Abstract
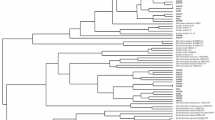
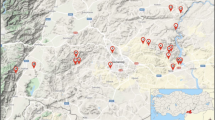
The present work aimed to characterize rhizobia nodulating Trigonella foenum-graecum L. (fenugreek) cultivated in six different geographical locations in Northwestern Morocco. Forty-seven rhizobial isolates from nodules of Trigonella foenum-graecum were grouped into thirteen clusters using the Rep-PCR technique. The phylogenetic analysis based on 16S rRNA gene sequences of 13 groups showed that all representative strains were closely related to members of the genus Ensifer (syn. Sinorhizobium) of the Alphaproteobacteria. All the representative strains shared 100% similarity with Ensifer medicae WSM419T. NodC and nifH gene analysis revealed a close phylogenetic relationship of the representative strains with those of the strains belonging to the symbiovar meliloti. Furthermore, nodulation ability of our rhizobial strains was efficient in their host plant (Trigonella foenum-graecum L.).




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abbes Z, Trabelsi I, Kharrat M, Amri M (2019) Intercropping with Fenugreek (Trigonella Foenum-Graecum) enhanced seed yield and reduced Orobanche Foetida infestation in Faba Bean (Vicia Faba). Biol Agric Hortic 35(4):238–247. https://doi.org/10.1080/01448765.2019.1616614
Ahmad A, Alghamdi SS, Mahmood K, Afzal M (2016) Fenugreek a multipurpose crop: potentialities and improvements. Saudi J Biol Sci 23(2):300–310. https://doi.org/10.1016/j.sjbs.2015.09.015
Ali SF, Lal G, Aishwath O et al (2012) Possibilities and potential of rhizobial inoculants in organic production of fenugreek in arid and semiarid regions of Rajasthan. Int J Seed Spices 2:39–45
Álvarez-Martínez ER, Valverde Á, Ramírez-Bahena MH et al (2009) The analysis of core and symbiotic genes of rhizobia nodulating vicia from different continents reveals their common phylogenetic origin and suggests the distribution of rhizobium leguminosarum strains together with vicia seeds. Arch Microbiol 191(8):659–668. https://doi.org/10.1007/s00203-009-0495-6
Andrews M, Andrews ME (2017) Specificity in legume-rhizobia symbioses. Int J Mol Sci 18(4):705. https://doi.org/10.3390/ijms18040705
Basu SK, Zandi P, Cetzal-Ix W (2019) Fenugreek (Trigonella Foenum-Graecum L.). In: The role of functional food security in global health. pp 471–97. https://doi.org/10.1016/B978-0-12-813148-0.00028-1
Benayad Z, Gómez-Cordovés C, Es-Safi M (2014) Characterization of flavonoid glycosides from fenugreek (Trigonella Foenum-Graecum) crude seeds by HPLC–DAD–ESI/MS analysis. Int J Mol Sci 15(11):20668–20685. https://doi.org/10.3390/ijms151120668
Bitarafan Z, Jensen SM, Liu F, Andreasen C (2019) Intercropping Fenugreek (Trigonella Foenum-Graecum L.) and Barley (Hordeum vulgare L.) with and without Biochar: tests along a competition gradient. J Agron Crop Sci 205(1):99–107. https://doi.org/10.1111/jac.12305
Chahboune R, Barrijal S, Moreno S, Bedmar EJ (2011) Characterization of bradyrhizobium species isolated from root nodules of cytisus Villosus Grown in Morocco. Syst Appl Microbiol 34(6):440–445. https://doi.org/10.1016/j.syapm.2011.04.008
De Bruijn FJ (1992) Use of repetitive (repetitive extragenic palindromic and enterobacterial repetitive intergeneric consensus) sequences and the polymerase chain reaction to fingerprint the genomes of Rhizobium meliloti isolates and other soil bacteria. Appl Environ Microbiol 58(7):2180–2187
De Meyer SE, Van Hoorde K, Vekeman B, Braeckman T, Willems A (2011) Genetic diversity of rhizobia associated with indigenous legumes in different regions of flanders (Belgium). Soil Biol Biochem 43(12):2384–2396. https://doi.org/10.1016/j.soilbio.2011.08.005
Edwards U, Rogall T, Blöcker H, Emde M, Böttger EC (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17(19):7843–7853
El Batanony NH, Castellano-Hinojosa A, Correa-Galeote D, Bedmar EJ (2015) The Diversity of Rhizobia Nodulating the Medicago, Melilotus and Trigonella Inoculation Group in Egypt is marked by the dominance of two genetic types. Symbiosis 67(1):3–10. https://doi.org/10.1007/s13199-015-0365-8
Elboutahiri N, Thami-Alami I, Udupa SM (2010) Phenotypic and genetic diversity in Sinorhizobium Meliloti and S. Medicae from Drought and Salt Affected Regions of Morocco. BMC Microbiol 10(1):1–13. https://doi.org/10.1186/1471-2180-10-15
El Idrissi MM, Abdelmoumen H (2021) Fenugreek biology and applications. In: Naeem M, Aftab T, Khan MMA (eds) Diversity of Trigonella foenum graecum microsymbionts in Morocco. Springer, Singapore, pp 283–299. https://doi.org/10.1007/978-981-16-1197-1_13
Ezzakkioui F, El Mourabit N, Chahboune R et al (2015) Phenotypic and genetic characterization of rhizobia isolated from hedysarum flexuosum in Northwest Region of Morocco: characterization of Rhizobia Nodulating H. Flexuosum. J Basic Microbiol 55(7):830–837. https://doi.org/10.1002/jobm.201400790
Gaur S, Tak N, Rathi S, Choudhary S, Gehlot HS (2018) Identification and molecular characterization of root nodule microsymbiont of Trigonella foenumgraecum L. Growing in different soils from Western Rajasthan, India. J Environ Biol 39(5):684–692. https://doi.org/10.22438/jeb/39/5/MRN-709
Goddard MR, Godfray HCJ, Burt A (2005) Sex increases the efficacy of natural selection in experimental yeast populations. Nature 434(7033):636–640. https://doi.org/10.1038/nature03405
Gritli T, Ellouze W, Chihaoui SA et al (2020) Genotypic and symbiotic diversity of native rhizobia nodulating red pea (Lathyrus Cicera L.) in Tunisia. Syst Appl Microbiol 43(1):126049. https://doi.org/10.1016/j.syapm.2019.126049
He YR, Wang JY, Wang ET et al (2011) Trigonella arcuata-associated rhizobia-an Ensifer (Sinorhizobium) meliloti population adapted to a desert environment. Plant Soil 345(1):89–102. https://doi.org/10.1007/s11104-011-0762-6
Hou BC, Wang ET, JrY Li et al (2009) Rhizobium Tibeticum sp. Nov., a symbiotic bacterium isolated from Trigonella Archiducis-Nicolai (Sirj.) Vassilcz. Int J Syst Evol Microbiol 59(12):3051–3057. https://doi.org/10.1099/ijs.0.009647-0
Kim OS, Cho YJ, Lee K et al (2012) Introducing EzTaxon-e: a prokaryotic 16S RRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kleczkowska J, Nutman PS, Skinner FA, Vincent JM (1968) Identification methods for microbiologists. In: Gibbs BM, Shapton DA (eds) The identification and classification of Rhizobium. Academic Press, London, pp 51–65
Kumar H, Dubey RC, Maheshwari DK (2018) Rhizobial genetic diversity in root nodules of Trigonella Foenum-Graecum cultivated in sub-Himalayan Region of Uttarakhand. Biocatal Agric Biotechnol 16:243–252. https://doi.org/10.1016/j.bcab.2018.08.011
Laguerre G, Nour SM, Macheret V et al (2001) Classification of rhizobia based on NodC and NifH gene analysis reveals a close phylogenetic relationship among phaseolus vulgaris symbionts. Microbiology 147:981–993. https://doi.org/10.1099/00221287-147-4-981
Layek J, Das A, Mitran T et al (2018) Legumes for soil health and sustainable management. In: Meena R, Das A, Yadav G, Lal R (eds) Cereal + legume intercropping: an option for improving productivity and sustaining soil health. Springer, Singapore, pp 347–386. https://doi.org/10.1007/978-981-13-0253-4_11
Lindström K, Lehtomäki S (1988) Metabolic properties, maximum growth temperature and phase sensitivity of Rhizobium sp. (Galega) compared with other fastgrowing rhizobia. FEMS Microbiol Lett 50:277–287. https://doi.org/10.1111/j.1574-6968.1988.tb02951.x
Mahdhi M, Fterich A, Rejili M, Rodriguez-Llorente ID, Mars M (2012) Legume-nodulating bacteria (LNB) from three pasture legumes (Vicia Sativa, Trigonella Maritima and Hedysarum Spinosissimum) in Tunisia. Ann Microbiol 62(1):61–68. https://doi.org/10.1007/s13213-011-0227-4
Mehrafarin A, Rezazadeh SH, Naghdi Badi H et al (2011) A review on biology, cultivation and biotechnology of fenugreek (Trigonella foenum-graecum L.) as a valuable medicinal plant and multipurpose. J Med Plants 37:6–24
Pandey P, Sahgal M, Maheswari DK, Johri BN (2004) Genetic diversity of rhizobia isolated from medicinal legumes growing in the sub-himalayan Region of Uttaranchal. Curr Sci 86(1):202–207
Rajendran G, Patel MH, Joshi SJ (2012) Isolation and characterization of nodule-associated Exiguobacterium sp. from the root nodules of fenugreek (Trigonella foenum-graecum) and their possible role in plant growth promotion. Int J Microbiol. https://doi.org/10.1155/2012/693982
Ramírez-Bahena MH, Vargas M, Martín M et al (2015) Alfalfa microsymbionts from different ITS and NodC lineages of ensifer meliloti and ensifer medicae symbiovar meliloti establish efficient symbiosis with Alfalfa in Spanish Acid Soils. Appl Microbiol Biotechnol 99(11):4855–4865. https://doi.org/10.1007/s00253-014-6347-6
Rangin C (2008) Phylogéographie, compétition et sélection chez les bactéries symbiotiques Sinorhizobium meliloti et Sinorhizobium medicae
Reeve W, Chain P, O’Hara G et al (2010) Complete genome sequence of the medicago microsymbiont Ensifer (Sinorhizobium) Medicae Strain WSM419. Stand Genom Sci 2(1):77–86. https://doi.org/10.4056/sigs.43526
Rigaud J, Puppo A (1975) Indole-3-acetic Acid Catabolism by Soybean Bacteroids. Microbiology 88(2):223–228. https://doi.org/10.1099/00221287-88-2-223
Rohlf FJ (1993) Numerical taxonomy and multivariate analysis system. NTSYS-Pc, Chennai
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Shamseldin A, Abdelkhalek A, Sadowsky MJ (2017) Recent changes to the classification of symbiotic, nitrogen-fixing, legume-associating bacteria: a review. Symbiosis 71(2):91–109. https://doi.org/10.1007/s13199-016-0462-3
Silva C, Kan FL, Martinez-Romero E (2007) Population Genetic Structure of Sinorhizobium Meliloti and S. Medicae Isolated from Nodules of Medicago spp. in Mexico: genetic structure of Sinorhizobium from Alfalfa in Mexico. FEMS Microbiol Ecol 60(3):477–489. https://doi.org/10.1111/j.1574-6941.2007.00301.x
Singh K, Gera R, Sharma R et al (2021) Mechanism and application of sesbania root-nodulating bacteria: an alternative for chemical fertilizers and sustainable development. Arch Microbiol 203(4):1259–1270. https://doi.org/10.1007/s00203-020-02137-x
Somasegaran P, Hoben HJ (1994) Handbook for rhizobia methods in legume-rhizobium technology. Springer, New York. https://doi.org/10.1007/978-1-4613-8375-8
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027. https://doi.org/10.1093/molbev/msab120
Toukabri W, Ferchichi N, Hlel D et al (2021) Response of intercropped barley and fenugreek to mono- and co-inoculation with sinorhizobium meliloti F42 and variovorax paradoxus F310 under contrasting agroclimatic regions. Arch Microbiol 203(4):1657–1670. https://doi.org/10.1007/s00203-020-02180-8
Vincent JM (1970) A manual for the practical study of the root-nodule bacteria. IBP Handbuch 15 Oxford and Edinburgh. Blackwell Scientific Publications, London
Wang SY, Yang XL, Li HF, Liu J (2006) Analysis of DNA homology and 16S rDNA sequence of rhizobia, a new phenotypic subgroup, isolated from Xizang Autonomous Region of China. (wei Sheng Wu Xue Bao) Acta Microbiol Sin 46(1):132–135
Wei GH, Wang ET, Tan ZY et al (2002) Rhizobium indigoferae sp. nov., and Sinorhizobium kummerowiae sp. nov., respectively isolated from Indigofera spp. and Kummerowia stipulacea. Int J Syst Evol Microbiol 52(6):2231–2239
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173(2):697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
Xu LM, Ge C, Cui Z, Li J, Fan H (1995) Bradyhizobium Liaoningense sp. nov., isolated from the root nodules of soybeans. Int J Syst Evol Microbiol 45(4):706–711
Zahran HH (1999) Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol Mol Biol Rev 63(4):968–989
Zandi P, Basu SK, Cetzal-Ix W et al (2017) Active ingredients from aromatic and medicinal plants. In: El-Shemy HA (ed) Fenugreek (Trigonella Foenum-Graecum L.): an important medicinal and aromatic crop. InTechOpen, Croatia, pp 207–214. https://doi.org/10.5772/66506
Acknowledgements
The authors would like to acknowledge the CNRST (Centre National pour la Recherche Scientifique et Technique) of Morocco for the sequencing of 16S rRNA, nodC, and nifH genes.
Funding
This work was supported by Projets thèmatiques-UAE, (Grant numbers 03/21). Author Barrijal Said and Belkadi Najlae has received research support from Abdelmalek Essaadi University.
Author information
Authors and Affiliations
Contributions
NB and SB wrote the main manuscript text. FE and IS participated in reviewing the manuscript. NB and RC prepared the figures. AR done the nod gene sequencing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Belkadi, N., Ezzakkioui, F., Saibari, I. et al. Genetic diversity of rhizobia isolated from nodules of Trigonella foenum-graecum L. (fenugreek) cultivated in Northwestern Morocco. Arch Microbiol 204, 574 (2022). https://doi.org/10.1007/s00203-022-03189-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03189-x