Abstract
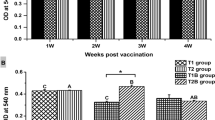
In this study, calcium phosphate nanoparticles-based (STCNV) and montanide oil adjuvant vaccine (STOAV) containing outer membrane proteins (Omps) of S. Typhi were evaluated for inducing oxidative stress indicators [reduced glutathione (GSH), lipid peroxidation (LPO), catalase, superoxide dismutase (SOD), and total protein] in the tissues of mice after vaccination. The GSH levels though slightly high in the liver, kidney, and lungs of STCNV group were not significantly different from STOAV and the control group (STC). There was no significant difference in LPO levels in any group for any tissue. The significantly lower activities of catalase were observed in the kidney and lungs of the STCNV group as compared to STOAV and STC group, while in the liver, STCNV group revealed lower catalase activity in comparison to the control group. No significant difference in the SOD activities between the two vaccinated groups was observed. The total protein contents in all the organs showed no significant difference in the vaccinated and the control group. The vaccines may induce long-term inflammatory response and consequently damage vital organs; this study revealed no long-term oxidative stress in all the three vital organs, suggesting that these vaccines may not cause oxidative damages in the vital organs of mice.





Similar content being viewed by others
References
Angelini G, Gardella S, Ardy M, Ciriolo MR, Filomeni G, Di Trapani G, Clarke F, Sitia R, Rubartelli A (2002) Antigen-presenting dendritic cells provide the reducing extracellular microenvironment required for T lymphocyte activation. Proc Natl Acad Sci 99(3):1491–1496. https://doi.org/10.1073/pnas.022630299
Bennett SJ, Griffiths HR (2013) Regulation of T-cell functions by oxidative stress. Maria Jose Alcaraz. Oreste Studies on arthritis and joint disorders. Humana Press, NY, pp 33–48
Bergmayer HU (1983) UV method of catalase assay. Methods Enzym Anal 3:273
Buchmeier NA, Heffron F (1990) Induction of Salmonella stress proteins upon infection of macrophages. Science 248(4956):730–732. https://doi.org/10.1126/science.1970672
Cheeseman KH, Slater TF (1993) An introduction to free radical biochemistry. Br Med Bull 49(3):481–493. https://doi.org/10.1093/oxfordjournals.bmb.a072625
Contreras M, Peres Rubio C, de la Fuente J, Villar M, Merino O, Mosqueda J, Cerón JJ (2020) Changes in serum biomarkers of oxidative stress in cattle vaccinated with tick recombinant antigens: a pilot study. Vaccines 9(1):5
Fraser A, Paul M, Goldberg E, Acosta CJ, Leibovici L (2007) Typhoid fever vaccines: systematic review and meta-analysis of randomised controlled trials. Vaccine 25(45):7848–7857. https://doi.org/10.1016/j.vaccine.2007.08.027
Gangathraprabhu B, Kannan S, Santhanam G, Suryadevara N, Maruthamuthu M (2020) A review on the origin of multidrug-resistant Salmonella and perspective of tailored phoP gene towards avirulence. Microb Pathog 147:104352
García-Sánchez A, Miranda-Díaz AG, Cardona-Muñoz EG (2020) The role of oxidative stress in physiopathology and pharmacological treatment with pro-and antioxidant properties in chronic diseases. Oxid Med Cell Longev 2020:1–16
Gross A, Bertholet S, Mauel J, Dornand J (2004) Impairment of Brucella growth in human macrophagic cells that produce nitric oxide. Microb Pathog 36(2):75–82. https://doi.org/10.1016/j.micpath.2003.09.003
Jackie T, Haleagrahara N, Chakravarthi S (2011) Antioxidant effects of Etlingera elatior flower extract against lead acetate-induced perturbations in free radical scavenging enzymes and lipid peroxidation in rats. BMC Res Notes 4(1):1–8. https://doi.org/10.1186/1756-0500-4-67
Jha R, Kumar A, Saxena A, Pandey M, Kumar R, Saxena MK (2015) Heterogeneous expression and functional evaluation of in silico characterized recombinant OmpC of Salmonella Typhimurium as a functional poultry vaccine to eradicate zoonotic transmission. Afri J Biotechnol 14(41):2862–2870. https://doi.org/10.5897/AJB2015.14865
Kaufmann SHE (1990) Heat shock proteins and the immune response. Immunol Today 11:129–136. https://doi.org/10.1016/0167-5699(90)90050-J
Kumar A, Verma AK, Rahal A, Panwar PK, Dhama K (2013) Recent trends in development of adjuvant of vaccine. Trends Med Res 8:32–35. https://doi.org/10.3923/tmr.2013.32.35
Kumar A, Gupta VK, Mandil R, Verma AK, Rahal A, Yadav SK (2018) Mapping of oxidative stress in immune response induced by polymer gel based Brucella melitensis vaccine in mice. Indian J Anim Sci 88(7):11–18
Kumar A, Gupta VK, Mandil R, Rahal A, Verma AK, Yadav SK (2019b) Interplay of oxidative stress and antioxidant bio markers in oil adjuvant Brucella melitensis vaccinated and challenged mice. Vaccine 37(25):3343–3351
Kumar A, Gupta VK, Rahal A, Mandil R, Verma AK, Yadav SK (2019) Nanoparticle based Brucella melitensis vaccine induced oxidative stress acts in synergism to immune response. Indian J Anim Res 53(5):648–654
Lawrence DA, Song R, Webert P (1996) Surface thiols of human lymphocytes and their changes after in vitro and in vivo activation. J Leukoc Biol 60(5):611–618. https://doi.org/10.1002/jlb.60.5.611
Lee JS, Mogasale VV, Mogasale V, Lee K (2016) Geographical distribution of typhoid risk factors in low and middle income countries. BMC Infect Dis 16(1):1–10
Lin Y, Wang X, Huang X, Zhang J, Xia N, Zhao Q (2017) Calcium phosphate nanoparticles as a new generation vaccine adjuvant. Expert Rev Vaccines 16(9):895–906. https://doi.org/10.1080/14760584.2017.1355733
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Madesh M, Balasubramanian KA (1998) Microtiter plate assay for superoxide dismutase using MTT reduction by superoxide. Indian J Biochem Biophys 35(3):184–188
Marathe SA, Lahiri A, Negi VD, Chakravortty D (2012) Typhoid fever & vaccine development: a partially answered question. Indian J Med Res 135(2):161–169
Murray PJ, Young RA (1992) Stress and immunological recognition in host-pathogen interactions. J Bacteriol 174(13):4193–4196. https://doi.org/10.1128/jb.174.13.4193-4196.1992
Rahal A, Kumar A, Singh V, Yadav B, Tiwari R, Chakraborty S, Dhama K (2014) Oxidative stress, prooxidants, and antioxidants: the interplay. BioMed Res Int. https://doi.org/10.1155/2014/761264
Ramani S, Pathak A, Dalal V, Paul A, Biswas S (2020) Oxidative stress in autoimmune diseases: an under dealt malice. Curr Protein Pept Sci 21(6):611–621
Rehman SU (1984) Lead induced regional lipid peroxidation in brain. Toxicol Lett 21:333–337. https://doi.org/10.1016/0378-4274(84)90093-6
Saad NJ, Lynch VD, Antillón M, Yang C, Crump JA, Pitzer VE (2018) Seasonal dynamics of typhoid and paratyphoid fever. Sci Rep 8(1):1–9
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal biochem 25:192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Singh Y, Saxena A, Singh SP, Verma MK, Kumar A, Kumar A, Mrigesh M, Saxena MK (2022) Calcium phosphate adjuvanted nanoparticles of outer membrane proteins of Salmonella Typhi as a candidate for vaccine development against Typhoid fever. J Med Microb 71(4):001529. https://doi.org/10.1099/jmm.0.001529
Snedecor GW, Cochran WG (1989) Statistical methods, 8thEdn. Ames: Iowa State Univ. Press Iowa 54:71–82
Sofy AR, Dawoud RA, Sofy MR, Mohamed HI, Hmed AA, El-Dougdoug NK (2020) Improving regulation of enzymatic and non-enzymatic antioxidants and stress-related gene stimulation in Cucumber mosaic cucumovirus-infected cucumber plants treated with glycine betaine, chitosan and combination. Molecules 25(10):2341
Vashishtha VM, Kalra A (2020) The need & the issues related to new-generation typhoid conjugate vaccines in India. Indian J Med Res 151(1):22–34. https://doi.org/10.4103/ijmr.IJMR_1890_17
Velisek J, Stara A, Li ZH, Silovska S, Turek J (2011) Comparison of the effects of four anaesthetics on blood biochemical profiles and oxidative stress biomarkers in rainbow trout. Aquaculture 310(3–4):369–375. https://doi.org/10.1016/j.aquaculture.2010.11.010
Yan Z, Garg SK, Banerjee R (2010) Regulatory T cells interfere with glutathione metabolism in dendritic cells and T cells. J Biol Chem 285(53):41525–41532. https://doi.org/10.1074/jbc.M110.189944
Acknowledgements
The authors are thankful to the Director, Experiment Station and the Dean, College of Veterinary and Animal Sciences, for providing the necessary facilities and support to conduct the present study. Dr. Bhaskar Sharma. Ex-National Professor, ICAR-Indian Veterinary Research Institute, Izatnagar, is highly acknowledged for his guidance and support during the study and preparation of the manuscript.
Funding
The research work was supported financially by the Department of Biotechnology, (Grant Number BT/PR796/ADV/116/2011), Ministry of Science and Technology, Government of India. The grant was awarded to Dr. Mumtesh Kumar Saxena as an R&D project.
Author information
Authors and Affiliations
Contributions
YS prepared nanovaccine formulation for vaccination of mice and drafted the manuscript. AK performed the experiments and contributed to the interpretation of data. AS guided and helped in the preparation of vaccine formulation. PB edited the manuscript. SPS guided during handling and vaccination of mice. AK and MM contributed to the aseptic collection of organs from different groups of mice. MKS designed the study, contributed to the interpretation of data and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest concerning to this manuscript.
Ethical approval
The entire animal experiments and trials were approved by Institutional Animal Ethics Committee (IAEC) of College of Veterinary & Animal Sciences, G.B. Pant University of Agriculture & Technology, Pantnagar, Uttarakhand, India.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, Y., Kumar, A., Saxena, A. et al. Assessment of free radicals and reactive oxygen species milieu in nanoparticles adjuvanted outer membrane proteins vaccine against Salmonella typhi. Arch Microbiol 204, 479 (2022). https://doi.org/10.1007/s00203-022-03096-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03096-1