Abstract
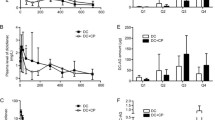
The use of diclofenac (DCF), a nonsteroidal anti-inflammatory drug, is associated with a high prevalence of gastrointestinal side effects. In vivo studies in rodents suggested that reactive metabolites of DCF produced by the liver or the intestine might be responsible for this toxicity. In the present study, precision-cut intestinal slices (PCIS) prepared from the jejunum of 18 human donors were used as an ex vivo model to investigate whether DCF intestinal metabolites are responsible for its intestinal toxicity in man. PCIS were incubated with a concentration range of DCF (0–600 µM) up to 24 h. DCF (≥400 µM) caused direct toxicity to the intestine as demonstrated by ATP depletion, morphological damage, caspase 3 activation, and lactate dehydrogenase leakage. Three main metabolites produced by PCIS (4′-hydroxy DCF, 5-hydroxy DCF, and DCF acyl glucuronide) were detected by HPLC. Protein adducts were detected by immunohistochemical staining and showed correlation with the intestinal metabolites. DCF induced similar toxicity to each of the samples regardless of the variation in metabolism among them. Less metabolites were produced by slices incubated with 400 µM DCF than with 100 µM DCF. The addition of the metabolic inhibitors such as ketoconazole, cimetidine, or borneol decreased the metabolite formation but increased the toxicity. The results suggest that DCF can induce intestinal toxicity in human PCIS directly at therapeutically relevant concentrations, independent of the reactive metabolites 4′-OH DCF, 5-OH DCF, or diclofenac acylglucuronide produced by the liver or formed in the intestine.






Similar content being viewed by others
References
Allison MC, Howatson AG, Torrance CJ, Lee FD, Russell RI (1992) Gastrointestinal damage associated with the use of nonsteroidal antiinflammatory drugs. N Engl J Med 327:749–754
Atchison CR, West AB, Balakumaran A, Hargus SJ, Pohl LR, Daiker DH, Aronson JF, Hoffmann WE, Shipp BK, Treinen-Moslen M (2000) Drug enterocyte adducts: possible causal factor for diclofenac enteropathy in rats. Gastroenterology 119:1537–1547
Bansal R, Prakash J, de Ruijter M, Beljaars L, Poelstra K (2011) Peptide-modified albumin carrier explored as a novel strategy for a cell-specific delivery of interferon gamma to treat liver fibrosis. Mol Pharm 8:1899–1909
Bjarnason I, Hayllar J, MacPherson AJ, Russell AS (1993) Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 104:1832–1847
Bjornsson E, Westgaard G, Bjarnason I (2008) Severe injury to the small bowel associated with a short course of diclofenac. Scand J Gastroenterol 43:759–760
Boelsterli UA, Ramirez-Alcantara V (2011) NSAID acyl glucuronides and enteropathy. Curr Drug Metab 12:245–252
Boelsterli UA, Redinbo MR, Saitta KS (2013) Multiple NSAID-induced hits injure the small intestine: underlying mechanisms and novel strategies. Toxicol Sci 131:654–667
Bort R, Ponsoda X, Jover R, Gomez-Lechon MJ, Castell JV (1999) Diclofenac toxicity to hepatocytes: a role for drug metabolism in cell toxicity. J Pharmacol Exp Ther 288:65–72
Boushey RP, Yusta B, Drucker DJ (1999) Glucagon-like peptide 2 decreases mortality and reduces the severity of indomethacin-induced murine enteritis. Am J Physiol 277:E937–E947
Cheng YT, Wu CH, Ho CY, Yen GC (2012) Catechin protects against ketoprofen-induced oxidative damage of the gastric mucosa by up-regulating Nrf2 in vitro and in vivo. J Nutr Biochem 24:475–483
Cho S, Wood A, Bowlby MR (2007) Brain slices as models for neurodegenerative disease and screening platforms to identify novel therapeutics. Curr Neuropharmacol 5:19–33
Davies NM, Saleh JY, Skjodt NM (2000) Detection and prevention of NSAID-induced enteropathy. J Pharm Pharm Sci 3:137–155
Fortun PJ, Hawkey CJ (2007) Nonsteroidal antiinflammatory drugs and the small intestine. Curr Opin Gastroenterol 23:134–141
Galetin A, Houston JB (2006) Intestinal and hepatic metabolic activity of five cytochrome P450 enzymes: impact on prediction of first-pass metabolism. J Pharmacol Exp Ther 318:1220–1229
Graaf IA, Groothuis GM, Olinga P (2007) Precision-cut tissue slices as a tool to predict metabolism of novel drugs. Expert Opin Drug Metab Toxicol 3:879–898
Groothuis GM, de Graaf IA (2012) Precision-cut intestinal slices as in vitro tool for studies on drug metabolism. Curr Drug Metab 14:112–119
Guengerich FP (2005) Principles of covalent binding of reactive metabolites and examples of activation of bis-electrophiles by conjugation. Arch Biochem Biophys 433:369–378
Hargus SJ, Amouzedeh HR, Pumford NR, Myers TG, McCoy SC, Pohl LR (1994) Metabolic activation and immunochemical localization of liver protein adducts of the nonsteroidal anti-inflammatory drug diclofenac. Chem Res Toxicol 7:575–582
Hui L, Gao S, Liu T, Cao C, Guo J, Hao R, Yi Y, Li C, Zhao Y, Liang A (2011) Evaluation of vitro hepatotoxicity of monocrotaline by precision-cut liver slice technique. Zhongguo Zhong Yao Za Zhi 36:628–632
Kalgutkar AS, Didiuk MT (2009) Structural alerts, reactive metabolites, and protein covalent binding: how reliable are these attributes as predictors of drug toxicity? Chem Biodivers 6:2115–2137
Kararli TT (1995) Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm Drug Dispos 16:351–380
Kasper M, Seidel D, Knels L, Morishima N, Neisser A, Bramke S, Koslowski R (2004) Early signs of lung fibrosis after in vitro treatment of rat lung slices with CdCl2 and TGF-beta1. Histochem Cell Biol 121:131–140
Kenny JR, Maggs JL, Meng X, Sinnott D, Clarke SE, Park BK, Stachulski AV (2004) Syntheses and characterization of the acyl glucuronide and hydroxy metabolites of diclofenac. J Med Chem 47:2816–2825
Khojasteh SC, Oishi S, Nelson SD (2010) Metabolism and toxicity of menthofuran in rat liver slices and in rats. Chem Res Toxicol 23:1824–1832
Kretz-Rommel A, Boelsterli UA (1993) Diclofenac covalent protein binding is dependent on acyl glucuronide formation and is inversely related to P450-mediated acute cell injury in cultured rat hepatocytes. Toxicol Appl Pharmacol 120:155–161
Kretz-Rommel A, Boelsterli UA (1994) Mechanism of covalent adduct formation of diclofenac to rat hepatic microsomal proteins. Retention of the glucuronic acid moiety in the adduct. Drug Metab Dispos 22:956–961
Laine L, Goldkind L, Curtis SP, Connors LG, Yanqiong Z, Cannon CP (2009) How common is diclofenac-associated liver injury? Analysis of 17,289 arthritis patients in a long-term prospective clinical trial. Am J Gastroenterol 104:356–362
Lim MS, Lim PL, Gupta R, Boelsterli UA (2006) Critical role of free cytosolic calcium, but not uncoupling, in mitochondrial permeability transition and cell death induced by diclofenac oxidative metabolites in immortalized human hepatocytes. Toxicol Appl Pharmacol 217:322–331
LoGuidice A, Ramirez-Alcantara V, Proli A, Gavillet B, Boelsterli UA (2010) Pharmacologic targeting or genetic deletion of mitochondrial cyclophilin D protects from NSAID-induced small intestinal ulceration in mice. Toxicol Sci 118:276–285
LoGuidice A, Wallace BD, Bendel L, Redinbo MR, Boelsterli UA (2012) Pharmacologic targeting of bacterial beta-glucuronidase alleviates nonsteroidal anti-inflammatory drug-induced enteropathy in mice. J Pharmacol Exp Ther 341:447–454
Macpherson AB, Jarnason I (1993) NSAIDS and gastropathy. Pharmacoeconomics 3:83–84
Maiden L, Thjodleifsson B, Theodors A, Gonzalez J, Bjarnason I (2005) A quantitative analysis of NSAID-induced small bowel pathology by capsule enteroscopy. Gastroenterology 128:1172–1178
Maiden L, Thjodleifsson B, Seigal A, Bjarnason II, Scott D, Birgisson S, Bjarnason I (2007) Long-term effects of nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 selective agents on the small bowel: a cross-sectional capsule enteroscopy study. Clin Gastroenterol Hepatol 5:1040–1045
McConnell EL, Fadda HM, Basit AW (2008) Gut instincts: explorations in intestinal physiology and drug delivery. Int J Pharm 364:213–226
Miners JO, Birkett DJ (1998) Cytochrome P4502C9: an enzyme of major importance in human drug metabolism. Br J Clin Pharmacol 45:525–538
Obach RS, Zhang QY, Dunbar D, Kaminsky LS (2001) Metabolic characterization of the major human small intestinal cytochrome p450s. Drug Metab Dispos 29:347–352
O’Brien PJ, Irwin W, Diaz D, Howard-Cofield E, Krejsa CM, Slaughter MR, Gao B, Kaludercic N, Angeline A, Bernardi P, Brain P, Hougham C (2006) High concordance of drug-induced human hepatotoxicity with in vitro cytotoxicity measured in a novel cell-based model using high content screening. Arch Toxicol 80:580–604
Ponsoda X, Bort R, Jover R, Gomez-Lechon MJ, Castell JV (1995) Molecular mechanism of diclofenac hepatotoxicity: association of cell injury with oxidative metabolism and decrease in ATP levels. Toxicol In Vitro 9:439–444
Poon GK, Chen Q, Teffera Y, Ngui JS, Griffin PR, Braun MP, Doss GA, Freeden C, Stearns RA, Evans DC, Baillie TA, Tang W (2001) Bioactivation of diclofenac via benzoquinone imine intermediates-identification of urinary mercapturic acid derivatives in rats and humans. Drug Metab Dispos 29:1608–1613
Power I, Cumming AD, Pugh GC (1992) Effect of diclofenac on renal function and prostacyclin generation after surgery. Br J Anaesth 69:451–456
Pumford NR, Myers TG, Davila JC, Highet RJ, Pohl LR (1993) Immunochemical detection of liver protein adducts of the nonsteroidal antiinflammatory drug diclofenac. Chem Res Toxicol 6:147–150
Quinn CM, Bjarnason I, Price AB (1993) Gastritis in patients on non-steroidal anti-inflammatory drugs. Histopathology 23:341–348
Roskott AM, Nieuwenhuijs VB, Leuvenink HG, Dijkstra G, Ottens P, de Jager MH, Gonalves Dias Pereira P, Fidler V, Groothuis GM, Ploeg RJ, de Graaf IA (2010) Reduced ischemia-reoxygenation injury in rat intestine after luminal preservation with a tailored solution. Transplantation 90:622–629
Sarda S, Page C, Pickup K, Schulz-Utermoehl T, Wilson I (2012) Diclofenac metabolism in the mouse: novel in vivo metabolites identified by high performance liquid chromatography coupled to linear ion trap mass spectrometry. Xenobiotica 42:179–194
Seitz S, Boelsterli UA (1998) Diclofenac acyl glucuronide, a major biliary metabolite, is directly involved in small intestinal injury in rats. Gastroenterology 115:1476–1482
Seitz S, Kretz-Rommel A, Oude Elferink RP, Boelsterli UA (1998) Selective protein adduct formation of diclofenac glucuronide is critically dependent on the rat canalicular conjugate export pump (Mrp2). Chem Res Toxicol 11:513–519
Sibilia J, Deray G, Montalescot G (2006) What do we know about the cardiovascular toxicity of the NSAIDs? Presse Med 35(Suppl 1):11–23
Somasundaram S, Rafi S, Hayllar J, Sigthorsson G, Jacob M, Price AB, Macpherson A, Mahmod T, Scott D, Wrigglesworth JM, Bjarnason I (1997) Mitochondrial damage: a possible mechanism of the “topical” phase of nsaid induced injury to the rat intestine. Gut 41:344–353
Somasundaram S, Sigthorsson G, Simpson RJ, Watts J, Jacob M, Tavares IA, Rafi S, Roseth A, Foster R, Price AB, Wrigglesworth JM, Bjarnason I (2000) Uncoupling of intestinal mitochondrial oxidative phosphorylation and inhibition of cyclooxygenase are required for the development of NSAID-enteropathy in the rat. Aliment Pharmacol Ther 14:639–650
Strassburg CP, Kneip S, Topp J, Obermayer-Straub P, Barut A, Tukey RH, Manns MP (2000) Polymorphic gene regulation and interindividual variation of UDP-glucuronosyltransferase activity in human small intestine. J Biol Chem 275:36164–36171
Tang W (2003) The metabolism of diclofenac-enzymology and toxicology perspectives. Curr Drug Metab 4:319–329
Tang W, Stearns RA, Bandiera SM, Zhang Y, Raab C, Braun MP, Dean DC, Pang J, Leung KH, Doss GA, Strauss JR, Kwei GY, Rushmore TH, Chiu SH, Baillie TA (1999) Studies on cytochrome P-450-mediated bioactivation of diclofenac in rats and in human hepatocytes: identification of glutathione conjugated metabolites. Drug Metab Dispos 27:365–372
van de Kerkhof EG, de Graaf IA, de Jager MH, Meijer DK, Groothuis GM (2005) Characterization of rat small intestinal and colon precision-cut slices as an in vitro system for drug metabolism and induction studies. Drug Metab Dispos 33:1613–1620
van de Kerkhof EG, Ungell AL, Sjoberg AK, de Jager MH, Hilgendorf C, de Graaf IA, Groothuis GM (2006) Innovative methods to study human intestinal drug metabolism in vitro: precision-cut slices compared with using chamber preparations. Drug Metab Dispos 34:1893–1902
van de Kerkhof EG, de Graaf IA, Groothuis GM (2007) In vitro methods to study intestinal drug metabolism. Curr Drug Metab 8:658–675
Vickers AE, Rose K, Fisher R, Saulnier M, Sahota P, Bentley P (2004) Kidney slices of human and rat to characterize cisplatin-induced injury on cellular pathways and morphology. Toxicol Pathol 32:577–590
Wang AG, Xia T, Yuan J, Yu RA, Yang KD, Chen XM, Qu W, Waalkes MP (2004) Effects of phenobarbital on metabolism and toxicity of diclofenac sodium in rat hepatocytes in vitro. Food Chem Toxicol 42:1647–1653
Watkins JB, Klaassen CD (1983) Chemically-induced alteration of UDP-glucuronic acid concentration in rat liver. Drug Metab Dispos 11:37–40
Wolfe MM, Lichtenstein DR, Singh G (1999) Gastrointestinal toxicity of nonsteroidal antiinflammatory drugs. N Engl J Med 340:1888–1899
Zhu Y, Zhang QY (2012) Role of intestinal cytochrome p450 enzymes in diclofenac-induced toxicity in the small intestine. J Pharmacol Exp Ther 343:362–370
Acknowledgments
The authors thank Dr. J. H. Proost and Dr. A. Taneja (University of Groningen) for their advice on the statistical analysis. Dr. Leonie Beljaars (University of Groningen) is acknowledged for her advice on the staining intensity measurement.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
204_2014_1242_MOESM1_ESM.tif
Supplementary table 1 Inhibition of metabolites formation raw values. Compared with DCF200 µM alone, the addition of borneol (B), ketoconazole (K), cimetidine (C) inhibited the formation of DAG, 4′-OH DCF, 5-OH DCF, respectively. UD: under detection level. NA: not analyzed. (TIFF 5608 kb)
Rights and permissions
About this article
Cite this article
Niu, X., de Graaf, I.A.M., Langelaar-Makkinje, M. et al. Diclofenac toxicity in human intestine ex vivo is not related to the formation of intestinal metabolites. Arch Toxicol 89, 107–119 (2015). https://doi.org/10.1007/s00204-014-1242-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1242-6