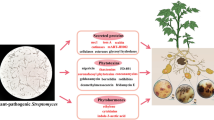
Abstract
Potato crop, currently, is the staple food crop of about 1.3 billion global population. Potato is attaining even more admiration globally day by day owing to its public acceptability. However, potato sustainable production is distinctly challenged by multiple factors like diseases, pests and climate change etc. Among diseases, common scab is one of the prime threats to potato crop due to its soil-borne nature and versatility in phytotoxins’ secretion. Common scab is caused multiple number of phytopathogenic streptomyces strains. Despite extensive research programs, researchers are still unable to identify a significant solution to this threat that is proliferating exceptional rate across the globe. To develop feasible remedies, adequate information regarding host–pathogen interaction should be available. This review possesses insights on existing pathogenic species, the evolution of novel pathogenic streptomyces spp. and phytotoxins produced by the pathogenic strains. Furthermore, which type of physiological, biochemical and genetic activities occur during pathogen’s infestation of the host are also canvassed.




Similar content being viewed by others
Data availability
The authors confirm that the data supporting the review are available within the article.
References
Adolf B, Andrade-Piedra J, Bittara Molina F, Przetakiewicz J, Hausladen H, Kromann P, Lees A, Lindqvist-Kreuze H, Perez W, Secor GA (2020) Fungal, oomycete, and plasmodiophorid diseases of potato. The potato crop: its agricultural, nutritional and social contribution to humankind. Springer International Publishing, Cham
Barry SM, Kers JA, Johnson EG, Song L, Aston PR, Patel B, Krasnoff SB, Crane BR, Gibson DM, Loria R, Challis GL (2012) Cytochrome P450-catalyzed L-tryptophan nitration in thaxtomin phytotoxin biosynthesis. Nat Chem Biol 8(10):814–816
Bernards MA, Razem FA (2001) The poly(phenolic) domain of potato suberin: a non-lignin cell wall bio-polymer. Phytochem 57:1115–1122
Bibb MJ (2005) Regulation of secondary metabolism in streptomycetes. Curr Opin Microbiol 8:208–215
Bignell DR, Seipke RF, Huguet-Tapia JC, Chambers AH, Parry RJ, Loria R (2010) Streptomyces scabies 87–22 contains a coronafacic acid-like biosynthetic cluster that contributes to plant–microbe interactions. Mol Plant Microbe Interact 23(2):161–175
Bignell DRD, Francis IM, Fyans JK, Loria R (2014a) Thaxtomin A production and virulence are controlled by several bld gene global regulators in Streptomyces scabies. Mol Plant Microbe Interact 27:875–885
Bignell DRD, Fyans JK, Cheng Z (2014b) Phytotoxins produced by plant pathogenic Streptomyces species. J App Microbiol 116:223–235
Bignell DRD, Cheng Z, Bown L (2018) The coronafacoyl phytotoxins: structure, biosynthesis, regulation and biological activities. Antonie Van Leeuwenhoek 111:649–666
Bischoff V, Cookson SJ, Wu S, Scheible WR (2009) Thaxtomin A affects CESA-complex density, expression of cell wall genes, cell wall composition, and causes ectopic lignification in Arabidopsis thaliana seedlings. J Exp Bot 60:955–965
Bordeleau E, Ghinet MG, Burrus V (2012) Diversity of integrating conjugative elements in actinobacteria. Mob Gen Elements 2:119–124
Bown L, Li Y, Berrué F, Verhoeven JT, Dufour SC, Bignell DRD (2017) Coronafacoyl phytotoxin biosynthesis and evolution in the common scab pathogen Streptomyces scabiei. App Environ Microbiol 83(19):e01169-e1217
Bukhalid RA, Chung SY, Loria R (1998) nec1, a gene conferring a necrogenic phenotype, is conserved in plant-pathogenic Streptomyces spp. and linked to a transposase pseudogene. Mol Plant Microbe Interact 11:960–967
Camire ME, Kubow S, Donnelly DJ (2009) Potatoes and human health. Cri Rev Food Sci Nut 49:823–840
Cao Z, Khodakaramian G, Arakawa K, Kinashi H (2012) Isolation of borrelidin as a phytotoxic compound from a potato pathogenic Streptomyces strain. Biosci Biotech Biochem 76(2):353–357
Chapleau M, Guertin JF, Farrokhi A, Lerat S, Burrus V, Beaulieu C (2016) Identification of genetic and environmental factors stimulating excision from Streptomyces scabiei chromosome of the toxicogenic region responsible for pathogenicity: the toxicogenic region in S. scabiei strains. Mol Plant Pathol 17:501–509
Charkowski A, Sharma K, Parker ML, Secor GA, Elphinstone J (2020) Bacterial diseases of potato. The potato crop: its agricultural, nutritional and social contribution to humankind. Springer International Publishing, Cham, pp 351–388
Chater KF, Chandra G (2008) The use of the rare UUA codon to de-fine “expression space” for genes involved in secondary metabolism, development and environmental adaptation in Streptomyces. J Microbiol 46:1–11
Cheng Z, Bown L, Tahlan K, Bignell DRD (2015) Regulation of coronafacoyl phytotoxin production by the PAS-LuxR family regulator CfaR in the common scab pathogen Streptomyces scabies. PLoS ONE 10(3):e0122450
Chu BC, Garcia-Herrero A, Johanson TH, Krewulak KD, Lau CK, Peacock RS, Slavinskaya Z, Vogel HJ (2010) Siderophore uptake in bacteria and the battle for iron with the host; a bird’s eye view. Biometals 23(4):601–611
Connorton JM, Balk J (2019) Iron biofortification of staple crops: lessons and challenges in plant genetics. Plant Cell Physiol 60:1447–1456
Craig M, Lambert S, Jourdan S, Tenconi E, Colson S, Maciejewska M, Ongena M, Martin JF, van Wezel G, Rigali S (2012) Unsuspected control of siderophore production by N-acetylglucosamine in streptomycetes. Environ Microbiol Rep 4:512–521
de Haan S, Burgos G, Liria R, Rodriguez F, Creed-Kanashiro HM, Bonierbale M (2019) The nutritional contribution of potato varietal diversity in Andean food systems: a case study. Am J Pot Res 96(2):151–163
Dees MW, Wanner LA (2012) In search of better management of potato common scab. Potato Res 55:249–268
Deflandre B, Rigali S (2021) Old enzyme, new role: the β-Glucosidase BglC of Streptomyces scabiei interferes with the plant defense mechanism by hydrolyzing scopolin. Biophysica 2(1):1–7
Deflandre B, Thiébaut N, Planckaert S, Jourdan S, Anderssen S, Hanikenne M, Devreese B, Francis I, Rigali S (2020) Deletion of bglC triggers a genetic compensation response by awakening the expression of alternative beta-glucosidase. Biochim Biophys Acta - Gene Reg Mech 1863(10):194615
Deflandre B, Stulanovic N, Planckaert S, Anderssen S, Bonometti B, Karim L, Coppieters W, Devreese B, Rigali S (2022) The virulome of Streptomyces scabiei in response to cello-oligosaccharide elicitors. Microb Gen 8(1)
Díaz-Cruz GA, Liu J, Tahlan K, Bignell DR (2022) Nigericin and geldanamycin are phytotoxic specialized metabolites produced by the plant pathogen Streptomyces sp 11-1-2. Microbiol Spect 10(2):e02314-e2321
Errakhi R, Dauphin A, Meimoun P, Lehner A, Reboutier D, Vatsa P, Briand J, Madiona K, Rona JP, Barakate M, Wendehenne D (2008) An early Ca2+ influx is a prerequisite to thaxtomin A-induced cell death in Arabidopsis thaliana cells. J Exp Bot 59(15):4259–4270
FAO (2021) http://www.fao.org/faostat/en/#data/QC. Accessed 16 Apr 2023
Francis IM, Jourdan S, Fanara S, Loria R, Rigali S (2015) The cellobiose sensor CebR is the gatekeeper of Streptomyces scabies pathogenicity. Mbio 6:e02018-e12014
Fukuchi N, Furihata K, Takayama S, Isogai A, Suzuki A (1992a) Rotihibin A, a novel plant growth regulator, from Streptomyces sp. Biosci Biotech Biochem 56(5):840–841
Fukuchi N, Nakayama J, Takayama S, Isogai A, Suzuki A (1992b) Structural elucidation of rotihibin B by tandem mass spectrometry. Biosci Biotech Biochem 56(7):1152–1153
Fukuchi N, Furihata K, Nakayama J, Toshinari G, Takayama S, Isogai A, Suzuki A (1995) Rotihibins, novel plant growth regulators from Streptomyces graminofadens. J Antibio 48(9):1004–1010
Fuqua WC, Winans SC, Greenberg EP (1994) Quorum sensing in bacteria: the LuxR-LuxI family of cell density-responsive transcriptional regulators. J Bacteriol 176:269–275
Fyans JK, Bown L, Bignell DRD (2016) Isolation and characterization of plant-pathogenic Streptomyces species associated with common scab-infected potato tubers in Newfoundland. Phytopathol 106:123–131
Guan D, Grau BL, Clark CA, Taylor CM, Loria R, Pettis GS (2012) Evidence that thaxtomin C is a pathogenicity determinant of Streptomyces ipomoeae, the causative agent of Streptomyces soil rot disease of sweet potato. Mol Plant Microbe Interact 25:393–401
Gudmestad NC, Taylor RJ, Pasche JS (2007) Management of soilborne diseases of potato. Australas Plant Pathol 36:109–115
Halder V, Oeljeklaus J, Heilmann G, Krahn JH, Liu Y, Xiong Y, Schlicht M, Schillinger J, Kracher B, Ehrmann M, Kombrink E (2018) Identification of the natural product rotihibin A as a TOR kinase signaling inhibitor by unbiased transcriptional profiling. Eur J Chem 24(48):12500–12504
Healy FG, Krasnoff SB, Wach M, Gibson DM, Loria R (2002) Involvement of a cytochrome P450 monooxygenase in thaxtomin A biosynthesis by Streptomyces acidiscabies. J Bacteriol 184:2019–2029
Henrissat B (1991) A classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem J 280:309–316
Herbst DA, Boll B, Zocher G, Stehle T, Heide L (2013) Structural basis of the interaction of MbtH-like proteins, putative regulators of nonribosomal peptide biosynthesis, with adenylating enzymes. J Biol Chem 288:1991–2003
Hill J, Lazarovits G (2005) A mail survey of growers to estimate potato common scab prevalence and economic loss in Canada. Can J Plant Pathol 27:46–52
Hopwood DA (2007) Streptomyces in nature and medicine: the antibiotic makers. Oxford University Press, New York, NY
Hsu SY (2010) IAA production by Streptomyces scabies and its role in plant microbe interaction. Ph. D. thesis. Ithaca NY: Cornell University.
Hudec C, Novinscak A, Filion M (2021) Diversity and virulence of Streptomyces spp. causing potato common scab in Prince Edward Island. Canada Phytopathol 111:617–626
Huguet-Tapia JC, Bignell DRD, Loria R (2014) Characterization of the integration and modular excision of the integrative conjugative element PAISt in Streptomyces turgidiscabies Car8. PLoS ONE 9:e99345
Huguet-Tapia JC, Lefebure T, Badger JH, Guan D, Pettis GS, Stanhope MJ, Loria R (2016) Genome content and phylogenomics reveal both ancestral and lateral evolutionary pathways in plant-pathogenic Streptomyces species. App Environ Microbiol 82(7):2146–2155
Hunt AC, Servin-Gonzalez L, Kelemen GH, Buttner MJ (2005) The bldC developmental locus of Streptomyces coelicolorencodes a member of a family of small DNA-binding proteins related to the DNA-binding domains of the MerR family. J Bacteriol 187:716–728
Jabloune R, Khalil M, Moussa IEB, Simao-Beaunoir AM, Lerat S, Brzezinski R, Beaulieu C (2020) Enzymatic degradation of p-nitrophenyl esters, polyethylene terephthalate, cutin, and suberin by Sub1, a suberinase encoded by the plant pathogen Streptomyces scabies. Microbes Environ 35(1):ME19086
Jiang G, Zuo R, Zhang Y, Powell MM, Zhang P, Hylton SM, Loria R, Ding Y (2018) One-pot biocombinatorial synthesis of herbicidal thaxtomins. ACS Cat 8(11):10761–10768
Johnson EG, Joshi MV, Gibson DM, Loria R (2007) Cello-oligosaccharides released from host plants induce pathogenicity in scab-causing Streptomyces species. Physiol Mol Plant Pathol 71(1–3):18–25
Johnson EG, Krasnoff SB, Bignell DRD, Chung WC, Tao T, Parry RJ, Loria R, Gibson DM (2009) 4-Nitrotryptophan is a substrate for the non-ribosomal peptide synthetase TxtB in the thaxtomin A biosynthetic pathway. Mol Microbiol 73(3):409–418
Joshi MV, Loria R (2007) Streptomyces turgidiscabies possesses a functional cytokinin biosynthetic pathway and produces leafy galls. Mol Plant Microbe Interact 20:751–758
Joshi M, Rong X, Moll S, Kers J, Franco C, Loria R (2007a) Streptomyces turgidiscabies secretes a novel virulence protein, Nec1, which facilitates infection. Mol Plant Microbe Interact 20:599–608
Joshi MV, Bignell DRD, Johnson EG, Sparks JP, Gibson DM, Loria R (2007b) The AraC/XylS regulator TxtR modulates thaxtomin biosynthesis and virulence in Streptomyces scabies. Mol Microbiol 66(3):633–642
Joshi MV, Mann SG, Antelmann H, Widdick DA, Fyans JK, Chandra G, Hutchings MI, Toth I, Hecker M, Loria R, Palmer T (2010) The twin arginine protein transport pathway exports multiple virulence proteins in the plant pathogen Streptomyces scabies. Mol Microbiol 77(1):252–271
Jourdan S, Francis IM, Kim MJ, Salazar JJC, Planckaert S, Frère JM, Matagne A, Kerff F, Devreese B, Loria R, Rigali S (2016) The CebE/MsiK transporter is a doorway to the cello-oligosaccharide-mediated induction of Streptomyces scabies pathogenicity. Sci Rep 6(1):1–12
Jourdan S, Francis IM, Deflandre B, Tenconi E, Riley J, Planckaert S, Tocquin P, Martinet L, Devreese B, Loria R, Rigali S (2018) Contribution of the β-glucosidase BglC to the onset of the pathogenic lifestyle of Streptomyces scabies. Mol Plant Pathol 19(6):1480–1490
Juhas M, van der Meer JR, Gaillard M, Harding RM, Hood DW, Crook DW (2009) Genomic islands: tools of bacterial horizontal transfer and evolution. FEMS Microbiol Rev 33:376–393
Katsir L, Schilmiller AL, Staswick PE, He SY, Howe GA (2008) COI1 is a critical component of a receptor for jasmonate and the bacterial virulence factor coronatine. Proceed Nat Acad Sci 105:7100–7105
Kers JA, Cameron KD, Joshi MV, Bukhalid RA, Morello JE, Wach MJ, Gibson DM, Loria R (2005) A large, mobile pathogenicity island confers plant pathogenicity on Streptomyces species. Mol Microbiol 55:1025–1033
King J, Slavin J (2013) White potatoes, human health, and dietary guidance. Adv Nut 4:393S-401S
Komeil D, Simao-Beaunoir AM, Beaulieu C (2013) Detection of potential suberinase-encoding genes in Streptomyces scabiei strains and other actinobacteria. Can J Microbiol 59:294–303
Komeil D, Padilla-Reynaud R, Lerat S, Simao-Beaunoir AM, Beaulieu C (2014) Comparative secretome analysis of Streptomyces scabiei during growth in the presence or absence of potato suberin. Prot Sci 12(1):1–16
Lapaz MI, López A, Huguet-Tapia JC, Pérez-Baldassari MF, Iglesias C, Loria R, Moyna G, Pianzzola MJ (2019) Isolation and structural characterization of a non-diketopiperazine phytotoxin from a potato pathogenic Streptomyces strain. Nat Prod Res 33(20):2951–2957
Lauzier A, Simao-Beaunoir AM, Bourassa S, Poirier GG, Talbot B, Beaulieu C (2008) Effect of potato suberin on Streptomyces scabies proteome. Mol Plant Pathol 9(6):753–762
Legault GS, Lerat S, Nicolas P, Beaulieu C (2011) Tryptophan regulates thaxtomin A and indole-3-acetic acid production in Streptomyces scabiei and modifies its interactions with radish seedlings. Phytopathol 101:1045–1051
Lerat S, Simao-Beaunoir AM, Beaulieu C (2009) Genetic and physiological determinants of Streptomyces scabies pathogenicity. Mol Plant Pathol 10:579–585
Li Y, Liu J, Adekunle D, Bown L, Tahlan K, Bignell DRD (2019) TxtH is a key component of the thaxtomin biosynthetic machinery in the potato common scab pathogen Streptomyces scabies. Mol Plant Pathol 20(10):1379–1393
Liu J, Nothias LF, Dorrestein PC, Tahlan K, Bignell DRD (2021) Genomic and metabolomic analysis of the potato common scab pathogen Streptomyces scabiei. ACS Omega 6(17):11474–11487
Loria R, Kers J, Joshi M (2006) Evolution of plant pathogenicity in Streptomyces. Ann Rev Phytopathol 44:469–487
Mata CG, Lamattina L, Cassia RO (2001) Involvement of iron and ferritin in the potato–Phytophthora infestans interaction. Eur J Plant Pathol 107:557–562
Natsume M, Yamada A, Tashiro N, Abe H (1998) Differential production of the phytotoxin thaxtomin A and concanamycins A and B by potato common scab-causing Streptomyces spp. Ann Phytopathol Soc Jpn 64:202–204
Natsume M, Komiya M, Koyanagi F, Tashiro N, Kawaide H, Abe H (2005) Phytotoxin produced by Streptomyces sp. causing potato russet scab in Japan. J Gen Plant Pathol 71(5):364–369
Natsume M, Tashiro N, Doi A, Nishi Y, Kawaide H (2017) Effects of concanamycins produced by Streptomyces scabies on lesion type of common scab of potato. J Gen Plant Pathol 83:78–82
Natsume M, Nagagata A, Aittamaa M, Okaniwa N, Somervuo P, Fiedler HP, Kreuze JF, Rokka VM, Bång H, Kawaide H, Valkonen J (2018) Phytotoxin produced by the netted scab pathogen, Streptomyces turgidiscabies strain 65, isolated in Sweden. J Gen Plant Pathol 84(2):108–117
Nisa T, Haq MI, Mukhtar T, Khan MA, Irshad G (2022) Incidence and severity of common scab of potato caused by Streptomyces scabies in Punjab. Pakistan Pak J Bot 54(2):723–729
Oda T, Hirabayashi H, Shikauchi G, Takamura R, Hiraga K, Minami H, Hashimoto H, Yamamoto M, Wakabayashi K, Shimizu T, Sato M (2017) Structural basis of autoinhibition and activation of the DNA-targeting ADP-ribosyltransferase pierisin-1. J Biol Chem 292(37):15445–15455
Padilla-Reynaud R, Simao-Beaunoir AM, Lerat S, Bernards MA, Beaulieu C (2015) Suberin regulates the production of cellulolytic enzymes in Streptomyces scabiei, the causal agent of potato common scab. Microb Environ 30:245–253
Patel P, Song L, Challis GL (2010) Distinct extracytoplasmic siderophore binding proteins recognize ferrioxamines and ferricoelichelin in Streptomyces coelicolor A3(2). Biochem 49(37):8033–8042
Planckaert S, Jourdan S, Francis IM, Deflandre B, Rigali S, Devreese B (2018) Proteomic response to thaxtomin phytotoxin elicitor cellobiose and to deletion of cellulose utilization regulator CebR in Streptomyces scabies. J Prot Res 17(11):3837–3852
Planckaert S, Deflandre B, De Vries AM, Ameye M, Martins JC, Audenaert K, Rigali S, Devreese B (2021) Identification of novel rotihibin analogues in Streptomyces scabies, including discovery of its biosynthetic gene cluster. Microbiol Spect 9(1):e00571-e621
Pollard M, Beisson F, Li Y, Ohlrogge JB (2008) Building lipid barriers: biosynthesis of cutin and suberin. Trends Plant Sci 13:236–246
Santos-Aberturas J, Vicente CM, Guerra SM, Payero TD, Martin JF, Aparicio JF (2011) Molecular control of polyene macrolide biosynthesis: direct binding of the regulator PimM to eight promoters of pimaricin genes and identification of binding boxes. J Biol Chem 286:9150–9161
Scheible WR, Fry B, Kochevenko A, Schindelasch D, Zimmerli L, Somerville S, Loria R, Somerville CR (2003) An Arabidopsis mutant resistant to thaxtomin A, a cellulose synthesis inhibitor from Streptomyces species. Plant Cell 15:1781–1794
Seipke RF, Loria R (2008) Streptomyces scabies 87–22 possesses a functional tomatinase. J Bacteriol 190:7684–7692
Seipke RF, Song L, Bicz J, Laskaris P, Yaxley AM, Challis GL, Loria R (2011) The plant pathogen Streptomyces scabies 87–22 has a functional pyochelin biosynthetic pathway that is regulated by TetR-and AfsR-family proteins. Microbiol 157(9):2681–2693
Taylor BL, Zhulin IB (1999) PAS domains: internal sensors of oxygen, redox potential, and light. Microbiol Mol Biol Rev 63:479–506
Wach MJ, Krasnoff SB, Loria R, Gibson DM (2007) Effect of carbohydrates on the production of thaxtomin A by Streptomyces acidiscabies. Arch Microbiol 188:81–88
Wanner LA (2006) A survey of genetic variation in Streptomyces isolates causing potato common scab in the United States. Phytopathol 96(12):1363–1371
Wei Q, Li J, Yang S, Wang W, Min F, Guo M, Zhang S, Dong X, Hu L, Li Z, Wang X (2022) Streptomyces rhizophilus causes potato common scab disease. Plant Dis 106(1):266–274
Weingart H, Ullrich H, Geider K, Völksch B (2001) The role of ethylene production in virulence of Pseudomonas syringae pvs. glycinea and phaseolicola. Phytopathol 91:511–518
Weisberg AJ, Kramer CG, Kotha RR, Luthria DL, Chang J, Clarke CR (2021) A novel species-level group of Streptomyces exhibits variation in phytopathogenicity despite conservation of virulence loci. Mol Plant Microbe Interact 34:39–48
Wilson CR (2004) A summary of common scab disease of potato research from Australia. Proceed Inter Potato Scab Symp 2004. Sapporo, Japan: Hokkaido University 198-214
Wozniak RAF, Waldor MK (2010) Integrative and conjugative elements: mosaic mobile genetic elements enabling dynamic lateral gene flow. Nat Rev Microbiol 8:552–563
Yang J, Tauschek M, Robins-Browne RM (2011) Control of bacterial virulence by AraC-like regulators that respond to chemical signals. Trends Microbiol 19:128–135
Zhang Y, Loria R (2017) Emergence of novel pathogenic Streptomyces species by site-specific accretion and cis-mobilization of pathogenicity islands. Mol Plant Microbe Interact 30:72–82
Zhang Y, Bignell DRD, Zuo R, Fan Q, Huguet-Tapia JC, Ding Y, Loria R (2016) Promiscuous pathogenicity islands and phylogeny of pathogenic Streptomyces spp. Mol Plant Microbe Interact 29(8):640–650
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Conceptualization: IUH, ZM and MAUH. Literature review, draft preparation and revision: IUH, SL and ZM. Editing and Proofreading: IUH, ZM and MAUH.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interests.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Biosynthetic gene cluster
-
An operon commonly encodes a non-ribosomal peptide synthetase and polyketide synthase.
- Cellubiose/cellotriose
-
A di/trisaccharide composed of glucose molecule lined via glycosidic bond.
- Cis-integrative and mobilizable element
-
Genetic mobile element possess ability to integrate and express in recipient/host cell.
- Colonization region
-
Region on TBGC that is involved in the colonization of pathogen in host.
- Elicitor
-
Signal metabolites trigger plant defense upon perception of pathogen.
- GC rich genome
-
Genome composed of high GC content percentage.
- Integrative and conjugative element
-
Mobile genetic elements that can integrate host genome also known as conjugative transposons.
- Pathogenicity island
-
Region of streptomyces genome that imparts pathogenicity feature.
- Phytotoxins
-
Molecules having toxic impacts over plants.
- Siderophores
-
Iron chelating molecules secreted by micro-organisms.
- Thaxtomin A
-
Prime phytotoxin produced by streptomyces spp.
- Toxicogenic region
-
Portion of PAI that encodes phytotoxins.
- Virulome
-
Virulent secretions produce by pathogenic streptomyces spp.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haq, I.U., Mukhtar, Z., Anwar-ul-Haq, M. et al. Deciphering host–pathogen interaction during Streptomyces spp. infestation of potato. Arch Microbiol 205, 222 (2023). https://doi.org/10.1007/s00203-023-03560-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03560-6