
-
Single-walled carbon nanotubes
- Th0:
-
Naïve T helper
- Th1:
-
T helper 1
- Th2:
-
T helper 2
- TSLP:
-
Thymic stromal lymphopoietin
References
Aiso S, Yamazaki K, Umeda Y et al (2010) Pulmonary toxicity of intratracheally instilled multiwall carbon nanotubes in male fischer 344 rats. Ind Health 48(6):783–795
Chen F, Liu Z, Wu W et al (2012) An essential role for TH2-type responses in limiting acute tissue damage during experimental helminth infection. Nat Med 18(2):260–266. doi:10.1038/nm.2628
De Volder MF, Tawfick SH, Baughman RH, Hart AJ (2013) Carbon nanotubes: present and future commercial applications. Science 339(6119):535–539. doi:10.1126/science.1222453
Donaldson K, Murphy FA, Duffin R, Poland CA (2010) Asbestos, carbon nanotubes and the pleural mesothelium: a review of the hypothesis regarding the role of long fibre retention in the parietal pleura, inflammation and mesothelioma. Part Fibre Toxicol 7:5. doi:10.1186/1743-8977-7-5
Dong J, Ma Q (2015a) Advances in mechanisms and signaling pathways of carbon nanotube toxicity. Nanotoxicology 9:658–676. doi:10.3109/17435390.2015.1009187
Dong J, Ma Q (2015b) Suppression of basal and carbon nanotube-induced oxidative stress, inflammation and fibrosis in mouse lungs by Nrf2. Nanotoxicology. doi:10.3109/17435390.2015.1110758
Dong J, Porter DW, Batteli LA, Wolfarth MG, Richardson DL, Ma Q (2015a) Pathologic and molecular profiling of rapid-onset fibrosis and inflammation induced by multi-walled carbon nanotubes. Arch Toxicol 89(4):621–633. doi:10.1007/s00204-014-1428-y
Dong J, Yu X, Porter DW, Battelli LA, Kashon ML, Ma Q (2015b) Common and distinct mechanisms of induced pulmonary fibrosis by particulate and soluble chemical fibrogenic agents. Arch Toxicol 90(2):385–402. doi:10.1007/s00204-015-1589-3
Duffield JS, Lupher M, Thannickal VJ, Wynn TA (2013) Host responses in tissue repair and fibrosis. Ann Rev Pathol 8:241–276. doi:10.1146/annurev-pathol-020712-163930
Han YG, Xu J, Li ZG, Ren GG, Yang Z (2012) In vitro toxicity of multi-walled carbon nanotubes in C6 rat glioma cells. Neurotoxicology 33(5):1128–1134. doi:10.1016/j.neuro.2012.06.004
He X, Young SH, Schwegler-Berry D, Chisholm WP, Fernback JE, Ma Q (2011) Multiwalled carbon nanotubes induce a fibrogenic response by stimulating reactive oxygen species production, activating NF-kappaB signaling, and promoting fibroblast-to-myofibroblast transformation. Chem Res Toxicol 24(12):2237–2248. doi:10.1021/tx200351d
Huang X, Zhang F, Sun X et al (2014) The genotype-dependent influence of functionalized multiwalled carbon nanotubes on fetal development. Biomaterials 35(2):856–865
Husain AN, Kumar V (2005) The lung. In: Kumar V, Abbas AK, Fausto N (eds) Robbins and cotran pathologic basis of disease, 7th edn. Elsevier Saunders, Philadelphia, pp 711–772
Johnston HJ, Hutchison GR, Christensen FM et al (2010) A critical review of the biological mechanisms underlying the in vivo and in vitro toxicity of carbon nanotubes: the contribution of physico-chemical characteristics. Nanotoxicology 4(2):207–246. doi:10.3109/17435390903569639
Lam CW, James JT, McCluskey R, Hunter RL (2004) Pulmonary toxicity of single-wall carbon nanotubes in mice 7 and 90 days after intratracheal instillation. Toxicol Sci 77(1):126–134. doi:10.1093/toxsci/kfg243
Ma-Hock L, Strauss V, Treumann S et al (2013) Comparative inhalation toxicity of multi-wall carbon nanotubes, graphene, graphite nanoplatelets and low surface carbon black. Part Fibre Toxicol 10:23. doi:10.1186/1743-8977-10-23
Morgan WKC, Seaton A (1995) Occupational lung diseases, 3rd edn. W.B. Saunders Company, Philadelphia
Porter DW, Hubbs AF, Mercer RR et al (2010) Mouse pulmonary dose- and time course-responses induced by exposure to multi-walled carbon nanotubes. Toxicology 269(2–3):136–147. doi:10.1016/j.tox.2009.10.017
Ravichandran P, Baluchamy S, Sadanandan B et al (2010) Multiwalled carbon nanotubes activate NF-kappaB and AP-1 signaling pathways to induce apoptosis in rat lung epithelial cells. Apoptosis Int J Program Cell Death 15(12):1507–1516. doi:10.1007/s10495-010-0532-6
Reddy AR, Reddy YN, Krishna DR, Himabindu V (2012) Pulmonary toxicity assessment of multiwalled carbon nanotubes in rats following intratracheal instillation. Environ Toxicol 27(4):211–219. doi:10.1002/tox.20632
Rydman EM, Ilves M, Koivisto AJ et al (2014) Inhalation of rod-like carbon nanotubes causes unconventional allergic airway inflammation. Part Fibre Toxicol 11:48. doi:10.1186/s12989-014-0048-2
Shipkowski KA, Taylor AJ, Thompson EA et al (2015) An allergic lung microenvironment suppresses carbon nanotube-induced inflammasome activation via STAT6-dependent inhibition of caspase-1. PLoS ONE 10(6):e0128888. doi:10.1371/journal.pone.0128888
Shvedova AA, Kisin ER, Mercer R et al (2005) Unusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in mice. Am J Physiol Lung Cell Mol Physiol 289(5):L698–L708. doi:10.1152/ajplung.00084.2005
Siegrist KJ, Reynolds SH, Kashon ML et al (2014) Genotoxicity of multi-walled carbon nanotubes at occupationally relevant doses. Part Fibre Toxicol 11:6. doi:10.1186/1743-8977-11-6
Takagi A, Hirose A, Futakuchi M, Tsuda H, Kanno J (2012) Dose-dependent mesothelioma induction by intraperitoneal administration of multi-wall carbon nanotubes in p53 heterozygous mice. Cancer Sci 103(8):1440–1444. doi:10.1111/j.1349-7006.2012.02318.x
Wang P, Nie X, Wang Y et al (2013) Multiwall carbon nanotubes mediate macrophage activation and promote pulmonary fibrosis through TGF-beta/Smad signaling pathway. Small 9(22):3799–3811. doi:10.1002/smll.201300607
Wynn TA (2004) Fibrotic disease and the T(H)1/T(H)2 paradigm. Nat Rev Immunol 4(8):583–594. doi:10.1038/nri1412
Wynn TA (2015) Type 2 cytokines: mechanisms and therapeutic strategies. Nat Rev Immunol 15(5):271–282. doi:10.1038/nri3831
Wynn TA, Ramalingam TR (2012) Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med 18(7):1028–1040. doi:10.1038/nm.2807
Zhang Q, Huang JQ, Qian WZ, Zhang YY, Wei F (2013) The road for nanomaterials industry: a review of carbon nanotube production, post-treatment, and bulk applications for composites and energy storage. Small 9(8):1237–1265. doi:10.1002/smll.201203252
Acknowledgments
This work was funded to QM by National Institute for Occupational Safety and Health, Health Effects Laboratory Division and Nanotechnology Research Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Additional information
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, J., Ma, Q. In vivo activation of a T helper 2-driven innate immune response in lung fibrosis induced by multi-walled carbon nanotubes. Arch Toxicol 90, 2231–2248 (2016). https://doi.org/10.1007/s00204-016-1711-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1711-1