Abstract
In Mediterranean climate regions, intermittent rivers (IRs) harbor highly dynamic communities with species and trait composition changing over time and space. Simultaneously considering multiple biodiversity facets and a spatiotemporal perspective is, therefore, key to developing effective conservation strategies for these ecosystems. We studied the spatiotemporal dynamics of aquatic macroinvertebrates in rivers of the western Mediterranean Basin by analysing (1) the taxonomic and functional richness and the local contribution to beta diversity (LCBD; measured considering taxonomic and functional facets) of perennial rivers and IRs over five sampling times, and (2) their relation with flow intermittence, local environmental uniqueness, and the number of anthropogenic impacts. Both analyses were also conducted for the subset of data including only IRs to compare values between their flowing and disconnected pool phases. According to our results, taxonomic and functional richness tended to be higher in perennial rivers than in IRs, while taxonomic and functional LCBD tended to be higher in IRs than in perennial rivers. When comparing IR sites over time, higher values of taxonomic and functional LCBD corresponded mostly to their disconnected pool phase. Flow intermittence, the number of anthropogenic impacts and the environmental uniqueness were significant predictors of taxonomic and functional richness, but only flow intermittence was an important predictor of taxonomic LCBD. For the IR-only data subset, disconnected pool permanence was the main predictor explaining spatiotemporal patterns. Our results highlight the importance of IRs to biodiversity conservation of Mediterranean climate rivers, especially during the disconnected pool phase, suggesting that these ecosystems cannot be ignored in conservation planning strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Incorporating spatiotemporal alpha (α), beta (β) and gamma (γ) diversity information into community composition analysis is crucial to developing effective biodiversity and ecosystem conservation strategies (Pereira et al. 2013; Hill et al. 2016). Commonly, conservation efforts have focused on protecting the number of species within a site (taxonomic α-diversity) or region (taxonomic γ-diversity or regional diversity), while efforts to characterize and incorporate the variation in species composition (taxonomic β-diversity) are relatively more recent (Koleff et al. 2003; Anderson et al. 2011; Socolar et al. 2016; Bush et al. 2016). Taxonomic β-diversity can be measured as the compositional dissimilarity in species assemblages, either across space (Baselga 2010; Anderson et al. 2011) or time (Legendre and Gauthier 2014; Legendre 2019; Shimadzu et al. 2015). Spatial taxonomic β-diversity aims to understand the processes governing spatial variation in community composition, while temporal taxonomic β-diversity provides information on how species composition changes over time (Anderson et al. 2011; Legendre and Gauthier 2014; Legendre and Condit 2019). The combination of spatial and temporal β-diversity has therefore the potential to better represent how and why community composition changes over time, particularly in response to environmental changes (Legendre and Condit 2019; Vander Vorste et al. 2021; Faustino de Queiroz et al. 2022). As a result, this integrated approach can help to better identify areas that may be more resilient to change and those that may be more vulnerable, allowing for targeted conservation efforts (McGill et al. 2015).
Functional diversity can provide complementary insights into the impacts of disturbance on ecosystem functioning (Flynn et al. 2011; Gross et al. 2017). For instance, the use of functional diversity allows us to clarify the role of each species in ecosystem processes and their resistance and/or resilience capacity to environmental changes, either natural or anthropogenic (Tobias and Monika 2011; Villéger et al. 2013, 2017; Soria et al. 2020). Functional β-diversity can be defined as the dissimilarity in trait composition across space (Villéger et al. 2013; Aspin et al. 2019) or time (Baselga et al. 2015; Magurran et al. 2019). As the increase in multiple anthropogenic impacts is threatening the stability of ecosystems, understanding how both taxonomic and functional diversity influence ecosystem functioning can contribute to better predicting the ecological consequences of biodiversity loss (Hooper et al. 2005; Flynn et al. 2011; Gutiérrez-Cánovas et al. 2015).
Intermittent rivers (IRs) are non-perennial watercourses that typically shift among flowing, disconnected pool and dry phases, and are therefore highly dynamic ecosystems (Gallart et al. 2012, 2017). Flow intermittence exerts a primary control on IR ecosystem structure and function over time and, consequently, biodiversity patterns typically follow these changes (Datry et al. 2014; Arroita et al. 2018; Arias-Real et al. 2020). In the case of aquatic macroinvertebrates, surface flow cessation and the subsequent formation of disconnected pools imply the disappearance of riffle-dwelling species and the appearance of pool-dwelling species (Bonada et al. 2006; Bogan et al. 2017; Tonkin et al. 2017). With the complete drying of the riverbed, some taxa may emerge, move to other wet habitats or to the hyporheic zone, or enter a desiccation-resistant life stage (Lytle and Poff 2004; Bogan et al. 2017; Stubbington et al. 2019). Shifts from dry to flowing phases following rewetting favour recolonization, contributing to the recovery of local communities in IRs (Leigh et al. 2016; Bogan et al. 2017). IR communities are highly variable in time, with species and trait composition changing from one period to another (Datry et al. 2014; Bogan et al. 2017). Nevertheless, climate change and increasing human water demand are altering biodiversity patterns and functional processes of IRs (Datry et al. 2014; Leigh et al. 2019), and thus a better understanding of their contribution to biodiversity is timely.
In comparison to IRs from other climatic regions, those in Mediterranean climate areas are characterized by being highly predictable in terms of seasonality, resulting in well-known community shifts between flowing and non-flowing phases (Hershkovitz and Gasith 2013; Tonkin et al. 2017). In addition, Mediterranean-climate IRs are global biodiversity hotspots, possessing high levels of endemism, but also being particularly vulnerable to anthropogenic impacts (Bonada and Resh 2013; Cid et al. 2017; Newbold et al. 2020). Considering their high spatiotemporal variability, identifying key sites and periods of time that contribute the most to regional diversity could be informative for conservation management (Ruhí et al. 2017; Sánchez-Montoya et al. 2020; Rodríguez-Lozano et al. 2023).
Here, we studied the spatial and temporal dynamics of aquatic macroinvertebrates in 20 rivers of the western Mediterranean Basin over different aquatic phases by analysing the taxonomic and functional richness (hereafter TRic and FRic) and the local contribution to overall taxonomic and functional β-diversity (hereafter T-LCBD and
F-LCBD, respectively; Legendre and De Cáceres 2013) of each site over five sampling times. Specifically, we analysed (1) the values of TRic, FRic, T-LCBD and F-LCBD of perennial rivers and IRs over five sampling times, and (2) their relationship with three main predictors: flow intermittence, number of anthropogenic impacts and local environmental uniqueness. Both analyses were also conducted for the subset of data including only IRs to compare values between their flowing and disconnected pool phases. Perennial rivers and IRs were sampled along gradients of natural flow intermittence and anthropogenic impacts to investigate their influence on LCBD. We used the LCBD because it allows identifying which sites and times contribute the most to total β-diversity. We hypothesized that TRic and FRic would be higher in perennial rivers than in IRs, whereas T-LCBD and F-LCBD would be higher in IRs than in perennial rivers. Macroinvertebrate communities in IRs from Mediterranean climatic regions are exposed to and adapted to higher spatiotemporal variability, and consequently host unique species adapted to non-flowing conditions (Bonada et al. 2006, 2020; Bonada and Resh 2013; Leigh et al. 2019). However, evolutionarily acquired macroinvertebrate resistance traits in response to drying (e.g. aerial respiration, diapause or dormancy resistance forms) are less frequent than resilience traits (e.g. life cycle duration, aerial dispersion) in these ecosystems (Datry et al. 2014; Leigh et al. 2016; Bogan et al. 2017), which may result in higher dispersal of individuals during the dry period (i.e. increase in β-diversity or LCBD) than individuals that remain (i.e. TRic and FRic). Moreover, we hypothesized that T-LCBD and F-LCBD values would be even higher during IRs’ periods of disconnected pools than during their flowing phase because species adapted to non-flowing conditions with some kind of resilience trait will move from dry river reaches in search of any site with water (i.e. disconnected pool). Finally, in addition to TRic, FRic, T-LCBD and F-LCBD being related to flow intermittence, we also expected them to be positively correlated with environmental uniqueness (Castro et al. 2019; Heino et al. 2022), as sites and sampling times with differentiated environmental conditions may have unique species or trait composition (Ruhí et al. 2017; Bonada et al. 2020).
Methods
Study sites and sampling design
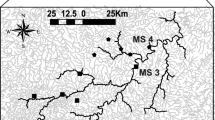
This study was conducted in 20 rivers located in the Mediterranean climate region of the Iberian Peninsula (Fig. 1). Sites were located in three predominantly calcareous catchments, comprising the Ebro, Júcar and Catalan basins. A 100-m site was defined in each river. Ten sites were intermittent and ten were perennial. The study area is characterized by a Mediterranean climate, with high seasonal and inter-annual variability in precipitation and flow regime (Bonada and Resh 2013; Cid et al. 2017). Sites ranged in altitude from 6 to 1100 m.a.s.l., and experienced different hydrological phases over the five sampling times (Table S1), with discharges ranging from 0 to 0.417 m3/s. See Table S2 and the following section on the Mediterranean Reference Criteria (MRC index)for further details on the study sites, such as riparian vegetation (e.g. cover and composition, lateral connectivity), introduced species, pollution (e.g. urban or industrial effluents), diffuse pollution sources and land use (e.g. agriculture, intensive grazing, urban use, burned vegetation), river morphology and habitat conditions (e.g. substrate diversity, canalization, gravel extraction), and hydrological alteration and regulation.
Biological dataset
Macroinvertebrates were collected during flowing and disconnected pool phases by sampling each site five times during a 6-week interval between April and December 2015: April–May (henceforth t1; spring in the Northern Hemisphere), June (t2; spring), July–August (t3; summer), September (t4; summer) and December (t5; autumn). Therefore, sampling included flowing (t1), drying (t2–t4) and rewetting (t5) periods with their corresponding flowing, disconnected pools and dry riverbed aquatic phases (see Table S1 for further details). Because five sites were dry on one to three occasions, a total of 91 samples were obtained. Our sampling procedure followed the official standardized protocol used by water agencies in Spain (MAGRAMA 2013). Samples were collected using a 250-μm-mesh D-net across all available microhabitats (i.e. hard substrates, plant detritus, vegetated banks, submerged macrophytes, sand and other fine sediments) and preserved in 4% formaldehyde (see Appendix S1 for more details). Macroinvertebrates were identified to the lowest taxonomic resolution possible, usually genus, but with some Chironomidae and Ceratopogonidae identified to subfamily or tribe. Overall, 194 macroinvertebrate taxa were identified (Soria et al. 2020).
As suggested in Soria et al. (2020), categories from seven specific resistance and resilience traits from Tachet et al. (2010) related to flow intermittence and anthropogenic impacts were used to study the functional facet of biodiversity: asexual reproduction, resistance forms (i.e. diapause or dormancy, cocoons), aerial respiration (i.e. spiracle, hydrostatic vesicle), flier and burrower (i.e. epibenthic) or interstitial (i.e. endobenthic) locomotion and substrate relation, less than a year life cycle duration, more than one reproduction cycles per year, aerial active and aquatic passive (i.e. drift) dissemination (Table S3).
Predictors of spatiotemporal β-diversity
Hydrology, anthropogenic impacts and general water quality parameters of the studied rivers were included as predictors of LCBD. The TREHS (Temporary Rivers Ecological and Hydrological Status) software (http://www.lifet rivers.eu/products/trehs-software/; Gallart et al. 2017) was used to classify rivers’ hydrological regime (i.e. perennial and IRs) and to differentiate river sites affected by natural flow intermittence from those with human-driven flow intermittence. To infer the IRs’ phases (i.e. flow, disconnected pools, dry riverbed), two temperature data loggers (UA-002 HOBO) were installed at each river site, which recorded data during the 30-week study period (see details in Soria et al. 2020). Thermal amplitude was then used to calculate the total zero-flow days (i.e. disconnected pools or dry riverbed) during the 30-week study period (hereafter ZFT) and the number of days in the disconnected pool phase since the last sample was taken (hereafter DPi) (Soria et al. 2020). The ZFT hydrological predictor was 0 for perennial rivers and ranged from 17 to 209 days of total zero-flow days for IRs. The DPi predictor ranged from 5 to 90 days in the disconnected pool phase since the last sample was taken (Table S1) and was only available for IRs. Thus, the ZFT provides information on the total cumulative drying period of each site, and thus informed on the degree of intermittency of an IR. The DPi provides information on the permanence of the disconnected pool phase between sampling times. For example, one IR may have been disconnected, connected and then disconnected again between t1 and t2, while another IR may have been disconnected for the entire time between t1 and t2. Since there was a moderate positive correlation between ZFT and DPi, the ZFT was used to test the spatiotemporal patterns of taxonomic and functional richness and LCBD between perennial rivers and IRs, while the DPi was used when analysing IRs’ spatiotemporal dataset.
Anthropogenic impacts were measured at each river site by using the number of impacts according to the Mediterranean Reference Criteria (MRC index) (Sánchez-Montoya et al. 2009). The MRC index includes information on invasive species, diffuse pollution sources, land-use intensity, riparian vegetation, river geomorphology, instream habitat conditions and hydrological alterations (Table S2), and ranges from 0 (highly impacted) to 20 (non-impacted). The information for each site was obtained from the list of pressures and impacts of the three different river basin water authorities included in our study combined with field observations (e.g. detection of invasive species) and, in the case of hydrological alterations, from the TREHS software. To facilitate interpretation, the inverse of the MRC index values was used (i.e. from 0 = non-impacted to 20 = highly impacted, hereafter number of impacts; see Soria et al. 2020). As a result, the number of impacts ranged from 0 (non-impacted) to 13.
For each sampling site and time, the following water quality parameters were measured in situ and analysed in the laboratory when required: conductivity (μS/cm), pH, temperature (ºC), dissolved oxygen (mg/l), chlorophyll-a (mg/m3), HCO3, Ca, TOC, Mg and SO4 (µg/l). Chlorophyll-a concentration, however, was not used because there was a moderate negative correlation with the number of impacts (Soria et al. 2020). Following Castro et al. (2019), the method described by Legendre and De Cáceres (2013) was used to calculate the local contribution to environmental heterogeneity (hereafter environmental uniqueness), a measure describing the environmental uniqueness of each site, which in this study was in terms of physicochemical characteristics. To do so, all physicochemical variables (except pH) were log-transformed and then a standardized Euclidean distance matrix was used to calculate the environmental uniqueness of each site. Samples with high values (closer to one) of the resultant vector are more unique in terms of environmental conditions (Castro et al. 2019). The predictor of environmental uniqueness ranged from 0.006 to 0.025, with a mean value of 0.011.
Statistical analysis
The TRic and FRic of each site and sampling time were calculated (Table S1). Data were rarefied prior to diversity calculations. FRic was obtained from Soria et al. (2020) and estimated as suggested by Villéger et al. (2008). T-LCBD and F-LCBD were estimated for each sample, i.e. for each site and sampling time (spatiotemporal approach; Legendre and De Cáceres 2013; Legendre and Gauthier 2014). T-LCBD and F-LCBD values indicate the degree of ecological uniqueness of each site at each sampling time in terms of taxa abundance or trait composition, respectively (Legendre and De Cáceres 2013). The T-LCBD was calculated using the taxa abundance matrix (Legendre and De Cáceres 2013). The F-LCBD was calculated using a matrix with the relative abundance of each trait category (columns) across the samples (rows) (Rodrigues-Capítulo et al. 2009). Following the procedures described by Legendre and De Cáceres (2013), T-LCBD and F-LCBD were estimated using the Euclidean distance on Hellinger-transformed data (Legendre and Gallagher 2001) and the significance of each LCBD value (i.e. each site in each sampling time) was tested with 999 permutations. For IRs, TRic ranged from 8 to 56 (mean: 24.63), FRic ranged from 0.002 to 0.418 (mean: 0.145), T-LCBD ranged from 0.006 to 0.029 (mean: 0.012) and F-LCBD ranged from 0.002 to 0.117 (mean: 0.013). For perennial rivers, TRic ranged from 18 to 61 (mean: 37.88), FRic ranged from 0.056 to 0.432 (mean: 0.2427), T-LCBD ranged from 0.005 to 0.016 (mean: 0.009) and F-LCBD ranged from 0.002 to 0.021 (mean: 0.008). A non-parametric Mann–Whitney U test with a Bonferroni correction was used to test significant differences between perennial rivers and IRs over the sampling times.
Linear mixed-effect models (LME) were used to test the relationship between TRic, FRic, T-LCBD and F-LCBD, and our set of environmental predictors. Specifically, the ZFT predictor was used when considering the whole dataset to compare the response variables along an intermittency gradient (i.e. including perennial rivers with ZFT = 0), while DPi was used when considering data from IRs only to compare the response variables between IRs’ periods of disconnected pools and their flowing phase. The number of impacts and environmental uniqueness predictors were used in both datasets to test whether TRic, FRic, T-LCBD and F-LCBD are negatively correlated with the number of impacts and positively correlated with environmental uniqueness. All models included sampling time (t1–t5) as a random factor to assess whether the relationship between TRic, FRic, T-LCBD and F-LCBD and predictors changed among sampling campaigns (i.e. spatial effects were assessed directly while time effects were assessed indirectly).
All analyses were conducted in R software version 3.6.2 (R Core Team 2015), using the packages “ade4” (Dray et al. 2007), “adespatial” (Dray et al. 2018), “car” (Fox and Weisberg 2011), “maptools” (Bivand and Lewin-Koh 2020), “nlme” (Pinheiro et al. 2016), “raster” (Hijmans 2020), “rgdal” (Bivand et al. 2020), “rgeos” (Bivand and Rundel 2020) and “vegan” (Oksanen et al. 2013). The code and functions used to run these analyses are available in the data availability statement.
Results
Our results suggest that both TRic and FRic tended to be higher in perennial rivers than in IRs over time (Fig. 1a, b and Table S1), while T-LCBD and F-LCBD showed the opposite pattern (Fig. 1c, d and Table S4), especially in summer. Significant differences (P < 0.05) between perennial rivers and IRs were found in t2–t5 sampling times for TRic (Fig. 1a) and t3 sampling time for T-LCBD and F-LCBD (Fig. 1c, d).
Boxplots showing taxonomic and functional local richness (a, b, respectively) and LCBD (c and d, respectively) over the five sampled times: flowing (t1: spring), drying (t2: spring and t3–t4: summer) and rewetting (t5: autumn) periods with their corresponding flowing, disconnected pools and dry riverbed aquatic phases. The thick horizontal bar indicates the median. The box indicates the interquartile range. Vertical bars indicate minimum and maximum values. Full boxes represent perennial rivers (PRs) and empty ones IRs. Significant differences (P < 0.05) between perennial rivers and IRs are indicated with (*)
When comparing the contributions of perennial rivers and IRs with overall taxonomic and functional β-diversity through the five sampling times, IR sites were the largest contributors. Specifically, significant site contributions to T-LCBD were found in five IRs corresponding to their disconnected pool phase (t3–t5; Fig. 2a and Tables S1 and S4). These five IRs had unique taxa that were found only at these sites, such as Hydrellia, Lispe, Diamesa, Anisops, Batracobdella and Hippeutis, or taxa that were found in these IRs and one (i.e. Eristalis, Sepedon, Naucoris, Stictotarsus, Cyphon, Aeschna, Parasigara) or two other rivers (i.e. Telmatoscopus, Hydroglyphus, Chaoborus, Helobdella, Sialis, Peltodytes). For F-LCBD, significant site contributions were observed at one specific sampling time (t2) of a perennial river, three IRs during its flowing phase (t1 and t3) and four IRs during its disconnected pool phase (t2, t4–t5; Fig. 2b and Tables S1 and S4).
Taxonomic (a) and functional (b) LCBD plots of each site through the five sampled times: flowing (t1: spring), drying (t2: spring and t3–t4: summer) and rewetting (t5: autumn) periods with their corresponding flowing, disconnected pools and dry riverbed aquatic phases. Circle size is proportional to the contribution to overall β-diversity for each period. Filled circles: observed perennial rivers; empty circles: observed intermittent rivers. Sites with significant values of LCBD (P < 0.05) are indicated in pink. See Table S2 for further details
When considering perennial rivers and IRs, TRic and FRic showed a significant negative correlation with all three predictors: ZFT, the number of impacts and environmental uniqueness (Table 1 and Fig. 3a–f). Our results also showed a significant and positive relationship between T-LCBD and ZFt, but no significant results were observed for the number of impacts and environmental uniqueness (Table 1 and Fig. 3g–i). None of the explanatory variables were correlated with F-LCBD (Table 1 and Fig. 3j–l).
Partial relationship between taxonomic and functional richness (TRic and FRic, respectively), taxonomic and functional LCBD (T-LCBD and F-LCBD, respectively), and the total zero-flow days (ZFT) during the 30-week study period, environmental uniqueness, and the number of impacts for all dataset including perennial and intermittent rivers. Regression lines are only shown in those plots with statistically significant relationships in the models
For the subset of data including only IRs, TRic showed a significant negative correlation with all three predictors, but FRic was only significantly and negatively correlated with the number of impacts (Table 2 and Fig. 4a–f). T-LCBD and F-LCBD were significantly correlated with DPi, but no significant patterns were observed for the other predictors (Table 2 and Fig. 4g–l).
Partial relationship between taxonomic and functional richness (TRic and FRic, respectively), taxonomic and functional LCBD (T-LCBD and F-LCBD, respectively), and the number of days in the disconnected pool phase (DPi) since the last sample was taken, environmental uniqueness and the number of impacts for only intermittent rivers. Regression lines are only shown in those plots with statistically significant relationships in the models
Discussion
Overall, our results showed that IRs had significantly higher taxonomic LCBD than perennial rivers, despite their lower local taxonomic and functional diversity. In early summer and in response to the loss of surface flow, the observed decline in taxonomic and functional richness in IRs can be explained by the disappearance of lotic species inhabiting riffle habitats (Bogan et al. 2017; Tonkin et al. 2017). Yet, during the disconnected pool phase in summer, the increase in taxonomic and functional LCBD in IRs can be related to the colonization of species with specific traits adapted to cope with such conditions (Bonada et al. 2006, 2020). A similar pattern, where sites with higher LCBD are those with lower richness, has been found in other studies of macroinvertebrates (e.g. Heino et al. 2017; da Silva et al. 2018; Valente-Neto et al. 2020) and other biological groups (e.g. Legendre and De Cáceres 2013; Vilmi et al. 2017; Landeiro et al. 2018). This negative relationship indicates that sites with low taxonomic and functional richness tend to hold unique species and trait compositions, highlighting the complementarity of alpha and beta diversity indices in describing biodiversity (Heino et al. 2017; da Silva et al. 2018). Incorporating temporal patterns of LCBD is therefore key to capturing the full variation in community composition that exists in these highly dynamic ecosystems (Ruhí et al. 2017; Sánchez-Montoya et al. 2020; Rodríguez-Lozano et al. 2023).
Our study also showed that higher values of taxonomic and functional LCBD occur mostly during the disconnected pool phase of IRs. During this phase, taxa inhabiting flowing conditions tend to be lost, while lentic taxa progressively colonize the remaining disconnected pools from nearby sites that are drying up, such as Odonata, Coleoptera and Hemiptera (Bogan et al. 2017; Bonada et al. 2020). Indeed, there are studies showing that lentic-dwelling species have higher dispersal abilities than lotic species (e.g. Ribera and Vogler 2000; Hjalmarsson et al. 2015). Disconnected pools can act as refuges for some aquatic taxa such as fish or amphibians during IRs’ dry season, which are fundamental for recolonizing the river network upon flow resumption (Hermoso et al. 2013; Gallart et al. 2017). In addition, for some species of macroinvertebrates (and for amphibians and fish), disconnected pools are also used as stepping-stones for their dispersal or as key sites for laying eggs and, thus, completing their life cycle (Bonada et al. 2006, 2020; Stubbington et al. 2017; Moidu et al. 2023). Considering that up to 60% of the world’s rivers by length are IRs and that they are expected to increase worldwide (Döll and Schmied 2012; Messager et al. 2021), it is expected that disconnected pools will become more abundant and, consequently, management actions will be needed to conserve them (Gallart et al. 2017; Bonada et al. 2020).
On the other hand, our results suggest that flow intermittence (i.e. indicated by ZFT), the number of impacts and the environmental uniqueness were significant predictors of taxonomic and functional richness, but only flow intermittence was an important predictor of taxonomic LCBD. Indeed, the permanence of disconnected pools (i.e. indicated by DPi) was the main predictor explaining the spatiotemporal patterns of IRs. However, there was no significant correlation with the number of impacts, despite some impacted IRs also showing higher LCBD values. This could be explained by the fact that resistance and resilience traits that can cope with flow intermittence in IRs (e.g. multi-voltinism, aerial respiration or mechanisms to tolerate low dissolved oxygen concentrations) may also be useful in coping with anthropogenic impacts (Bonada and Resh 2013; Stubbington et al. 2017; Soria et al. 2020), a phenomenon known as co-tolerance (Boulton et al. 2000). In fact, Mediterranean IRs hold unique species composition adapted to natural flow intermittence, such as a dominance of pool-dwelling species during the disconnected pool phase (Bonada et al. 2006, 2020; Cid et al. 2017), which could give them the ability to resist and to recover from drying periods and, at the same time, from anthropogenic impacts. However, persistent and intensifying anthropogenic impacts over time could reduce the ability of these species to cope with flow intermittence (Datry et al. 2017).
Considering that IRs have been commonly ignored in conservation planning (Bogan and Lytle 2007; Leigh et al. 2019), it is timely to provide complementary measures to adequately assess their biodiversity. In this sense, the LCBD approach shows a high potential to be used for conservation purposes, as the ecological uniqueness of a site can be compared with other sites sampled in a region (Legendre and De Cáceres 2013; da Silva et al. 2018; Valente-Neto et al. 2020). In highly dynamic systems such as IRs, not only spatial but temporal patterns should also be considered to better identify key sites (Ruhí et al. 2017; Rogosch and Olden 2019; Stubbington et al. 2019; Sánchez-Montoya et al. 2020). However, analysing only spatiotemporal species richness and community composition might not be sufficient to design conservation plans aimed at protecting the processes that maintain their ecosystem functioning (Leigh et al. 2016, 2019; Villéger et al. 2017; Crabot et al. 2020). Therefore, given the hydrological variability of IRs and the increasing anthropogenic impacts they receive, freshwater conservation planning should consider monitoring the temporal variability of both taxonomic and functional biodiversity in these ecosystems. This might be even more relevant in Mediterranean climate regions worldwide where IRs constitute one of their predominant freshwater ecosystems (Bonada and Resh 2013; Cid et al. 2017). Special attention should also be given to the disconnected pool phase, as this is key to maintaining local and regional aquatic biodiversity (Gallart et al. 2017; Bonada et al. 2020). In this regard, several management-related tools have recently been developed to better predict the flow patterns of IRs, such as wet-dry mapping, remote sensing (e.g. satellite images, fixed cameras), field sensors (e.g. conductivity, temperature, water level and/or presence/absence of water), hydrological metrics (e.g. zero-flow days) or models (e.g. IHACRES [Identification of unit Hydrographs And Component flows from Rainfall, Evaporation and Streamflow data] or SWAT [Soil & Water Assessment Tool]) (Datry et al. 2017). Indeed, as the distribution of disconnected pools in IR networks can vary spatially and temporally from one year to another, tools have been developed even to account for their temporal and spatial occurrence (e.g. TRESH software; Gallart et al. 2017), as well as to assess their priority as biodiversity refuges and incorporate them into conservation planning (Hermoso et al. 2013; Yu et al. 2022). Citizen science can also result in a powerful tool to fully understand the hydrological characteristics of these ecosystems (e.g. RiuNet app, DRYRivERS app). The integration of these tools in the conservation management of IRs, together with the use of community metrics able to capture their spatiotemporal biodiversity patterns, are key to improving freshwater conservation in the Mediterranean climate region.
Data availability
Data available via the Dryad Digital Repository https://doi.org/10.5061/dryad.n2z34tms5 (Soria et al. 2019).
References
Anderson MJ, Crist TO, Chase JM et al (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14:19–28. https://doi.org/10.1111/j.1461-0248.2010.01552.x
Arias-Real R, Muñoz I, Gutiérrez-Cánovas C, Granados V, Lopez-Laseras P, Menéndez M (2020) Subsurface zones in intermittent streams are hotspots of microbial decomposition during the non-flow period. Sci Total Environ 703:135485. https://doi.org/10.1016/j.scitotenv.2019.135485
Arroita M, Flores L, Larrañaga A, Chauvet E, Elosegi A (2018) Hydrological contingency: drying history affects aquatic microbial decomposition. Aquat Sci 80:31
Aspin TWH, Khamis K, Matthews TJ et al (2019) Extreme drought pushes stream invertebrate communities over functional thresholds. Glob Change Biol 25:230–244. https://doi.org/10.1111/gcb.14495
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A, Bonthoux S, Balent G (2015) Temporal beta diversity of bird assemblages in agricultural landscapes: land cover change vs stochastic processes. PLoS ONE 10:e0127913. https://doi.org/10.1371/journal.pone.0127913
Bivand R, Lewin-Koh N (2020). maptools: Tools for Handling Spatial Objects. R package version 1.0–1. Accessed in July 2021. https://CRAN.R-project.org/package=maptools
Bivand R, Rundel C (2020). rgeos: Interface to Geometry Engine - Open Source ('GEOS'). R package version 0.5–3. Accessed in July 2021. https://CRAN.R-project.org/package=rgeos
Bivand R, Keitt T, Rowlingson B (2020) rgdal: Bindings for the 'Geospatial' Data Abstraction Library. R package version 15–10. Accessed in July 2021. https://CRAN.R-project.org/package=rgdal
Bogan MT, Lytle DA (2007) Seasonal flow variation allows ‘time-sharing’ by disparate aquatic insect communities in montane desert streams. Freshw Biol 52:290–304. https://doi.org/10.1111/j.1365-2427.2006.01691.x
Bogan MT, Datry T, Chester ET et al (2017) Resistance, resilience, and community recovery in intermittent rivers and ephemeral streams. In: Datry T, Bonada N, Boulton AJ (eds) Intermittent rivers and ephemeral streams: ecology and management. Elsevier, Waltham, pp 349–376
Bonada N, Resh VH (2013) Mediterranean-climate streams and rivers: Geographically separated but ecologically comparable freshwater systems. Hydrobiologia 719(1):1–29. https://doi.org/10.1007/s10750-013-1634-2
Bonada N, Rieradevall M, Prat N, Resh VH (2006) Benthic macroinvertebrate assemblages and macrohabitat connectivity in Mediterranean-climate streams of northern California. J N Am Benthol Soc 25:32–43
Bonada N, Cañedo-Argüelles M, Gallart F et al (2020) Conservation and management of isolated pools in temporary rivers. Water 12(10):2870. https://doi.org/10.3390/w12102870
Boulton AJ, Sheldon F, Thoms MC, Stanley EH (2000) Problems and constraints in managing rivers with variable flow regimes. In: Boon PJ, Davies BR, Petts GE (eds) Global perspectives on river conservation: science, policy and practice. Wiley, London, pp 415–425
Bush A, Harwood T, Hoskins AJ, Mokany K, Ferrier S (2016) Current uses of beta-diversity in biodiversity conservation: a response to. Trends Ecol Evol 31:337
Castro E, Siqueira T, Melo AS, Bini LM, Landeiro VL, Schneck F (2019) Compositional uniqueness of diatoms and insects in subtropical streams is weakly correlated with riffle position and environmental uniqueness. Hydrobiologia 842(1):219–232
Cid N, Bonada N, Carlson SM, Grantham TE, Gasith A, Resh VH (2017) High variability is a defining component of Mediterranean climate rivers and their biota. Water 9:1–24. https://doi.org/10.3390/w9010052
Crabot J, Heino J, Launay B, Datry T (2020) Drying determines the temporal dynamics of stream invertebrate structural and functional beta diversity. Ecography 43:620–635. https://doi.org/10.1111/ecog.04835
da Silva PG, Hernández MIM, Heino J (2018) Disentangling the correlates of species and site contributions to beta diversity in dung beetle assemblages. Divers Distrib 24:1674–1686. https://doi.org/10.1111/ddi.12785
Datry T, Larned ST, Fritz KM, Bogan MT, Wood PJ, Meyer EI, Santos AN (2014) Broad-scale patterns of invertebrate richness and community composition in temporary rivers: effects of flow intermittence. Ecography 37:94–104. https://doi.org/10.1111/j.1600-0587.2013.00287.x
Datry T, Bonada N, Boulton AJ (2017) Intermittent rivers and ephemeral streams: ecology and management. Elsevier, Waltham
Döll P, Schmied HM (2012) How is the impact of climate change on river flow regimes related to the impact on mean annual runoff? A global-scale analysis. Environ Res Lett 7(1):14037–14111. https://doi.org/10.1088/1748-9326/7/1/014037
Dray S, Dufour AB, Chessel D (2007) The ade4 package-II: Two-table and K-table methods. R News 7(2):47–52
Dray S, Blanchet G, Borcard D, Guenard G, Jombart T, Larocque G, et al (2018) Package ‘adespatial’. R Package, 3–8
Faustino de Queiroz AC, Anton-Pardo M, Bini LM et al (2022) Invertebrate beta diversity in permanent and temporary lentic water bodies: a meta-analytic assessment. Hydrobiologia 849:1273–1285. https://doi.org/10.1007/s10750-021-04788-3
Flynn DFB, Mirotchnick N, Jain M, Palmer MI, Naeem S (2011) Functional and phylogenetic diversity as predictors of biodiversity–ecosystem-function relationships. Ecology 92:1573–1581. https://doi.org/10.1890/10-1245.1
Fox J, Weisberg S (2011) An {R} Companion to Applied Regression, Second Edition. Thousand Oaks CA: Sage. Available: http://socserv.socsci.mcmaster.ca/jfox/Books/Companion. Accessed 15 February 2020
Gallart F, Prat N, Garcia-Roger EM et al (2012) A novel approach to analysing the regimes of temporary streams in relation to their controls on the composition and structure of aquatic biota. Hydrol Earth Syst Sci 16:3165–3182. https://doi.org/10.5194/hess-16-3165-2012
Gallart F, Cid N, Latron J et al (2017) TREHS: An open-access software tool for investigating and evaluating temporary river regimes as a first step for their ecological status assessment. Sci Total Environ 607:519–540. https://doi.org/10.1016/j.scitotenv.2017.06.209
Gross N, Bagousse-Pinguet Y, Liancourt P et al (2017) (2017) Functional trait diversity maximizes ecosystem multifunctionality. Nat Ecol Evol 1:0132. https://doi.org/10.1038/s41559-017-0132
Gutiérrez-Cánovas C, Sánchez-Fernández D, Velasco J, Millán A, Bonada N (2015) Similarity in the difference: Changes in community functional features along natural and anthropogenic stress gradients. Ecology 96(9):2458–2466. https://doi.org/10.1890/14-1447.1
Heino J, Bini LM, Andersson J, Bergsten J, Bjelke U, Johansson F (2017) Unravelling the correlates of species richness and ecological uniqueness in a metacommunity of urban pond insects. Ecol Indic 73:422–431. https://doi.org/10.1016/j.ecolind.2016.10.006
Heino J, García Girón J, Hämäläinen H et al (2022) Assessing the conservation priority of freshwater lake sites based on taxonomic, functional and environmental uniqueness. Divers Distrib 28(9):1966–1978
Hermoso V, Ward DP, Kennard MJ (2013) Prioritizing refugia for freshwater biodiversity conservation in highly seasonal ecosystems. Divers Distrib 19:1031–1042. https://doi.org/10.1111/ddi.12082
Hershkovitz Y, Gasith A (2013) Resistance, resilience, and community dynamics in Mediterranean-climate streams. Hydrobiologia 719:59–75. https://doi.org/10.1007/s10750-012-1387-3
Hijmans RJ (2020) raster: Geographic Data Analysis and Modeling. R package version 3.1–5. https://CRAN.R-project.org/package=raster
Hill SL, Harfoot M, Purvis A et al (2016) Reconciling Biodiversity Indicators to Guide Understanding and Action. Conserv Lett 9:405–412. https://doi.org/10.1111/conl.12291
Hooper DU, Chapin FS, Ewel JJ et al (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75:3–35. https://doi.org/10.1890/04-0922
Koleff P, Gaston KJ, Lennon JJ (2003) Measuring beta diversity for presence-absence data. J Anim Ecol 72:367–382. https://doi.org/10.1046/j.1365-2656.2003.00710.x
Landeiro VL, Franz B, Heino J, Siqueira T, Bini LM (2018) Species-poor and low-lying sites are more ecologically unique in a hyperdiverse Amazon region: Evidence from multiple taxonomic groups. Divers Distrib 24:966–977. https://doi.org/10.1111/ddi.12734
Legendre P (2019) A temporal beta-diversity index to identify sites that have changed in exceptional ways in space–time surveys. Ecol Evol 9(6):3500–3514
Legendre P, Condit R (2019) Spatial and temporal analysis of beta diversity in the Barro Colorado Island forest dynamics plot. Panama for Ecosyst 6:7. https://doi.org/10.1186/s40663-019-0164-4
Legendre P, De Cáceres M (2013) Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecol Lett 16:951–963. https://doi.org/10.1111/ele.12141
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129(2):271–280
Legendre P, Gauthier O (2014) Statistical methods for temporal and space–time analysis of community composition data. Proc R Soc B 281:2013–2728. https://doi.org/10.1098/rspb.2013.2728
Leigh C, Bonada N, Boulton AJ, Hugueny B, Larned ST, Vander Vorste R, Datry T (2016) Invertebrate assemblage responses and the dual roles of resistance and resilience to drying in intermittent rivers. Aquat Sci 78:291. https://doi.org/10.1007/s00027-015-0427-2
Leigh C, Aspin TWH, Matthews TJ, Rolls RJ, Ledger ME (2019) Drought alters the functional stability of stream invertebrate communities through time. J Biogeogr 46:1988–2000. https://doi.org/10.1111/jbi.13638
Lytle DA, Poff NL (2004) Adaptation to natural flow regimes. Trends Ecol Evol 19(2):94–100. https://doi.org/10.1016/j.tree.2003.10.002
MAGRAMA (2013) “Guía de Playas de España. Base de Datos”, en Cartografía y SIG, Infraestructura de datos espaciales-Descargas, Madrid, Ministerio de Agricultura, Alimentación y Medio Ambiente. Retrieved from http://www.magrama.gob.es/es/carto grafia-y-sig/ide/desca rgas/costas-medio-marino/default.aspx. Accessed 17 Oct 2019
Magurran AE, Dornelas M, Moyes F, Henderson PA (2019) Temporal β diversity—a macroecological perspective. Glob Ecol Biogeogr 28(12):1949–1960. https://doi.org/10.1111/geb.13026
McGill BJ, Dornelas M, Gotelli NJ, Magurran AE (2015) Fifteen forms of biodiversity trend in the Anthropocene. Trends Ecol Evol 30(2):104–113
Messager ML, Lehner B, Cockburn C et al (2021) Global prevalence of non-perennial rivers and streams. Nature 594:391–397. https://doi.org/10.1038/s41586-021-03565-5
Moidu H, Rodríguez-Lozano P, Leidy RA, Bogan MT, Grantham TE, Carlson SM (2023) Ecological consequences of shifting habitat mosaics within and across years in an intermittent stream. Freshw Biol 68:1161–1175. https://doi.org/10.1111/fwb.14094
Newbold T, Oppenheimer P, Etard A, Williams JJ (2020) Tropical and Mediterranean biodiversity is disproportionately sensitive to land-use and climate change. Nat Ecol Evol 4:1630–1638. https://doi.org/10.1038/s41559-020-01303-0
Oksanen JFG, Blanchet R, Kindt P et al (2013) Vegan: Community Ecology Package. R package version 2.0–9. Available from URL: http://CRAN.Rproject.org/package Accessed 14 Dec 2019
Pereira HM, Ferrier S, Walters M et al (2013) Essential Biodiversity Variables. Science 339(6117):277–278. https://doi.org/10.1126/science.1229931
Pinheiro J, Bates D, DebRoy S, Sarkar D, Heisterkamp S, Van Willigen B (2016) Linear and nonlinear mixed effects models. R package version 3.1–127. Available from http://bugs.r-project.org. Accessed 14 Dec 2019
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ribera I, Vogler AP (2000) Habitat type as a determinant of species range sizes: the example of lotic-lentic differences in aquatic Coleoptera. Biol J Linn Soc 71:33–52
Rodrigues-Capítulo A, Muñoz I, Bonada N, Gaudes A, Tomanova S (2009) La biota de los ríos: los invertebrados. In: Elosegi A, Sabater S (eds) Conceptos y técnicas en ecologia fluvial. Fundación BBVA, pp 253–270
Rodríguez-Lozano P, Lobera G, Pardo I, García L, García C (2023) Conservation of temporary streams: the relevance of spatiotemporal variation in beta diversity. Aquat Conserv Mar Freshw Ecosyst 33(10):1014–1027. https://doi.org/10.1002/aqc.4005
Rogosch JS, Olden JD (2019) Dynamic contributions of intermittent and perennial streams to fish beta diversity in dryland rivers. J Biogeogr 46:2311–2322. https://doi.org/10.1111/jbi.13673
Ruhí A, Datry T, Sabo JL (2017) Interpreting beta-diversity components over time to conserve metacommunities in highly dynamic ecosystems. Conserv Biol. https://doi.org/10.1111/cobi.12906
Sánchez-Montoya MM, Vidal-Abarca MR, Puntí T et al (2009) Defining criteria to select reference sites in Mediterranean streams. Hydrobiologia 619(1):39–54. https://doi.org/10.1007/s10750-008-9580-0
Sánchez-Montoya MM, Tockner K, von Schiller D et al (2020) Dynamics of ground-dwelling arthropod metacommunities in intermittent streams: the key role of dry riverbeds. Biol Conserv 241:108328. https://doi.org/10.1016/j.biocon.2019.108328
Shimadzu H, Dornelas M, Magurran AE (2015) Measuring temporal turnover in ecological communities. Methods Ecol Evol 6:1384–1394. https://doi.org/10.1111/2041-210X.12438
Socolar JB, Gilroy JJ, Kunin WE, Edwards DP (2016) How Should Beta-Diversity Inform Biodiversity Conservation? Trends Ecol Evol 31:67–80. https://doi.org/10.1016/j.tree.2015.11.005
Soria M, Gutiérrez-Cánovas C, Bonada N et al (2019) Data from: Natural disturbances can produce misleading bioassessment results: identifying metrics to detect anthropogenic impacts in intermittent rivers. Dryad Dataset. https://doi.org/10.5061/dryad.n2z34tms5
Soria M, Gutiérrez-Cánovas C, Bonada N et al (2020) Natural disturbances can produce misleading bioassessment results: Identifying metrics to detect anthropogenic impacts in intermittent rivers. J Appl Ecol 57(2):283–295. https://doi.org/10.1111/1365-2664.13538
Stegen JC, Freestone AL, Crist TO et al (2013) Stochastic and deterministic drivers of spatial and temporal turnover in breeding bird communities. Glob Ecol Biogeogr 22(2):202–212 (PMID: ISI:000313267000006)
Stubbington R, Bogan MT, Bonada N, Boulton AJ, Datry T, Leigh C, Vander Vorste R (2017) The biota of intermittent rivers and ephemeral streams: aquatic invertebrates. In: Datry T, Bonada N, Boulton AJ (eds) Intermittent rivers and ephemeral streams: ecology and management. Elsevier, Waltham, pp 217–243
Stubbington R, Sarremejane R, Datry T (2019) Alpha and beta diversity of connected benthic–subsurface invertebrate communities respond to drying in dynamic river ecosystems. Ecography 42:2060–2073. https://doi.org/10.1111/ecog.04592
Tachet H, Richoux P, Bournaud M, Usseglio-Polatera P (2010) Invertébrés d’eau Douce: Systématique, Biologie, Écologie. CNRS Éditions, Paris
Tobias N, Monika W (2011) Does taxonomic homogenisation imply functional homogenisation in temperate forest herb layer communities? Plant Ecol 213:431–443. https://doi.org/10.1007/s11258-011-9990-3
Tonkin JD, Bogan MT, Bonada N, Rios-Touma B, Lytle DA (2017) Seasonality and predictability shape temporal species diversity. Ecology 98:1201–1216. https://doi.org/10.1002/ecy.1761
Valente-Neto F, da Silva FH, Covich AP et al (2020) Streams dry and ecological uniqueness rise: environmental selection drives aquatic insect patterns in a stream network prone to intermittence. Hydrobiologia 847:617–628. https://doi.org/10.1007/s10750-019-04125-9
Vander Vorste R, Stubbington R, Acuña V, Bogan MT, Bonada N, Cid N, Datry T, Storey R, Wood PJ, Ruhí A (2021) Climatic aridity increases temporal nestedness of invertebrate communities in naturally drying rivers. Ecography 44:860–869. https://doi.org/10.1111/ecog.05349
Villéger S, Mason NWH, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301. https://doi.org/10.1890/07-1206.1
Villéger S, Grenouillet G, Brosse S (2013) Decomposing functional beta-diversity reveals that low functional beta-diversity is driven by low functional turnover in European fish assemblages. Glob Ecol Biogeogr 22:671–681. https://doi.org/10.1111/geb.12021
Villéger S, Brosse S, Mouchet M, Mouillot D, Vanni MJ (2017) Functional ecology of fish: current approaches and future challenges. Aquat Sci 79:783–801. https://doi.org/10.1007/s00027-017-0546-z
Vilmi A, Karjalainen SM, Heino J (2017) Ecological uniqueness of stream and lake diatom communities shows different macroecological patterns. Divers Distrib 23:1042–1053. https://doi.org/10.1111/ddi.12594
Yu S, Rose PM, Bond NR, Bunn SE, Kennard MJ (2022) Identifying priority aquatic refuges to sustain freshwater biodiversity in intermittent streams in eastern Australia. Aquat Conserv Mar Freshw 32(10):1584–1595. https://doi.org/10.1002/aqc.3871
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. The study was supported by the LIFE+ TRivers (LIFE13 ENV/ES/000341) project. MS was supported by the Scholarship for studies or projects outside Catalonia funded by “Fundació Universitària Agustí Pedro i Pons” and the Iberoamerican Mobility Scholarship funded by “Banco Santander”. CG-C was supported by a Junior Leader Fellowship contract (LCF/BQ/PR22/11920005) funded by “la Caixa” Foundation (ID 100010434). PR-L was supported by a Margalida Comas postdoctoral contract (PD/031/2018) funded by the Government of the Balearic Islands and the European Social Fund and by a Juan de la Cierva-Incorporación fellowship (IJC2019-041601-I).
Author information
Authors and Affiliations
Contributions
MS, JO, NC, LMB and NB conceived the ideas and designed the methodology; NC, PR-L, PF and DV collected the data; RA identified the macroinvertebrate data; MS, JO and LMB analysed the data; MS, JO, LMB, NB and NC led the writing of the manuscript; and CG-C, RA, PR-L, PF, DV, FG and NP contributed to the drafts. All authors gave final approval for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soria, M., Cid, N., Ortega, J. et al. Analysing the contribution of intermittent rivers to beta diversity can improve freshwater conservation in Mediterranean rivers. Aquat Sci 86, 66 (2024). https://doi.org/10.1007/s00027-024-01074-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-024-01074-x