Abstract
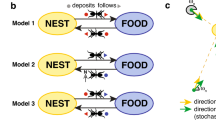
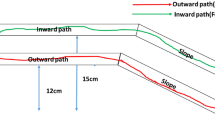
Social insects utilise a complex spatial orientation system mediated by chemical signals. This study investigated how the foraging behaviour of ants (Dorymyrmex thoracicus) varies depending on the quantity of an available resource using a field experiment. Further, we demonstrated computationally that ant displacement is compatible with a model based on pheromone deposition. Our experiment tested how the resource size (large or small) and availability (one or two simultaneous patches) of resources offered influence the number of recruited ants (traffic flow) and the speed of traffic flow both moving towards a resource and returning to the colony. The results showed that the returning flow was higher than the going flow independent of resource. The traffic flow towards a single resource was higher than the flow for either of two simultaneous resources patches offered; thus, multiple resources sources split the flow, regardless of the fact that resource size did not affect foraging choices. Our results indicated that the ants used an orientation mechanism that can be reproduced by a theoretical computer model. With our model, we showed that initially, the displacement of ants followed no clearly detectable pattern. However, with increasing levels of ant recruitment and consequent pheromone deposition on the most used trails, returning displacement revealed the formation of shorter and more organised trails. The model revealed key transition between periods of order and disorder that continued until the flow of information reached an organised state (Shannon entropy). This study highlights an exceptional mechanism of foraging optimisation in eusocial insects.





Similar content being viewed by others
References
AntWeb (2018) Publishing AntWeb. https://www.antweb.org/description.do?genus=dorymyrmex&species=thoracicus&rank=species&countryName=Paraguay. Accessed 25 Jun 2018
Bartumeus F, Da Luz MGE, Viswanathan GM, Catalan J (2005) Animal search strategies: a quantitative random-walk analysis. Ecology 86:3078–3087. https://doi.org/10.1890/04-1806
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Beckers R, Deneubourg JL, Goss S (1992) Trails and U-turns in the selection of a path by the ant Lasius niger. J Theor Biol 159:395–415
Chowdhury D, Guttal V, Nishinari K, Schadschneider A (2002) A cellular-automata model of flow in ant trails: non-monotonic variation of speed with density. J Phys A 35:L573. https://doi.org/10.1088/0305-4470/35/41/103
Contreras-Torres H, Canals M (2010) Effect of colony, patch distance, and trajectory sense on movement complexity in foraging ants. J Insect Behav 23:319–328. https://doi.org/10.1007/s10905-010-9216-x
Crawley MJ (2012) The R book, 2nd edn. John Wiley and Sons, USA
Deneubourg JL, Aron S, Goss S, Pasteels JM, Duerinck G (1986) Random behavior, amplification processes and number of participants: how they contribute to the foraging properties of ants. Physica D 22:176–186. https://doi.org/10.1016/0167-2789(86)90239-3
Deneubourg JL, Goss S, Franks N, Pasteels J (1989) The blind leading the blind: modeling chemically mediated army ant raid patterns. J Insect Behav 2:719–725. https://doi.org/10.1007/BF01065789
Denny JA, Wright JH, Grief B (2001) Foraging efficiency in the wood ant, Formica rufa: is time of the essence in trail following? Anim Behav 62:139–146. https://doi.org/10.1006/anbe.2001.1718
Dussutour A, Fourcassie V, Helbing D, Deneubourg JL (2004) Optimal traffic organization in ants under crowded conditions. Nature 428:70–73
Flanagan TP, Letendre K, Burnside WR, Fricke GM, Moses ME (2012) Quantifying the effect of colony size and food distribution on harvester ant foraging. PLoS One 7:e39427. https://doi.org/10.1371/journal.pone.0039427
Gordon DM (1991) Behavioral flexibility and the foraging ecology of seed-eating ants. Am Nat 138:379–411. https://doi.org/10.1086/285223
Gordon DM (1995) The development of an ant colony’s foraging range. Anim Behav 49:649–659
Gordon DM (1999) Ants at work: how an insect society is organized. Free Press, New York
Gordon DM (2010) Ant encounters: interaction, networks and colony behavior. Princeton University Press, New Jersey
Goss S, Aron S, Deneubourg JL, Pasteels JM (1989) Self-organized shortcuts in the Argentine ant. Naturwissenschaften 76:579–581
Hölldobler B, Wilson EO (1990) The ants. Harvard University Press, Cambridge
Jesus RM, Rolim SG (2005) Fitossociologia da floresta atlântica de tabuleiro em Linhares (ES). Boletim Técnico SIF 19:1–149
Merkle D, Middendorf M (2000) An ant algorithm with a new pheromone evaluation rule for total tardiness problems. In: Cagnoni S (ed) Real-world applications of evolutionary computing. EvoWorkshops, Springer, Berlin, Heidelberg, pp 287–296
Miramontes O (1995) Order- disorder transitions in the behavior of ant societies. Complexity 1:56–60. https://doi.org/10.1002/cplx.6130010313
Miramontes O, Solé R, Goodwin BC (1993) Antichaos in ants: the excitability metaphor at two hierarchical levels. In: Deneubourg JL, Goss S, Nicolis G, Bersini H, Dagonnier R, Taylor T (eds) Proceedings of the second European Conference on Artificial Life (ECAL '93). Centre for non-linear phenomena and complex systems, Brussels, pp 790–807
Oliveira GV, Corrêa MM, Góes IM, Machado AF, de Sá-Neto RJ, Delabie JH (2015) Interactions between Cecropia (Urticaceae) and ants (Hymenoptera: Formicidae) along a longitudinal east-west transect in the Brazilian Northeast. Ann Soc Entomol Fr (NS) 51:153–160
Pacala SW, Gordon D, Godfray HCJ (1996) Effects of social group size on information transfer and task allocation. Evol Ecol 10:127–165. https://doi.org/10.1007/BF01241782
Perna A, Granovskiy B, Garnier S, Nicolis SC, Labe´dan M, Theraulaz G, Fourcassie V, Sumpter DJT (2012) Individual rules for trail pattern formation in Argentine ants (Linepithema humile). PLOS Comp Bio 8:e1002592. https://doi.org/10.1371/journal.pcbi.1002592
Ramos-Fernández G, Mateos JL, Miramontes O, Cocho G, Larralde H, Ayala-Orozco B (2004) Lévy walk patterns in the foraging movements of spider monkeys (Ateles geoffroyi). Behav Ecol Sociobiol 55:223–230. https://doi.org/10.1007/s00265-003-0700-6
R-Core-Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Rico-Gray V, Oliveira PS (2007) The ecology and evolution of ant-plant interactions. University of Chicago Press, Chicago
Rosario-Martinez H (2015) Phia: Post-hoc interaction analysis. R package version 0.2-1. https://CRAN.R-project.org/package=phia. Accessed 26 April 2018
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois, Urbana
Solé RV, Miramontes O, Goodwin BC (1993) Oscillations and chaos in ant societies. J Theor Biol 161:343–357. https://doi.org/10.1006/jtbi.1993.1060
Sumpter DJ, Beekman M (2003) From nonlinearity to optimality: pheromone trail foraging by ants. Anim Behav 66:273–280. https://doi.org/10.1006/anbe.2003.2224
Taylor F (1977) Foraging behavior of ants: experiments with two species of Myrmecine ants. Behav Ecol Sociobiol 2:147–167
Torres-Contreras H, Vasquez RA (2007) Spatial heterogeneity and nestmate encounters affect locomotion and foraging success in the ant Dorymyrmex goetschi. Ethology 113:76–86
Traniello JFA (1989) Foraging strategies of ants. Ann Rev Entomol 34:191–210. https://doi.org/10.1146/annurev.en.34.010189.001203
Whitford WG, Steinberger Y (2009) Harvester ants (Hymenoptera: Formicidae) discriminate among artificial seeds with different protein contents. Sociobiology 53:549–558
Wilson EO (1962) Chemical communication among workers of the fire ant Solenopsis saevissima (Fr. Smith) 1. The organization of mass-foraging. Anim Behav 10:134–147
Wolfram S (1994) Cellular automata and complexity. Westview Press, Champaign
Acknowledgements
The authors thank the Postgraduate Programs in Ecologia de Biomas Tropicais of Universidade Federal de Ouro Preto and in Biodiversidade e Uso dos Recusos Naturais of Universidade Estadual de Montes Claros for organising the field course in 2012 and provide logistical support. We gratefully acknowledge the staff of the Reserva Natural Vale for allowing us to work in the reserve. We thank the researcher Fabiana Cuezzo for the identification of the species Dorymyrmex thoracicus, and researcher Reisla Oliveira for photographing the field activities. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. GML thanks for a research scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq (155895/2014-1) and CAPES (88881.133074/2016-01). FK thanks for a research scholarship from CNPq (140647/2018-0). RF is granted by the CNPq. PL has a student fellowship (CVU 771366 from the Consejo Nacional de Ciencia y Tecnología (CONACYT), Mexico. SPR is granted by the CNPq (304024/2015-5). EA is granted by the CAPES (2657-12-0) and CNPq (425333/2018-3).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lourenço, G.M., Keesen, F., Fagundes, R. et al. Recruitment and entropy decrease during trail formation by foraging ants. Insect. Soc. 67, 59–69 (2020). https://doi.org/10.1007/s00040-019-00728-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-019-00728-6