Abstract
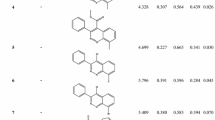
Chlorinated agrochemicals play a major role in toxicity due especially to the labile C − Cl bond and high lipophilicity of organochlorines. In turn, urea and thiourea herbicides are widely used for weed control. A series of substituted N-benzoyl-N’-pyrimidin-2-yl thioureas has been recently synthesized and tested against Brassica napus L., demonstrating promising herbicidal activities, particularly for chlorinated derivatives. We have therefore modeled these activities using multivariate image analysis applied to quantitative structure–activity relationships (MIA-QSAR) to find out a significant and reliable correlation between measured and predicted inhibition of B. napus L. root growth (%) and, ultimately, to propose effective, non-chlorinated and/or less lipophilic N-(4-methanesulfonyl)benzoyl-N’-(pyrimidin-2-yl)thiourea candidates. The model was found to be predictive, giving an average r2pred in the external validation of 0.833. The predicted data for the proposed herbicides, interpreted in terms of MIA-plots of the chemical moieties responsible for bioactivity and supported by docking studies towards the photosystem II enzyme, suggest that substituents at both R1 and R2 positions modulate the agrochemical (R1 = Cl increases and R2 = OR decreases bioactivity) and environmental friendship (particularly with R2 = OH) performances of this class of compounds.







Similar content being viewed by others
References
Ago H, Adachi H, Umena Y, Tashiro T, Kawakami K, Kamiya N, Tian L, Han G, Kuang T, Liu Z, Wang F, Zou H, Enami I, Miyano M, Shen J-R (2016) Novel features of eukaryotic photosystem II revealed by its crystal structure analysis from a red alga. J Biol Chem 291:5676–5687. https://doi.org/10.1074/jbc.M115.711689
Arnot JA, Gobas FAPC (2003) A generic QSAR for assessing the bioaccumulation potential of organic chemicals in aquatic food webs. QSAR Comb Sci 22:337–345. https://doi.org/10.1002/qsar.200390023
Barber C, Amberg A, Custer L, Dobo KL, Glowienke S, Gompel JV, Gutsell S, Harvey J, Honma M, Kenyon MO, Kruhlak N, Muster W, Stavitskaya L, Tealsdale A, Vessey J, Wichard J (2015) Establishing best practise in the application of expert review of mutagenicity under ICH M7. Reg Toxicol Pharmacol 73:367–377. https://doi.org/10.1016/j.yrtph.2015.07.018
Barigye SJ, Duarte MH, Nunes CA, Freitas MP (2016) MIA-plot: a graphical tool for viewing descriptor contributions in MIA-QSAR. RSC Adv 6:49604–49612. https://doi.org/10.1039/C6RA09593C
Brown HM, Cotterman JC (1994) Recent advances in sulfonylurea herbicides. In: Stetter J (ed) Herbicides inhibiting branched-chain amino acid biosynthesis. Chemistry of plant protection, vol 10. Springer, Berlin Heidelberg
Consonni V, Ballabio D, Todeschini R (2009) Comments on de definition of the Q2 parameter for QSAR validation. J Chem Inf Model 49:1669–1678. https://doi.org/10.1021/ci900115y
Dennington RD II, Keith TA, Millam JM (2008) GaussView. Gaussian Inc, Wallingford
Fiser A (2010) Template-based protein structure modeling. In: Fenyö D (ed) Computational Biology. Methods in Molecular Biology (Methods and Protocols). Humana Press, Totowa, pp 73–94
Gupta PK (2014) Herbicides and fungicides. In: Gupta RC (ed) Biomarkers in toxicology. Academic Press, Cambridge, pp 409–431
Hooft RWW, Vriend G, Sander C, Abola EE (1996) Errors in protein structures. Nature 381:272. https://doi.org/10.1038/381272a0
Huang X, Miller W (1991) A time-efficient, linear-space local similarity algorithm. Adv Appl Math 12:337–357. https://doi.org/10.1016/0196-8858(91)90017-D
Karasali H, Maragou N (2016) Pesticides and herbicides: Types of pesticide. In: Caballero B, Finglas PM, Toldrá F (eds) Encyclopedia of food and health. Elsevier, Amsterdam, pp 319–325
Kiralj R, Ferreira MMC (2009) Basic validation procedures for regression models in QSAR and QSPR studies: Theory and application. J Braz Chem Soc 20:770–787. https://doi.org/10.1590/S0103-50532009000400021
Li J-H, Wang Y, Wu Y-P, Li R-H, Liang S, Zhang J, Zhu Y-G, Xie B-J (2021) Synthesis, herbicidal activity study and molecular docking of novel pyrimidine thiourea. Pest Biochem Physiol 172:104766. https://doi.org/10.1016/j.pestbp.2020.104766
Liang X, Huang T, Li M, Mack J, Wildervanck M, Nyokong T, Zhu W (2017) Highly efficient C−Cl bond cleavage and unprecedented C−C bond cleavage of environmentally toxic DDT through molecular electrochemical catalysis. Appl Cat A 545:44–53. https://doi.org/10.1016/j.apcata.2017.07.026
Liu J (2010) Phenylurea herbicides. In: Krieger R (ed) Hayes’ Handbook of Pesticide Toxicology. Academic Press, Cambridge, pp 1725–1731
Metz J, Pakrasi H, Seibert M, Arntzer C (1986) Evidence for a dual function of the herbicide-binding D1 protein in photosystem II. FEBS Lett 205:269–274. https://doi.org/10.1016/0014-5793(86)80911-5
Mitra I, Saha A, Roy K (2010) Exploring quantitative structure–activity relationship studies of antioxidant phenolic compounds obtained from traditional Chinese medicinal plants. Mol Simul 36:1067–1079. https://doi.org/10.1080/08927022.2010.503326
Moses SK, Harley JR, Lieske CL, Muir DCG, Whiting AV, O’Hara TM (2015) Variation in bioaccumulation of persistent organic pollutants based on octanol–air partitioning: Influence of respiratory elimination in marine species. Mar Poll Bull 100:122–127. https://doi.org/10.1016/j.marpolbul.2015.09.020
Nunes CA, Freitas MP, Pinheiro ACM, Bastos SC (2012) Chemoface: A novel free user-friendly interface for chemometrics. J Braz Chem Soc 23:2003–2010. https://doi.org/10.1590/S0103-50532012005000073
Orry AJW, Abagyan R (2012) Homology modeling: methods and protocols. Humana Press, Totowa. https://doi.org/10.1007/978-1-61779-588-6
Ramalho TC, França TCC, Rennó MN, Guimarães AP, da Cunha EFF, Kuca K (2010) Development of new acetylcholinesterase reactivators: molecular modeling versus in vitro data. Chem Biol Interact 185:73–77. https://doi.org/10.1016/j.cbi.2010.05.016
Roy K, Chakraborty P, Mitra I, Ojha PK, Kar S, Das RN (2013) Some case studies on application of ‘“rm2”’ metrics for judging quality of quantitative structure–activity relationship predictions: Emphasis on scaling of response data. J Comput Chem 34:1071–1082. https://doi.org/10.1002/jcc.23231
Roy K, Kar S, Ambure P (2015) On a simple approach for determining applicability domain of QSAR models. Chemom Intell Lab Sys 145:22–29. https://doi.org/10.1016/j.chemolab.2015.04.013
The UniProt Consortium (2021) UniProt: the universal protein knowledgebase in 2021. Nucleic Acids Res 49:D480–D489. https://doi.org/10.1093/nar/gkaa1100
Thomann RV (1989) Bioaccumulation model of organic chemical distribution in aquatic food chains. Environ Sci Technol 23:699–707. https://doi.org/10.1021/es00064a008
Thomsen R, Christensen MH (2006) MolDock: A new technique for high-accuracy molecular docking. J Med Chem 49:3315–3321. https://doi.org/10.1021/jm051197e
Waterhouse A, Bertoni M, Bienert S, Studer G, Tauriello G, Gumienny R, Heer FT, de Beer TAP, Rempfer C, Bordoli L, Lepore R, Schwede T (2018) SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res 46:W296–W303. https://doi.org/10.1093/nar/gky427
Acknowledgements
The authors are thankful to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, funding code 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Grant number 301371/2017-2), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for financial support of this research.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Natânia E. Rodrigues, Adriana C. de Faria, Ingrid V. Pereira, Elaine F. F. da Cunha and Matheus P. Freitas. The first draft of the manuscript was written by Matheus P. Freitas and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rodrigues, N.E., de Faria, A.C., Pereira, I.V. et al. QSAR-Guided Proposition of N-(4-methanesulfonyl)Benzoyl-N’-(Pyrimidin-2-yl)Thioureas as Effective and Safer Herbicides. Bull Environ Contam Toxicol 108, 1019–1025 (2022). https://doi.org/10.1007/s00128-022-03467-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-022-03467-w