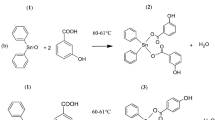
Abstract
A series of novel benzisoselenazolone derivatives bearing 1,3,4-thiadiazole moiety were designed, synthesized and evaluated for their in vitro antiproliferative activities against human cancer cell lines SMMC-7721, MCF-7, A549, and normal cell lines L929 by CCK-8 assay. The bioassay results demonstrated that most of the tested compounds showed potent antitumor activities, and some compounds exhibited better effects than positive control ebselen and ethaselen against various cancer cell lines. Among these compounds, compound 7l showed significant antiproliferative activity against SMMC-7721 cells, with IC50 values of 2.18 μM. Compound 7j exhibited the best inhibitory effect against MCF-7 cells, with IC50 values of 2.89 μM. Compounds 7e and 7i displayed highly effective antitumor activities against A549 cells, with IC50 values of 2.33 and 2.33 μM, respectively. Furthermore, most of the tested compounds showed weak cytotoxic effect against the normal cell lines L929. The pharmacological results suggest that the substituents of benzoylmethylthio-moiety at position-2 on 1,3,4-thiadiazole are vital for modulating antiproliferative activities against various tumor cell lines.





Similar content being viewed by others
References
Amir M, Kumar H, Javed SA (2007) Non-carboxylic analogues of naproxen: design, synthesis, and pharmacological evaluation of some 1,3,4-oxadiazole/thiadiazole and 1,2,4-triazole derivatives. Arch Pharm Chem Life Sci 340:577–585
Clerici F, Pocar D (2001) Synthesis of 2-amino-5-sulfanyl-1,3,4-thiadiazole derivatives and evaluation of their antidepressant and anxiolytic activity. J Med Chem 44:931–936
Cressier D, Prouillac C, Hernandez P, Amourette C, Diserbo M, Lion C, Rima G (2009) Synthesis, antioxidant properties and radioprotective effects of new benzothiazoles and thiadiazoles. Bioorg Med Chem 17:5275–5284
Dekhane DV, Pawar SS, Gupta S, Shingare MS, Patil CR, Thore SN (2011) Synthesis and anti-inflammatory activity of some new 4,5-dihydro-1,5-diaryl-1H-pyrazole-3- substituted-heteroazole derivatives. Bioorg Med Chem Lett 21:6527–6532
Dubey V, Pathak M, Bhat HR, Singh UP (2012) Design, facile synthesis, and antibacterial activity of hybrid 1,3,4-thiadiazole-1,3,5-triazine derivatives tethered via –S– bridge. Chem Biol Drug Des 80:598–604
El-Bayoumy K (1997) Organoselenium compounds: a novel class of cancer chemopreventive agents. Drugs Future 22:539–545
Foroumadi A, Firoozpour L, Emami S, Mansouri S, Ebrahimabadi AH, Asadipour A, Amini M, Saeid-Adeli N, Shafiee A (2007) Synthesis and antibacterial activity of N-[5-(chlorobenzylthio)-1,3,4-thiadiazol-2-yl] piperazinyl quinolone derivatives. Arch Pharm Res 30:138–145
Grozav AI, Gaina L, Kuete V, Silaghi-Dumitrescu L, Efferth T, Zaharia V (2013) Microwave-assisted synthesis of new selenazole derivatives with antiproliferative activity. Molecules 18:4679–4688
Hamama WS, Gouda MA, Badr MH, Zoorob HH (2013) Synthesis, antioxidant, and antitumor evaluation of certain new N-substituted-2-amino-1,3,4-thiadiazoles. Med Chem Res 22:3556–3565
He J, Li DD, Xiong K, Ge YJ, Jin HW, Zhang GZ, Hong MS, Tian YL, Yu J, Zeng HH (2012) Inhibition of thioredoxin reductase by a novel series of bis-1,2-benzisoselenazol-3(2H)-ones: organoselenium compounds for cancer therapy. Bioorg Med Chem 20:3816–3827
Hosseinzadeh L, Khorand A, Aliabadi A (2013) Discovery of 2-phenyl-N-(5-(trifluoro-methyl)-1,3,4-thiadiazol-2-yl)acetamide derivatives as apoptosis inducers via the caspase pathway with potential anticancer activity. Arch Pharm Chem Life Sci 346:812–818
Khan I, Ali S, Hameed S, Rama NH, Hussain MT, Wadood A, Uddin R, Ul-Had Z, Khan A, Ali S, Choudhary MZ (2010) Synthesis, antioxidant activities and urease inhibition of some new 1,2,4-triazole and 1,3,4-thiadiazole derivatives. Eur J Med Chem 45:5200–5207
Kumar D, Kumar NM, Chang KH, Shah K (2010) Synthesis and anticancer activity of 5-(3-indolyl)-1,3,4-thiadiazoles. Eur J Med Chem 45:4664–4668
Kumar D, Vaddula BR, Chang KH, Shah K (2011) One-pot synthesis and anticancer studies of 2-arylamino-5-aryl-1,3,4-thiadiazoles. Bioorg Med Chem Lett 21:2320–2323
Lee J, Lee HS, Seo HJ, Son EJ, Lee SH, Jung ME, Lee MW, Han HK, Kim J, Kang J, Lee J (2010) Novel C-aryl glucoside SGLT2 inhibitors as potential antidiabetic agents: 1,3,4-thiadiazolylmethylphenyl glucoside congeners. Bioorg Med Chem 18:2178–2194
Liu F, Luo XQ, Song BA, Bhadury PS, Yang S, Jin LH, Xue W, Hu DY (2008) Synthesis and antifungal activity of novel sulfoxide derivatives containing trimethoxyphenyl substituted 1,3,4-thiadiazole and 1,3,4-oxadiazole moiety. Bioorg Med Chem 16:3632–3640
Mantovani G, Maccio A, Madeddu C, Serpe R, Massa E, Gramignano G, Lusso MR, Curreli N, Rinaldi A (2004) Selenium is effective in inducing lymphocyte progression through cell cycle in cancer patients: potential mechanisms for its activity. J Exp Ther Oncol 4:69–78
Muller A, Cadenas E, Graf P, Sies H (1984) A novel biologically active seleno-organic compound-I: glutathione peroxidase-like activity in vitro and antioxidant capacity of PZ 51 (ebselen). Biochem Pharmacol 33:3235–3239
Parnham MJ, Graf E (1987) Seleno-organic compounds and therapy of hydroperoxide-linked pathological conditions. Biochem Pharmacol 36:3095–3102
Parnham MJ, Leyck S, Graf E, Dowling EJ, Blake DR (1991) The pharmacology of ebselen. Agents Actions 32:4–9
Pattn SR, Kittur BS, Sastry BS, Jadav SG, Thakur DK, Madamwar SA, Shinde HV (2011) Synthesis evaluation of some novel 1,3,4-thiadiazoles for antidiabetic activity. Indian J Chem 50B:615–618
Reich HJ, Jasperse CP (1987) Orgnoselenium chemistry. Redox chemistry of selenocysteine model systems. J Am Chem Soc 109:5549–5551
Saluk J, Bijak M, Nowak P, Wachowicz B (2013) Evaluating the antioxidative activity of diselenide containing compounds inhuman blood. Bioorg Chem 50:26–33
Sharma B, Verma A, Sharma UK, Prahapati S (2014) Efficient synthesis, anticonvulsant and muscle relaxant activities of new 2-((5-amino-1,3,4-thiadiazol-2-yl)methyl)-6-phenyl-4,5-dihydropyridazin-3(2H)-one derivatives. Med Chem Res 23:146–157
Shi YP, Chen BQ, Ma J, Liu YM, Li CW (2011) Synthesis and antitumor activity of 2-(2-substituted 1,3,4-thiadiazol-5-yl)benzisoselenazol-3(2H)-one derivatives. Acta Chim Sin 69:2561–2566
Siddiqui N, Ahuja P, Malik S, Arya SK (2013) Design of benzothiazole-1,3,4-thiadiazole conjugates: synthesis and anticonvulsant evaluation. Arch Pharm Chem Life Sci 346:819–831
Sies H (1993) Ebselen, a selenoorganic compound as glutathione peroxidase mimic. Free Radic Biol Med 14:313–323
Tan Q, Li J, Yin HW, Wang LH, Tang WC, Zhao F, Liu XM, Zeng HH (2010) Augmented antitumor effects of combination therapy of cisplatin with ethaselen as a novel thioredoxin reductase inhibitor on human A549 cell in vivo. Invest New Drugs 28:205–215
Terazawa R, Garud DR, Hamada N, Fujita Y, Itoh T, Nozawa Y, Nakane K, Deguchi T, Koketsu M, Ito M (2010) Identification of organoselenium compounds that possess chemopreventive properties in human prostate cancer LNCaPcells. Bioorg Med Chem 18:7001–7008
Wendel A, Tiegs G (1986) A novel biologically active seleno-organic compound-VI: protection by ebselen (PZ 51) against galactosamine/endotoxin-induced hepatitis in mice. Biochem Pharmacol 35:2115–2118
Wood SM, Beckhaml C, Yosioka A, Darban H, Watson RR (1999) β-Carotene and selenium supplementation enhances immune response in aged humans. Integr Med 2:85–92
Xing FX, Li SL, Ge XY, Wang CY, Zeng HH, Li D, Dong L (2008) The inhibitory effect of a novel organoselenium compound BBSKE on the tongue cancer Tca8113 in vitro and in vivo. Oral Oncol 44:963–969
Xu WM, Li SZ, He M, Yang S, Li XY, Li P (2013) Synthesis and bioactivities of novel thioether/sulfone derivatives containing 1,2,3-thiadiazole and 1,3,4-oxadiazole/thiadiazole moiety. Bioorg Med Chem Lett 23:5821–5824
Yusuf M, Khan RA, Ahmed B (2008) Syntheses and anti-depressant activity of 5-amino-1,3,4-thiadiazole-2-thiolimines and thiobenzyl derivatives. Bioorg Med Chem 16:8029–8034
Zaharia V, Ignat A, Ngameni B, Kueke V, Moungang ML, Fokunang CN, Vasilescu M, Palibroda N, Cristea C, Silaghi-Dumitrescu L, Ngadjui BT (2013) Heterocycles 23: synthesis, characterization and anticancer activity of new hydrazinoselenazole derivatives. Med Chem Res 22:5670–5679
Zhao F, Yan J, Deng SJ, Lan LX, He F, Kuang B, Zeng HH (2006) A thioredoxin reductase inhibitor induces growth inhibition and apoptosis in five cultured human carcinoma cell lines. Cancer Lett 236:46–53
Zhao HC, Shi YP, Liu YM, Li CW, Xuan LN, Wang P, Zhang K, Chen BQ (2013) Synthesis and antitumor-evaluation of 1,3-selenazole-containing 1,3,4-thiadiazole derivatives. Bioorg Med Chem Lett 23:6577–6579
Zhou ZB, Xia XP, Xu HB, Zhao CX, Chen CY (1993) Synthesis and anti-tumor activity of ebselen and its derivatives. Chem J Chin Univ 14:220–222
Acknowledgments
We are grateful to the National Natural Science Foundation of China (20971097) and Tianjin Municipal Natural Science Foundation (13JCYBJC24500) for financial support.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xuan, Ln., Wang, P., Zhang, K. et al. Synthesis and in vitro antiproliferative activity of novel benzisoselenazolone derivatives. Med Chem Res 24, 543–552 (2015). https://doi.org/10.1007/s00044-014-1149-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1149-z