Abstract
Summary
The Ultra-Orthodox Jewish population has behaviors that can influence the risk for osteoporotic fractures. We investigated whether this population is more prone to osteoporotic fractures than non-Orthodox Jewish. We did not find a significant difference in osteoporotic fracture rates between the two populations despite major differences in exposure to potential risk factors.
Introduction
The Ultra-Orthodox Jewish population is a conservative population with unique cultural behaviors such as modest clothing and specific dietary restrictions, which can influence bone density and risk for osteoporotic fractures. The aim of this study is to investigate whether the Ultra-Orthodox Jewish population is more prone to osteoporotic fractures than the non-Orthodox Jewish population.
Methods
This retrospective cohort study utilized computerized records from Maccabi Health Service. Study population included patients 65 years and older without a history of osteoporotic fracture, who reside in regions of Ultra-Orthodox and non-Orthodox Jews. The primary outcome was the adjusted risk to osteoporotic fracture during 9 years of follow-up. Cox regression included patient characteristics and risk factors for osteoporosis.
Results
A total of 115,134 patients were included in this study: 5397 patients residing in Ultra-Orthodox regions (51.0% female) and 109,737 patients residing in non-Orthodox regions (52.6% female). A total of 16,352 (14.2%) patients had an osteoporotic fracture during the study period. There was no significant difference in fracture rate between Ultra-Orthodox and non-Orthodox (14.3% vs. 14.2%, p = 0.827). Among Ultra-Orthodox and non-Orthodox females and males, there were no significant differences in fracture rates (19.1% vs. 19.1% p = 0.982 and 9.3% vs. 8.8% p = 0.311, respectively). The adjusted hazard risk for the Ultra-Orthodox Jews was 1.026, 95% CI: 0.95–1.11, p = 0.512.
Conclusion
We did not find a significant difference in the rate of osteoporotic fractures between Ultra-Orthodox and non-Orthodox populations despite major differences in exposure to potential risk factors. Results suggest that the perception of risk factors relevant for the religious communities should be re-evaluated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
All countries include populations with a variety of national, ethnic, religious, and linguistic minorities that enrich the diversity of their societies. In 2019, Jews make up to 79% of the total population in Israel. The Ultra-Orthodox Jewish population is a conservative and highly religious Jewish population that represents approximately 18% of the Jews in Israel [1]. A health issue that can be influenced by the ultra-orthodox unique cultural characteristics is bone density, and the risk for osteoporotic fractures. Some of the known risk factors for osteoporosis and for osteoporotic fractures may be significantly different between the Ultra-Orthodox and the non-Orthodox communities due to their religious values, which affect the behaviors and exposures of each population.
Literature suggests that lower vitamin D levels may be correlated with lower bone mineral density (BMD) [2, 3] and with osteoporotic fractures [4, 5]. The Ultra-Orthodox community is less exposed to sunlight than other communities. First, the Ultra-Orthodox people give a central place to learning the “Tora” (Bible) and Jewish Education. From a young age, boys receive religious education in the “Yeshiva” from morning to evening with decreased exposure to sunlight. In addition, both men and woman need to follow the rules of “Tzniut” (modesty) which require to substantially covering their bodies. Less sun exposure might influence vitamin D synthesis [6], suggesting that vitamin D levels may be lower among the Ultra-Orthodox community members [7, 8].
Another possible risk factor in this religious community is their lower calcium consumption. Literature suggests there is a correlation of low daily calcium consumption and osteoporotic fractures [9, 10] and to low BMD [11, 12]. Ultra-Orthodox Jews keep a set of dietary laws according to the Jewish laws. One of which is the separation of milk and meat products; therefore, when a person eats meat or its derivatives, he or she would need to wait several hours before eating dairy products (up to 6 h between the meat and dairy meals). The religious law lowers opportunities for the Ultra-Orthodox population to consume dairy food throughout the day. Furthermore, research indicates that Ultra-Orthodox women’s daily calcium consumption was lower than those of non-Orthodox Jewish women, although this finding was not statistically significant [13].
In addition, studies suggest that long duration of breastfeeding is correlated to lower BMD [14, 15] and higher risk of osteoporotic fractures [16]. Ultra-Orthodox Jewish women tend to breastfeed their children for longer periods of time [17] in comparison to other populations in Israel.
Hormone replacement therapy reduces the risk of osteoporotic fractures [18, 19]. A study suggested that, compared with secular persons, a considerably lower proportion of orthodox women reported using HRT [13]. In this case, HRT may be a protective factor to which the Ultra-Orthodox are not exposed.
The main objective of this study was to investigate whether the Ultra-Orthodox Jewish population is more prone to osteoporotic fractures. By comparing the Ultra-Orthodox population and different communities with similar genetic backgrounds in the same geographic area, a cohort study can shed a light on the impact of the abovementioned risk factors.
Methods
This was a retrospective cohort study, where the primary outcome was time-to-osteoporotic fracture in patients without a previous record of fracture. Comparisons were made between patients residing in Ultra-Orthodox Jews regions (the “Ultra-Orthodox Jews” group) and patients residing in non-Orthodox Jews regions (the control group or “non-Orthodox Jews” group) in Israel, with adjustment for known risk factors for fractures.
Data source
The study used de-identified data from the Maccabi Healthcare Services (MHS) central computerized database. MHS is the second largest health maintenance organization operating in Israel, serving more than 2.5 million members (25% of the national population). The MHS fully computerized database captures all information on patient interaction with the medical system (including demographics, visits, diagnoses, imaging, medication prescriptions, medication dispenses, procedures, and laboratory measurements).
Study population and design
This retrospective cohort study included male and female patients 65 years and older at the index date, 01/01/2010, with follow-up data until 01/06/2019. All patients were registered in MHS for at least 1 year before index date and had at least 5 years of continuous enrollment in MHS. As we aimed to investigate the occurrence of the first osteoporotic fracture, any patients with a previous record of osteoporotic fractures before 01/01/2010 were excluded. Areas in Israel, including areas within cities, are coded according to the probability of a person belonging to a certain community. MHS patients living in a geographic area coded as “Arab” within Israel were excluded from the study in order to compare two groups that are distinct in their exposure to risk factors. The Arab–Israeli population may share some exposures with the Ultra-Orthodox Jewish communities (i.e., covered skin for religious reasons) and other similarities with the non-Ultra-Orthodox population (not separating meat and milk products).
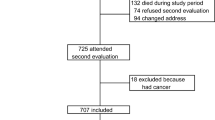
After exclusion criteria were met, the cohort was divided into two comparison groups: the Ultra-Orthodox Jews and the rest of the Israeli Jewish population—“non-Ultra-Orthodox” (Fig. 1). The division was based on geographic location. In this study, the different community sections are separated into two groups. For example, in an Ultra-Orthodox neighborhood, the probability of a person to belong to Ultra-Orthodox group would be very high, and the probability of that person belonging to a non-religious group would be low. Each MHS patient has a “section” code based on his or her permanent address. For this study, to be considered Ultra-Orthodox, the patients’ domestic address was required to be in an area where residents are identified as a “high” or “very high” likelihood of being Ultra-Orthodox. In the non-Ultra-Orthodox group, patients were included if their recorded address was located in an area where the probability of being Ultra-Orthodox was “medium likelihood,” “weak likelihood,” or “no likelihood.”
Variable definitions
Several variables were assessed for comparison between groups at baseline and for statistical analyses. Age of participants was calculated at the index date, socioeconomic status was expressed in three categories (low [1–4], medium [5, 6], and high [7–10]), and smoking was divided into “ever” and “never” categories, defining patients that were considered smokers at least once in their medical records. Diagnoses which correlated with higher risk of developing osteoporotic fractures such as diabetes, cancer, chronic kidney disease (CKD), hyperthyroidism, hypogonadism, Paget’s disease, rheumatoid arthritis, early menopause, Crohn’s disease, hypertension, and osteoporosis were defined by a diagnosis code or with a specific MHS disease registry within the database (Appendix 1).
Glucocorticoid use was defined as a purchase of any oral glucocorticoid for more than 3 months at a dose equivalent to 5 mg or higher of prednisolone per day. Body mass index (BMI) variable was defined as four categories: underweight (BMI < 18.5), normal weight (BMI = 18.5 to 25), overweight (BMI = 25 to 30), or obese (BMI > 30) at baseline. Certain variables that contain a laboratory measurement or a physician visit questionnaire included a “missing” category (i.e., smoking, BMI, and vitamin D levels). The patients’ vitamin D level was categorized as deficient if patients had vitamin D levels below 11 ng/mL (according to laboratory standard definition of vitamin D deficiency).Vitamin D levels were the baseline levels at the beginning of the study. Vitamin D levels were ordered when the physician decided it was recommended for his patient.
The outcome event in this study was defined as a major osteoporotic fracture (MOF) within the MHS Osteoporosis Registry data [20]. Therefore, patients with the specific MOF code from the registry had fracture commonly associated with osteoporosis, such as closed fractures of femur, vertebra, distal radius, and proximal humerus. The MOF code was used to limit the likelihood of fractures occurring from traumas, such as car accidents or other injuries that may not represent an osteoporotic fracture.
Statistical analyses
The primary outcome in this study was the adjusted time to osteoporotic fracture, using Cox regression for comparison between the two groups. This was measured for the full population, and then as a secondary outcome for women and men separately. Only the covariates that differed significantly at baseline were added into each model. The covariates included were presumed risk factors for osteoporotic fractures, namely the group (Ultra-Orthodox vs. the non-Ultra-Orthodox), age, socioeconomic status (SES), smoking, cancer, CKD, hyperthyroidism, BMI, and vitamin D deficiency.
To compare the baseline characteristics for significant differences between groups, a Pearson’s chi-square test was performed for all categorical variables, and Student’s t-test for continuous variables. After assessing the primary results from the Cox regressions, a sub-analysis using Kaplan–Meier (KM) estimator was done for the male population. A Mantel-Cox Log Rank test was selected in the KM analysis to compare any significant differences between groups (Ultra-Orthodox vs non Ultra-Orthodox).
Results
A total of 115,134 patients were included in this study (Fig. 1), where 5397 patients were in the Ultra-Orthodox group and 109,737 patients in the non-Ultra-Orthodox. At baseline, age was very similar between groups (mean age for Ultra-Orthodox = 74.44 (± 7.02) and for non-Ultra-Orthodox = 74.25 (± 6.77), p = 0.043), where 51.0% of the Ultra-Orthodox group and 52.6% of the control group were female (p = 0.022). Seventy-six percent of Ultra-Orthodox and 16.6% of non-Ultra-Orthodox were considered at low SES (other categories of SES in Table 1; p < 0.001). For the smoking variable, 17.9% of Ultra-Orthodox “ever” smoked compared to 22.0% in the non-Ultra-Orthodox group (p < 0.001). The groups also differed significantly in prior cancer diagnoses (Ultra-Orthodox 10.8% vs non Ultra-Orthodox 13.4%; p < 0.001), CKD (Ultra-Orthodox 68.1% vs non Ultra-Orthodox 70.4%; p < 0.001), hyperthyroidism (Ultra-Orthodox 2.1% vs non Ultra-Orthodox 2.6%; p = 0.023), early menopause (Ultra-Orthodox 0.5% vs non Ultra-Orthodox 0.2%; p = 0.001, the denominator was all women in the group), and BMI (categories in Table 1, p < 0.001). Menopause date was missing for 47% of all women (regardless of their religious group). Vitamin D deficiency was more common in the Ultra-Orthodox group at baseline (13.7% compared to 9.0%, p < 0.001). Missing data for vitamin D levels was also higher for the orthodox-Jews (16.9% compared to 14%). 30.3% of the Orthodox-Jews and 28.5% of the non-Orthodox-Jews died during the follow-up period.
There was no significant difference in fracture rate between Ultra-Orthodox Jews and non-Orthodox-Jews (14.3% vs. 14.2%, p = 0.827). Among Orthodox and non-Orthodox females and males, there were no significantly statistic difference in fracture rate (19.1% vs. 19.1% p = 0.982 and 9.3% vs. 8.8% p = 0.311, respectively).
In the first Cox regression, including both sexes (n = 115,134), 16,352 (14.2%) patients had an osteoporotic fracture during the study period. In this analysis, the “group” variable, defining the Ultra-Orthodox vs non Ultra-Orthodox, was not significant with hazard ratio (HR) of 1.026 [CI: 0.95–1.11] (p = 0.512). However, age, sex, SES, smoking, cancer, CKD, hyperthyroidism, BMI, and vitamin D deficiency (Table 2) were significant in the model.
In the Cox regression for females only (n = 60,483), there were 11,544 (19.1%) patients with a fracture outcome during the study period. The HR for fracture in ultraorthodox was 0.98 [CI: 0.89–1.07]. There were no significant difference between Ultra-Orthodox and non-Ultra-Orthodox groups (p = 0.675). Age, high SES, smoking, hyperthyroidism, CKD, and BMI were statistically significant in this model (Table 3).
In the Cox regression for male patients only (n = 54,651), 4808 (8.8%) patients had a fracture outcome. In this model, the “group” (i.e., Ultra-Orthodox vs. non Ultra-Orthodox) was closest to being statistically significant, with HR of 1.14 [CI: 0.99–1.30] and a p-value of 0.064 (Table 4).
A univariate Kaplan–Meier survival curve (Fig. 2) displays male patients’ time-to-fracture outcome, with no significant difference in outcomes between the Ultra-Orthodox men (above) and the non-Ultra-Orthodox men (below) in this figure (Log Rank test p = 0.206).
Discussion
The main finding in this study is that despite large differences in exposures that are supposed to be related to osteoporosis, no significant differences in osteoporotic fracture rates were found between Ultra-Orthodox and non-Orthodox populations. The large sample allowed for a narrow confidence interval, and the higher limit for the hazard ratio is 1.1, meaning that at most, Ultraorthodox group would have a very modest increase in the likelihood of a first osteoporotic fracture. There was a trend toward higher rates in Ultra-Orthodox men; however, it was non-significant and very subtle in absolute differences.
These findings oppose the preliminary hypothesis of this study, but have implications for the understanding of osteoporotic fractures and their risk factors. First, the results could be explained by a protective factor that was not taken into account, or that was only partially adjusted for in the analyses. For example, research findings suggest that frequent attendance at religious services is associated with significantly lower risk of all-cause mortality and that religious activities and attitudes might be associated with better physical health [21, 22]. This may explain why Ultra-Orthodox people, whom their faith in God is considered a major part in their lives, are in a better health condition. In addition, one of the religious values followed by Ultra-Orthodox Jewish population is “Peru Urevu” which can roughly be interpreted as “Be fruitful and multiply.” In this community’s families, the average number of children is 7.1 compared to approximately 3 in non-religious Jewish communities in Israel [1]. Research suggests that high birth rate may be associated with reduced risk of osteoporotic fracture [23,24,25]. This fact might be a protective factor among Ultra-Orthodox women. This can also give explanation for the small difference we found between men and women: in women, there was no difference while in men there was a trend toward more fractures in the Ultra-Orthodox group with a p-value of 0.064 and hazard ratio of 1.138.
Second, in big data studies, the information was not collected prospectively and missing data will lead to residual confounding. In our study, this is relevant especially for smoking status, BMI, age at menopause, and blood tests. Literature suggests that smoking is correlated with lower BMD [26, 27] and higher risk of osteoporotic fractures [28, 29]. Israeli Ultra-Orthodox men and women have very low prevalence of smoking when compared to the non-Orthodox population [13, 30]. Although smoking was an explanatory variable in the models, missing smoking status for some patients could potentially lead to biased results. For example, when not adjusted for, fractures that could be attributed to smoking would appear more in the non-Ultra-Orthodox group, since smoking prevalence is higher.
A third explanation suggests that factors considered important in the causation of fractures are less influential than expected: although the Ultra-Orthodox community is far less exposed to sun and therefore have lower vitamin D levels [7], we did not find a significant difference in osteoporotic fracture rate. It is possible that sun exposure, at least in sunny geographic areas like Israel, is less influential on bone health and osteoporotic fractures. This is maybe in contrast with finding in less sunny geographical areas [31].
Similarly, lower calcium intake may not be an important risk factor for osteoporotic fractures. This is supported by a meta-analysis, which concluded that dietary calcium intake is not associated with risk of fractures [32]. In 2020, the International Osteoporosis Foundation published a recommendation for sun exposure to prevent osteoporosis [33]. Considering known adverse effects of sun exposure, perhaps this recommendation should be re-evaluated.
This study is limited by its retrospective nature. The primary outcome, osteoporotic fracture, was not examined prospectively but according to a specific diagnosis in the MHS registry. In addition, we considered a patient Ultra-Orthodox and non-Orthodox according to area of their residential location, which was coded in the MHS data according to the majority population living in that area. This leads to contamination of groups, and result in reducing effects of population type on measured outcomes. On the other hand, residential areas in Israel are generally homogenous in regards to Ultra-Orthodox Jewish persons [34], so this bias toward no effect of exposure is limited. As previously mentioned, missing data may have created partial adjustment and bias. To be noted are the high rates (around 30%) of missing data for smoking, BMI, and especially age at menopause (with 47% of missing data). These variables are documented in the medical file by the physician; therefore, many times it is missing. Another possible limitation is the study retrospective nature; in our analysis, we included only patients over 65 years old, who had never had fracture before the index date. It is possible that the ultra-orthodox group develops fracture in younger age than 65 years and, therefore, we excluded them from the analysis. In addition, because we excluded patients with a fracture before the age of 65, we could not adjust for this variable, which is one of the major risk factors for another fracture.
This study also comprises several strengths such as the use of big-data from a large state-mandated healthcare provider. This real-world longitudinal data enabled the implication of exclusion criteria that ensured only persons without prior osteoporotic fracture (since 1998) were included in this study. The utilization of medical registries from MHS also permitted the description of patient characteristics, including SES and comorbidities, at baseline for comparison between groups. An additional strength is the size of the cohort itself, containing over 115,000 patients included in the research analyses.
Further research is needed on younger age population and on patients with a history of fractures.
In conclusion, big data and the osteoporosis registry, which contains information regarding incidence of osteoporotic fractures, serve as an opportunity to study two communities, which share similar genetic background, but have major different exposures. This study did not find a significant association between osteoporotic fracture rates and being an Ultra-Orthodox-Jew. This is despite major differences in exposure to factors, which are considered risk factors. These results may suggest the need to re-evaluate our perception and recommendations about sun exposure and diet as means to prevent osteoporotic fractures.
References
Cahaner L, Malach G (2019) The Yearbook of the Ultra-Orthodox Society in Israel 2019
Garg MK, Tandon N, Marwaha RK, Menon AS, Mahalle N (2014) The relationship between serum 25-hydroxy vitamin D, parathormone and bone mineral density in Indian population. Clin Endocrinol (Oxf) 80(1):41–46. https://doi.org/10.1111/cen.12248
Jiang X, Good LE, Spinka R, Schnatz PF (2016) Osteoporosis screening in postmenopausal women aged 50–64 years: BMI alone compared with current screening tools. Maturitas. https://doi.org/10.1016/j.maturitas.2015.09.009
Tai V, Leung W, Grey A, Reid IR, Bolland MJ (2015) Calcium intake and bone mineral density: systematic review and meta-analysis. BMJ. https://doi.org/10.1136/bmj.h4183
Cauley JA, Parimi N, Ensrud KE et al (2010) Serum 25-hydroxyvitamin D and the risk of hip and nonspine fractures in older men. J Bone Miner Res. https://doi.org/10.1359/jbmr.090826
Nair R, Maseeh A (2012) Vitamin D: the sunshine vitamin. J Pharmacol Pharmacother. https://doi.org/10.4103/0976-500X.95506
Tsur A, Metzger M, Dresner-Pollak R (2011) Effect of different dress style on vitamin D level in healthy young Orthodox and ultra-Orthodox students in Israel. Osteoporos Int. https://doi.org/10.1007/s00198-010-1492-y
Mukamel MN, Weisman Y, Somech R, et al (2001) Vitamin D deficiency and insufficiency in orthodox and non-orthodox Jewish mothers in Israel. Isr Med Assoc J
Warensjö E, Byberg L, Melhus H et al (2011) Dietary calcium intake and risk of fracture and osteoporosis: prospective longitudinal cohort study. BMJ. https://doi.org/10.1136/bmj.d1473
Vannucci L, Masi L, Gronchi G, Fossi C, Carossino AM, Brandi ML (2017) Calcium intake, bone mineral density, and fragility fractures: evidence from an Italian outpatient population. Arch Osteoporos. https://doi.org/10.1007/s11657-017-0333-4
Kim KM, Choi SH, Lim S et al (2014) Interactions between dietary calcium intake and bone mineral density or bone geometry in a low calcium intake population (KNHANES IV 2008–2010). J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2014-1006
Boot AM, de Ridder MAJ, Pols HAP, Krenning EP, de Muinck K-S (1997) Bone mineral density in children and adolescents: relation to puberty, calcium intake, and physical activity 1. J Clin Endocrinol Metab. https://doi.org/10.1210/jcem.82.1.3665
Werner P, Olchovsky D, Shemi G, Vered I (2003) Osteoporosis health-related behaviors in secular and orthodox Israeli Jewish women. Maturitas. https://doi.org/10.1016/S0378-5122(03)00197-X
Hwang IR, Choi YK, Lee WK et al (2016) Association between prolonged breastfeeding and bone mineral density and osteoporosis in postmenopausal women: KNHANES 2010–2011. Osteoporos Int. https://doi.org/10.1007/s00198-015-3292-x
Yeo UH, Choi CJ, Choi WS, Kim KS (2016) Relationship between breast-feeding and bone mineral density among Korean women in the. J Bone Miner Metab. https://doi.org/10.1007/s00774-015-0649-3
Yun BH, Chon SJ, Choi YS, Cho S, Lee BS, Seo SK (2016) The effect of prolonged breast-feeding on the development of postmenopausal osteoporosis in population with insufficient calcium intake and vitamin D level. Osteoporos Int. https://doi.org/10.1007/s00198-016-3585-8
Birenbaum E, Fuchs C, Reichman B (1989) Demographic factors influencing the initiation of breast-feeding in an Israeli urban population. Pediatrics
Torgerson DJ, Bell-Syer SEM (2001) Hormone replacement therapy and prevention of nonvertebral fractures: a meta-analysis of randomized trials. J Am Med Assoc. https://doi.org/10.1001/jama.285.22.2891
Nelson HD, Humphrey LL, Nygren P, Teutsch SM, Allan JD (2002) Postmenopausal hormone replacement therapy: scientific review. J Am Med Assoc. https://doi.org/10.1001/jama.288.7.872
Lan TY, Zeng YF, Tang GJ et al (2015) The use of hypnotics and mortality - a population-based retrospective cohort study. PLoS One 10(12):1–14. https://doi.org/10.1371/journal.pone.0145271
Li S, Stampfer MJ, Williams DR, Vanderweele TJ (2016) Association of religious service attendance with mortality amongWomen. JAMA Intern Med. https://doi.org/10.1001/jamainternmed.2016.1615
Koenig HG, George LK, Titus P (2004) Religion, spirituality, and health in medically ill hospitalized older patients. J Am Geriatr Soc. https://doi.org/10.1111/j.1532-5415.2004.52161.x
Kauppi M, Heliövaara M, Impivaara O, Knekt P, Jula A (2011) Parity and risk of hip fracture in postmenopausal women. In Osteoporosis International. https://doi.org/10.1007/s00198-010-1392-1
Wang Q, Huang Q, Zeng Y et al (2016) Parity and osteoporotic fracture risk in postmenopausal women: a dose-response meta-analysis of prospective studies. Osteoporos Int. https://doi.org/10.1007/s00198-015-3351-3
Hillier TA, Rizzo JH, Pedula KL et al (2003) Nulliparity and fracture risk in older women: the study of osteoporotic fractures. J Bone Miner Res. https://doi.org/10.1359/jbmr.2003.18.5.893
Siris ES, Miller PD, Barrett-Connor E et al (2001) Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: results from the National Osteoporosis Risk Assessment. J Am Med Assoc. https://doi.org/10.1001/jama.286.22.2815
Akkawi I, Zmerly H (2018) Osteoporosis: current concepts. Joints. https://doi.org/10.1055/s-0038-1660790
Kanis JA, Johnell O, Oden A, Johansson H, McCloskey E (2008) FRAX™ and the assessment of fracture probability in men and women from the UK. Osteoporos Int. https://doi.org/10.1007/s00198-007-0543-5
Kanis JA, Johnell O, Oden A et al (2005) Smoking and fracture risk: a meta-analysis. Osteoporos Int. https://doi.org/10.1007/s00198-004-1640-3
Kopel E, Keinan-Boker L, Enav T, Dichtiar R, Shohat T (2013) Cigarette smoking and correlates among Ultra-Orthodox Jewish males. Nicotine Tob Res. https://doi.org/10.1093/ntr/nts139
Nilson F, Moniruzzaman S, Andersson R (2014) A comparison of hip fracture incidence rates among elderly in Sweden by latitude and sunlight exposure. Scand J Public Health. https://doi.org/10.1177/1403494813510794
Bolland MJ, Leung W, Tai V et al (2015) Calcium intake and risk of fracture: systematic review. BMJ. https://doi.org/10.1136/bmj.h4580
Lin LP, Lai WJ, Hsu SW, Lin JD (2020) Early osteoporosis risks and associated factors among caregivers working in disability institutions: IOF one-minute osteoporosis risk check. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph17093319
Alfasi N, Flint Ashery S, Benenson I (2013) Between the individual and the community: residential patterns of the haredi population in jerusalem. Int J Urban Reg Res 37(6):2152–2176. https://doi.org/10.1111/j.1468-2427.2012.01187.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Jacobson Bensky, M., Adler, L., Banon, T. et al. Risk of major osteoporotic fractures among ultra-orthodox Jews. Osteoporos Int 34, 111–118 (2023). https://doi.org/10.1007/s00198-022-06522-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-022-06522-8