Abstract
Summary
A cocktail of many different antioxidants might be more effective than supplementation with a single molecule, and it closely resembles the natural environment in which active compounds were found. This is the first study well-grounded in stereological examination that showed that black olive extract effectively can ameliorate the quantitative changes of the bone structure and prevented bone loss in this osteoporosis animal model.
Introduction
The aim of this study was to quantitatively evaluate the effects of black olive extract consumption on treatment of ovariectomized (OVX) induced osteoporosis in rats. This is the first study well-grounded in stereological examination.
Methods
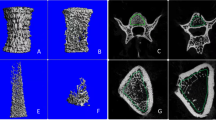
Ninety adult rats were allocated to control, sham-operated, OVX, and olive-supplemented OVX groups (received 250-, 500-, and 750-mg/kg body weight black olive hydroalcoholic extract orally) for 16 weeks. At the end of the experiment, blood samples were collected, and plasma levels of calcium, phosphorus, and alkaline phosphatase were assayed. Then, the specimens from both the tibia and fifth lumbar vertebra (L5) bones were processed, and stereological analysis was performed.
Results
Administration of extract resulted in decrease of alkaline phosphatase level during the treatment. After treatment of OVX rats with three doses of extract, the total number of the osteocytes revealed an increment in 500- and 750-mg/kg treated groups in comparison to the OVX group. This increment was significant only in L5. Compared to the OVX group, a significant increase was observed in the number of osteoblastsin L5 vertebra in three doses of extract-treated groups. However, this increment in tibia was statistically significant only in 750-mg/kg black olive hydroalcoholic extract-treated group. Moreover, the number of osteoclast cells were significantly decreased in vertebra and tibia in the treated groups compared to the OVX group (P < 0.05).
Conclusion
Black olive hydroalcoholic extract effectively can ameliorate the quantitative changes of the bone structure and prevented bone loss in this osteoporosis animal model. Thus, it can be a promising candidate for treatment of accelerated bone loss especially in postmenopausal osteoporosis.


Similar content being viewed by others
References
Donmez BO, Ozdemir S, Sarikanat M, Yaras N, Koc P, Demir N, Karayalcin B (2012) OguzN. Effect of angiotensin II type 1 receptor blocker on osteoporotic rat femurs. Pharmacol Rep 64(4):878–88
Jeddi M, Roosta MJ, Dabbaghmanesh MH, Omrani GR, Ayatollahi SM, Bagheri Z, Showraki AR, Bakhshayeshkaram M (2013) Normative data and percentile curves of bone mineral density in healthy Iranian children aged 9–18 years. Arch Osteoporos 8(1–2):114
Azizi ZA, Zamani A, Omrani LR, Omrani L, Dabaghmanesh MH, Mohammadi A, Namavar MR, Omrani GR (2010) Effects of hyperhomocysteinemia during the gestational period on ossification in rat embryo. Bone 46(5):1344–8
Hassan HA, EL Wakf AM, EL Gharib NE (2013) Role of phytoestrogenic oils in alleviating osteoporosis associated with ovariectomy in rats. Cytotechnology 65(4):609–619
Muthusami S, Ramachandran I, Muthusamy B, Vasudevan G, Prabhu V, Subramaniam V, Jagadeesan A, Narasimhan S (2005) Ovariectomy induces oxidative stress and impairs bone antioxidant system in adult rats. Clin Chim Acta 360(1–2):81–86
Sánchez-Rodríguez MA, Ruiz-Ramos M, Correa-Muñoz E, Mendoza-Núñez VM (2007) Oxidative stress as a risk factor for osteoporosis in elderly Mexicans as characterized by antioxidant enzymes. BMC Musculoskelet Disord 8:124
Zhang R, Liu ZG, Li C, Hu SJ, Liu L, Wang JP, Mei QB (2009) Du-Zhong (EucommiaulmoidesOliv.) cortex extract prevent OVX-induced osteoporosis in rats. Bone 45(3):553–9
Pines A (2010) Guidelines and recommendations on hormone therapy in the menopause. J Midlife Health 1(1):41–42
Fernández-Real JM, Bulló M, Moreno-Navarrete JM, Ricart W, Ros E, Estruch R, Salas-Salvadó JA (2012) Mediterranean diet enriched with olive oil is associated with higher serum total osteocalcin levels in elderly men at high cardiovascular risk. J Clin Endocrinol Metab 97(10):3792–8
Carrión Y, Ntinou M, Badal E (2010) Oleaeuropaea L. in the North Mediterranean Basin during the Pleniglacial and the Early-Middle Holocene. Quarter Scie Rev 29(7–8):952–968
Chen J, Hui E, Ip T, Thompson LU (2004) Dietary flaxseed enhances the inhibitory effect of tamoxifen on the growth of estrogen dependent human breast cancer (MCF-7) in nude mice. Clin Cancer Res 10(22):7703–11
Coni E, Benedetto R, Di Pasquale M, Masella R, Modesti D, Mattei R, Carlini EA (2000) Protective effect of oleuropein, an olive oil biophenol, on low density lipoprotein oxidizability in rabbits. Lipids 35(1):45–54
Hassan WN, Saed AM (2011) Protective effect of anise fruit (pimpinellaanisum) against osteoporosis in rat model. Am J Biomed Sci 3(1):49–56
Cicerale S, Lucas L, Keast R (2010) Biological activities of phenolic compounds present in virgin olive oil. Int J Mol Sci 11(2):458–479
Greenwald P, Anderson D, Nelson SA, Taylor PR (2007) Clinical trials of vitamin and mineral supplements for cancer prevention. Am J Clin Nutr 85(1):314S–317S
Waterhouse AL (2003) Determination of total phenolics. In: Wrolstad RE (ed) Current protocols in food analytical chemistry. Wiley, New York, pp I1.1.1–I1.1.8
Rainha N, Lima E, Baptista J, Rodrigues C (2011) Antioxidant properties, total phenolic, total carotenoid and chlorophyll content of anatomical parts of Hypericum foliosum. J Med Plant Res 5(10):1930–40
Karbalay-Doust S, Noorafshan A, Pourshahid SM (2012) Taxol and taurine protect the renal tissue of rats after unilateral ureteral obstruction: a stereological survey. Korean J Urol 53(5):360–367
Mattfeldt T, Mall G, Gharehbaghi H, Möller P (1990) Estimation of surface area and length with the orientator. J Microsc 159(Pt 3):301–17
Nyengaard JR (1999) Stereologic methods and their application in kidney research. J Am Soc Nephrol 10(5):1100–23
Gundersen HJ, Bendtsen TF, Korbo L, Marcussen N, Møller A, Nielsen K, Nyengaard JR, Pakkenberg B, Sørensen FB, Vesterby A (1988) Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. APMIS 96(5):379–94
Noorafshan A, Hoseini L, Karbalay-Doust S, Nadimi E (2012) A simple stereological method for estimating the number and the volume of the pancreatic beta cells. JOP 13(4):427–432
Puel C, Quintin A, Agalias A, Mathey J, Obled C, Mazur A, DaviccoM J, Lebecque P, Skaltsounis AL, Coxam V (2004) Olive oil and its main phenolic micronutrient (oleuropein) prevent inflammation-induced bone loss in the ovariectomised rat. Br J Nutr 92(1):119–27
García-Villalba R, Larrosa M, Possemiers S, Tomás-Barberán FA, Espín JC (2014) Bioavailability of phenolics from an oleuropein-rich olive (Oleaeuropaea) leaf extract and its acute effect on plasma antioxidant status: comparison between pre- and postmenopausal women. Eur J Nutr 53(4):1015–27
Swaminathan R (2001) Biochemical markers of bone turnover. Clin Chim Acta 313(1–2):95–105
Sheweita SA, Khoshhal KI (2007) Calcium metabolism and oxidative stress in bone fractures: role of antioxidants. Curr Drug Metab 8(5):519–25
Campos MS, López-Aliaga I, Barrionuevo M, Lisbona F, Coves F (1989) Nutritive utilization of calcium in rats: effects of dietary fat components and vitamin D3 on intestinal resected rats. J Nutr Sci Vitaminol (Tokyo) 35(5):511–21
Claassen N, Potgieter HC, Seppa M, Vermaak WJ, Coetzer H, Van Papendorp DH, Kruger MC (1995) Supplemented gamma-linolenic acid and eicosapentaenoic acid influence bone status in young male rats: effects on free urinary collagen crosslinks, total urinary hydroxyproline, and bone calcium content. Bone 16(4 Suppl):385S–392S
Lim DW, Kim YT (2014) Anti-osteoporoticeffects of Angelicasinensis (Oliv.) Diels extract on ovariectomized rats and its oral toxicity in rats. Nutrients 6(10):4362–72
Peel N (2009) Bone remodelling and disorders of bone metabolism. Surgery 27(2):70–74
Nishide Y, Tadaishi M, Kobori M, Tousen Y, Kato M, Inada M, Miyaura C, Ishimi Y (2013) Possible role of S-equol on bone loss via amelioration of inflammatory indices in ovariectomizedmice. J Clin Biochem Nutr 53(1):41–8
Johnson-Lynn SE, Francis R, McCaskie A (2008) Osteoporosis treatments and their effect on fracture healing. Curr Orthopaed 22:336–340
Redlich K, Smolen JS (2012) Inflammatory bone loss: pathogenesis and therapeutic intervention. Nat Rev Drug Discov 11(3):234–50
Taxvig C, Specht IO, Boberg J, Vinggaard AM, Nellemann C (2013) Dietary relevant mixtures of phytoestrogens inhibit adipocyte differentiation in vitro. Food Chem Toxicol 55:265–71
Reinwald S, Weaver CM (2006) Soy isoflavones and bone health: a double-edged sword? J Nat Prod 69(3):450–9
López-Grueso R, Gambini J, Abdelaziz KM, Monleón D, Díaz A, El Alami M, Bonet-Costa V, Borrás C, Viña J (2014) Early, but not late onset estrogen replacement therapy prevents oxidative stress and metabolic alterations caused by ovariectomy. Antioxid Redox Signal 20(2):236–46
Santiago-Mora R, Casado-Díaz A, De Castro MD, Quesada-Gómez JM (2011) Oleuropein enhances osteoblastogenesis and inhibits adipogenesis: the effect on differentiation in stem cells derived from bone marrow. Osteoporos Int 22(2):675–84
Acknowledgments
This study was financially supported by the Research Vice-chancellor of Shiraz University of Medical Sciences (Grant No. 89-01-33-2707). The authors would like to thank Ms. A. Keivanshekouh at the Research Improvement Center of Shiraz University of Medical Sciences for improving the use of English in the manuscript.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noorafshan, A., Dabbaghmanesh, M.H., Tanideh, N. et al. Stereological study of the effect of black olive hydroalcoholic extract on osteoporosis in vertebra and tibia in ovariectomized rats. Osteoporos Int 26, 2299–2307 (2015). https://doi.org/10.1007/s00198-015-3126-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-015-3126-x