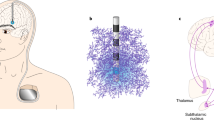
Abstract
Brain-machine interfaces (BMI) are now entering the clinical realm, where signals measured from the human brain are utilized to provide innovative therapies and enhance quality of life for individuals affected by neurological diseases and injury. Motor BMIs describe devices driven by signals from the motor system, which includes regions both on the surface and in deeper portions of the brain. These devices can be used to restore or enhance function for those with deficits in motor output as well as to provide therapy for individuals with deficits in part of the motor circuit function. Implanted motor BMIs, particularly those based on electrocorticography (ECoG) and deep brain stimulation (DBS) technologies, are burgeoning as a result of advances in wireless, implanted technologies in humans, and are based on foundational advances developed in research laboratories. Motor BMIs have now enabled an individual with amyotrophic lateral sclerosis (ALS) to communicate with the external world in a novel manner, and are being explored for closed-loop, adaptive deep brain stimulation therapies. In these therapies, electrical stimulation of deep brain structures used to treat movement disorders such as Parkinson’s disease and essential tremor is modulated in real time to provide more efficient stimulation with potentially fewer side effects. Future advances will be based upon further hardware and algorithmic developments, co-adaptive strategies that utilize learning both by the human brain and the implanted device, and integration with stimulation to provide more effective therapy for an expanding repertoire of movement disorders, restore sensation, and modulate cortical activity.
Similar content being viewed by others
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- BMI:
-
Brain-machine interface
- CM:
-
Centromedian region of thalamus
- CM-PF:
-
Centromedian-parafascicular complex
- CT:
-
Computed tomography
- DBS:
-
Deep brain stimulation
- ECoG:
-
Electrocorticography
- EEG:
-
Electroencephalography
- ERP:
-
Event related potential
- ET:
-
Essential tremor
- FDA:
-
Food and Drug Administration
- FES:
-
Functional electrical stimulation
- fMRI:
-
Functional magnetic resonance imaging
- fNIRS:
-
Functional near-infrared spectroscopy
- GPe:
-
Globus pallidus externus
- GPi:
-
Globus pallidus internus
- Hz:
-
Hertz
- IDE:
-
Investigational device exemption
- iEEG:
-
Intracranial electroencephalography
- LFP:
-
Local field potential
- MEG:
-
Magnetoencephalography
- MRI:
-
Magnetic resonance imaging
- NHP:
-
Nonhuman primate
- OCD:
-
Obsessive compulsive disorder
- PAC:
-
Phase amplitude coupling
- PD:
-
Parkinson’s disease
- SCI:
-
Spinal cord injury
- sEEG:
-
Stereoelectroencephalography
- STN:
-
Subthalamic nucleus
- Vim:
-
Ventral intermediate nucleus of thalamus
- VNS:
-
Vagus nerve stimulation
- VTA:
-
Volume of tissue activated
References
Thakor, N.V.: Translating the brain-machine interface. Sci. Transl. Med. 5, 210ps17 (2013). https://doi.org/10.1126/scitranslmed.3007303
Leuthardt, E.C., Schalk, G., Moran, D., Ojemann, J.G.: The emerging world of motor neuroprosthetics: a neurosurgical perspective. Neurosurgery. 59, 1–13 (2006). https://doi.org/10.1227/01.NEU.0000221506.06947.AC
Buzsáki, G., Anastassiou, C.A., Koch, C.: The origin of extracellular fields and currents – EEG, ECoG, LFP and spikes. Nat. Rev. Neurosci. 13, 407–420 (2012). https://doi.org/10.1038/nrn3241
Pesaran, B., Vinck, M., Einevoll, G.T., et al.: Investigating large-scale brain dynamics using field potential recordings: analysis and interpretation. Nat. Neurosci. 21, 903–919 (2018). https://doi.org/10.1038/s41593-018-0171-8
Armour, B.S., Courtney-Long, E.A., Fox, M.H., et al.: Prevalence and causes of paralysis – United States, 2013. Am. J. Public Health. 106, 1855–1857 (2016). https://doi.org/10.2105/AJPH.2016.303270
Lloyd-Jones, D., Adams, R.J., Brown, T.M., et al.: Heart disease and stroke statistics – 2010 update: a report from the American Heart Association. Circulation. 121, e46–e215 (2010). https://doi.org/10.1161/CIRCULATIONAHA.109.192667
Ziegler-Graham, K., MacKenzie, E.J., Ephraim, P.L., et al.: Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch. Phys. Med. Rehabil. 89, 422–429 (2008). https://doi.org/10.1016/j.apmr.2007.11.005
Arthur, K.C., Calvo, A., Price, T.R., et al.: Projected increase in amyotrophic lateral sclerosis from 2015 to 2040. Nat. Commun. 7, 1–6 (2016). https://doi.org/10.1038/ncomms12408
Anderson, K.D.: Targeting recovery: priorities of the spinal cord-injured population. J. Neurotrauma. 21, 1371–1383 (2004). https://doi.org/10.1089/neu.2004.21.1371
Snoek, G.J., Ijzerman, M.J., Hermens, H.J., et al.: Survey of the needs of patients with spinal cord injury: impact and priority for improvement in hand function in tetraplegics. Spinal Cord. 42, 526–532 (2004). https://doi.org/10.1038/sj.sc.3101638
Collinger, J.L., Boninger, M.L., Bruns, T.M., et al.: Functional priorities, assistive technology, and brain-computer interfaces after spinal cord injury. J. Rehabil. Res. Dev. 50, 145 (2013). https://doi.org/10.1682/JRRD.2011.11.0213
Lahr, J., Schwartz, C., Heimbach, B., et al.: Invasive brain–machine interfaces: a survey of paralyzed patients’ attitudes, knowledge and methods of information retrieval. J. Neural Eng. 12, 043001 (2015). https://doi.org/10.1088/1741-2560/12/4/043001
Dorsey, E.R., Constantinescu, R., Thompson, J.P., et al.: Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology. 68, 384–386 (2007). https://doi.org/10.1212/01.wnl.0000247740.47667.03
Jerbi, K., Vidal, J.R., Mattout, J., et al.: Inferring hand movement kinematics from MEG, EEG and intracranial EEG: from brain-machine interfaces to motor rehabilitation. ITBM-RBM. 32, 8–18 (2011). https://doi.org/10.1016/j.irbm.2010.12.004
LaFleur, K., Cassady, K., Doud, A., et al.: Quadcopter control in three-dimensional space using a noninvasive motor imagery-based brain-computer interface. J. Neural Eng. 10, 046003 (2013). https://doi.org/10.1088/1741-2560/10/4/046003
Hillman, E.M.C.: Coupling mechanism and significance of the BOLD signal: a status report. Annu. Rev. Neurosci. 37, 161–181 (2014). https://doi.org/10.1146/annurev-neuro-071013-014111
Lee, J.H., Ryu, J., Jolesz, F.A., et al.: Brain-machine interface via real-time fMRI: preliminary study on thought-controlled robotic arm. Neurosci. Lett. 450, 1–6 (2009). https://doi.org/10.1016/j.neulet.2008.11.024
Coyle, S.M., Ward, T.E., Markham, C.M.: Brain-computer interface using a simplified functional near-infrared spectroscopy system. J. Neural Eng. 4, 219–226 (2007). https://doi.org/10.1088/1741-2560/4/3/007
Fukuma, R., Yanagisawa, T., Saitoh, Y., et al.: Real-time control of a neuroprosthetic hand by magnetoencephalographic signals from paralysed patients. Sci. Rep. 6, 21781 (2016). https://doi.org/10.1038/srep21781
Waldert, S., Preissl, H., Demandt, E., et al.: Hand movement direction decoded from MEG and EEG. J. Neurosci. 28, 1000–1008 (2008). https://doi.org/10.1523/JNEUROSCI.5171-07.2008
Collinger, J.L., Wodlinger, B., Downey, J.E., et al.: High-performance neuroprosthetic control by an individual with tetraplegia. Lancet. 381, 557–564 (2013). https://doi.org/10.1016/S0140-6736(12)61816-9
Hochberg, L.R., Bacher, D., Jarosiewicz, B., et al.: Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature. 485, 372–375 (2012). https://doi.org/10.1038/nature11076
Jarosiewicz, B., Sarma, A.A., Bacher, D., et al.: Virtual typing by people with tetraplegia using a self-calibrating intracortical brain-computer interface. Sci. Transl. Med. 7, 313ra179 (2015). https://doi.org/10.1126/scitranslmed.aac7328
Ball, T., Kern, M., Mutschler, I., et al.: Signal quality of simultaneously recorded invasive and non-invasive EEG. NeuroImage. 46, 708–716 (2009). https://doi.org/10.1016/j.neuroimage.2009.02.028
Kanth, S.T., Ray, S.: Electrocorticogram (ECoG) is highly informative in primate visual cortex. J. Neurosci. 40, 2430–2444 (2020). https://doi.org/10.1523/JNEUROSCI.1368-19.2020
Kellis, S., Sorensen, L., Darvas, F., et al.: Multi-scale analysis of neural activity in humans: implications for micro-scale electrocorticography. Clin. Neurophysiol. 127, 591–601 (2016). https://doi.org/10.1016/j.clinph.2015.06.002
Kandel, E.R., Schwartz, J.H., Jessell, T.M., et al.: Principles of Neural Science. McGraw-Hill, New York (2013)
Zhang, Y., Larcher, K.M.H., Misic, B., Dagher, A.: Anatomical and functional organization of the human substantia nigra and its connections. elife. 6, 1–23 (2017). https://doi.org/10.7554/eLife.26653
Bostan, A.C., Strick, P.L.: The basal ganglia and the cerebellum: nodes in an integrated network. Nat. Rev. Neurosci. 19, 338–350 (2018). https://doi.org/10.1038/s41583-018-0002-7
Lanciego, J.L., Luquin, N., Obeso, J.A.: Functional neuroanatomy of the basal ganglia. Cold Spring Harb. Perspect. Med. 2, a009621 (2012). https://doi.org/10.1101/cshperspect.a009621
Herrero, M.T., Barcia, C., Navarro, J.M.: Functional anatomy of thalamus and basal ganglia. Childs Nerv. Syst. 18, 386–404 (2002). https://doi.org/10.1007/s00381-002-0604-1
Obeso, J.A., Rodriguez-Oroz, M.C., Rodriguez, M., et al.: Pathophysiology of the basal ganglia in Parkinson’s disease. Trends Neurosci. 23, S8–S19 (2000). https://doi.org/10.1016/S1471-1931(00)00028-8
Lehericy, S., Benali, H., Van de Moortele, P.-F., et al.: Distinct basal ganglia territories are engaged in early and advanced motor sequence learning. Proc. Natl. Acad. Sci. U. S. A. 102, 12566–12571 (2005). https://doi.org/10.1073/pnas.0502762102
Ackerley, R., Kavounoudias, A.: The role of tactile afference in shaping motor behaviour and implications for prosthetic innovation. Neuropsychologia. 79, 192–205 (2015). https://doi.org/10.1016/j.neuropsychologia.2015.06.024
Saal, H.P., Bensmaia, S.J.: Touch is a team effort: interplay of submodalities in cutaneous sensibility. Trends Neurosci. 37, 689–697 (2014). https://doi.org/10.1016/j.tins.2014.08.012
Sanes, J.N., Donoghue, J.P.: Plasticity and primary motor cortex. Annu. Rev. Neurosci. 23, 393–415 (2000). https://doi.org/10.1146/annurev.neuro.23.1.393
Brown, C.E., Aminoltejari, K., Erb, H., et al.: In vivo voltage-sensitive dye imaging in adult mice reveals that somatosensory maps lost to stroke are replaced over weeks by new structural and functional circuits with prolonged modes of activation within both the peri-infarct zone and distant sites. J. Neurosci. 29, 1719–1734 (2009). https://doi.org/10.1523/JNEUROSCI.4249-08.2009
Darian-Smith, C.: Plasticity of somatosensory function during learning, disease and injury. In: The Senses: A Comprehensive Reference, pp. 259–297. Elsevier, Amsterdam/Boston (2008)
Krakauer, J.W., Hadjiosif, A.M., Xu, J., et al.: Motor learning. Compr. Physiol. 9, 613–663 (2019). https://doi.org/10.1002/cphy.c170043
Bashashati, A., Fatourechi, M., Ward, R.K., Birch, G.E.: A survey of signal processing algorithms in brain-computer interfaces based on electrical brain signals. J. Neural Eng. 4, R32–R57 (2007). https://doi.org/10.1088/1741-2560/4/2/R03
Bensch, M., Martens, S., Halder, S., et al.: Assessing attention and cognitive function in completely locked-in state with event-related brain potentials and epidural electrocorticography. J. Neural Eng. 11, 026006 (2014). https://doi.org/10.1088/1741-2560/11/2/026006
Buzsaki, G.: Neuronal oscillations in cortical networks. Science. 304, 1926–1929 (2004). https://doi.org/10.1126/science.1099745
Bruns, A.: Fourier-, Hilbert- and wavelet-based signal analysis: are they really different approaches? J. Neurosci. Methods. 137, 321–332 (2004). https://doi.org/10.1016/j.jneumeth.2004.03.002
Miller, K.J., Leuthardt, E.C., Schalk, G., et al.: Spectral changes in cortical surface potentials during motor movement. J. Neurosci. 27, 2424–2432 (2007). https://doi.org/10.1523/JNEUROSCI.3886-06.2007
Canolty, R.T., Edwards, E., Dalal, S.S., et al.: High gamma power is phase-locked to theta oscillations in human neocortex. Science. 313, 1626–1628 (2006). https://doi.org/10.1126/science.1128115
Miller, K.J., Shenoy, P., den Nijs, M., et al.: Beyond the gamma band: the role of high-frequency features in movement classification. IEEE Trans. Biomed. Eng. 55, 1634–1637 (2008). https://doi.org/10.1109/TBME.2008.918569
Hermes, D., Nguyen, M., Winawer, J.: Neuronal synchrony and the relation between the blood-oxygen-level dependent response and the local field potential. PLoS Biol. 15(7), e2001461 (2017)
Wander, J.D., Blakely, T., Miller, K.J., et al.: Distributed cortical adaptation during learning of a brain-computer interface task. Proc. Natl. Acad. Sci. U. S. A. 110, 10818–10823 (2013). https://doi.org/10.1073/pnas.1221127110
Brown, P.: Oscillatory nature of human basal ganglia activity: relationship to the pathophysiology of Parkinson’s disease. Mov. Disord. 18, 357–363 (2003). https://doi.org/10.1002/mds.10358
Ashkan, K., Rogers, P., Bergman, H., Ughratdar, I.: Insights into the mechanisms of deep brain stimulation. Nat. Rev. Neurol. 13, 548–554 (2017). https://doi.org/10.1038/nrneurol.2017.105
Flint, R.D., Rosenow, J.M., Tate, M.C., et al.: Continuous decoding of human grasp kinematics using epidural and subdural signals. J. Neural Eng. 14, 016005 (2017). https://doi.org/10.1088/1741-2560/14/1/016005
Chestek, C.A., Gilja, V., Blabe, C.H., et al.: Hand posture classification using electrocorticography signals in the gamma band over human sensorimotor brain areas. J. Neural Eng. 10, 026002 (2013). https://doi.org/10.1088/1741-2560/10/2/026002
Hotson, G., McMullen, D.P., Fifer, M.S., et al.: Individual finger control of a modular prosthetic limb using high-density electrocorticography in a human subject. J. Neural Eng. 13, 026017 (2016). https://doi.org/10.1088/1741-2560/13/2/026017
Leuthardt, E.C., Schalk, G., Wolpaw, J.R., et al.: A brain-computer interface using electrocorticographic signals in humans. J. Neural Eng. 1, 63–71 (2004). https://doi.org/10.1088/1741-2560/1/2/001
Leuthardt, E.C., Miller, K.J., Schalk, G., et al.: Electrocorticography-based brain computer interface – the seattle experience. IEEE Trans. Neural Syst. Rehabil. Eng. 14, 194–198 (2006). https://doi.org/10.1109/TNSRE.2006.875536
Santello, M., Flanders, M., Soechting, J.F.: Postural hand synergies for tool use. J. Neurosci. 18, 10105–10115 (1998). https://doi.org/10.1523/JNEUROSCI.18-23-10105.1998
Branco, M.P., Freudenburg, Z.V., Aarnoutse, E.J., et al.: Decoding hand gestures from primary somatosensory cortex using high-density ECoG. NeuroImage. 147, 130–142 (2017). https://doi.org/10.1016/j.neuroimage.2016.12.004
Candrea, D.N., McMullen, D.P., Fifer, M.S., et al.: Decoding native cortical representations for flexion and extension at upper limb joints using electrocorticography. IEEE Trans. Neural Syst. Rehabil. Eng. 27, 293–303 (2019). https://doi.org/10.1109/tnsre.2019.2891362
Voytek, B., Knight, R.T.: Dynamic network communication as a unifying neural basis for cognition, development, aging, and disease. Biol. Psychiatry. 77, 1089–1097 (2015). https://doi.org/10.1016/j.biopsych.2015.04.016
Yuste, R.: From the neuron doctrine to neural networks. Nat. Rev. Neurosci. 16, 487–497 (2015). https://doi.org/10.1038/nrn3962
Opri, E., Cernera, S., Okun, M.S., et al.: The functional role of thalamocortical coupling in the human motor network. J. Neurosci. 39, 1153–1119 (2019). https://doi.org/10.1523/jneurosci.1153-19.2019
De Hemptinne, C., Ryapolova-Webb, E.S., Air, E.L., et al.: Exaggerated phase-amplitude coupling in the primary motor cortex in Parkinson disease. Proc. Natl. Acad. Sci. U. S. A. 110, 4780–4785 (2013). https://doi.org/10.1073/pnas.1214546110
de Hemptinne, C., Swann, N.C., Ostrem, J.L., et al.: Therapeutic deep brain stimulation reduces cortical phase-amplitude coupling in Parkinson’s disease. Nat. Neurosci. 18, 779–786 (2015). https://doi.org/10.1038/nn.3997
Bruurmijn, M.L.C.M., Pereboom, I.P.L., Vansteensel, M.J., et al.: Preservation of hand movement representation in the sensorimotor areas of amputees. Brain. 140, 3166–3178 (2017). https://doi.org/10.1093/brain/awx274
Yanagisawa, T., Hirata, M., Saitoh, Y., et al.: Real-time control of a prosthetic hand using human electrocorticography signals: technical note. J. Neurosurg. 114, 1715–1722 (2011). https://doi.org/10.3171/2011.1.JNS101421
Yanagisawa, T., Hirata, M., Saitoh, Y., et al.: Neural decoding using gyral and intrasulcal electrocorticograms. NeuroImage. 45, 1099–1106 (2009). https://doi.org/10.1016/j.neuroimage.2008.12.069
Freudenburg, Z.V., Branco, M.P., Leinders, S., et al.: Sensorimotor ECoG signal features for BCI control: a comparison between people with locked-in syndrome and able-bodied controls. Front. Neurosci. 13, 1–18 (2019). https://doi.org/10.3389/fnins.2019.01058
Blakely, T., Miller, K.J., Zanos, S.P., et al.: Robust, long-term control of an electrocorticographic brain-computer interface with fixed parameters. Neurosurg. Focus. 27, E13 (2009). https://doi.org/10.3171/2009.4.FOCUS0977
Chao, Z.C., Nagasaka, Y., Fujii, N.: Long-term asynchronous decoding of arm motion using electrocorticographic signals in monkey. Front. Neuroeng. 3, 3 (2010). https://doi.org/10.3389/fneng.2010.00003
Pels, E.G.M., Aarnoutse, E.J., Leinders, S., et al.: Stability of a chronic implanted brain-computer interface in late-stage amyotrophic lateral sclerosis. Clin. Neurophysiol. 130, 1798–1803 (2019). https://doi.org/10.1016/j.clinph.2019.07.020
Moran, D.: Evolution of brain-computer interface: action potentials, local field potentials and electrocorticograms. Curr. Opin. Neurobiol. 20, 741–745 (2010). https://doi.org/10.1016/j.conb.2010.09.010
Ward, M.P., Rajdev, P., Ellison, C., Irazoqui, P.P.: Toward a comparison of microelectrodes for acute and chronic recordings. Brain Res. 1282, 183–200 (2009). https://doi.org/10.1016/j.brainres.2009.05.052
Perge, J.A., Homer, M.L., Malik, W.Q., et al.: Intra-day signal instabilities affect decoding performance in an intracortical neural interface system. J. Neural Eng. 10, 036004 (2013). https://doi.org/10.1088/1741-2560/10/3/036004
Downey, J.E., Schwed, N., Chase, S.M., et al.: Intracortical recording stability in human brain-computer interface users. J. Neural Eng. 15, 046016 (2018). https://doi.org/10.1088/1741-2552/aab7a0
Degenhart, A.D., Bishop, W.E., Oby, E.R., et al.: Stabilization of a brain–computer interface via the alignment of low-dimensional spaces of neural activity. Nat. Biomed. Eng. (2020). https://doi.org/10.1038/s41551-020-0542-9
Winestone, J.S., Zaidel, A., Bergman, H., Israel, Z.: The use of macroelectrodes in recording cellular spiking activity. J. Neurosci. Methods. 206, 34–39 (2012). https://doi.org/10.1016/j.jneumeth.2012.02.002
Thompson, J.A., Lanctin, D., Ince, N.F., Abosch, A.: Clinical implications of local field potentials for understanding and treating movement disorders. Stereotact. Funct. Neurosurg. 92, 251–263 (2014). https://doi.org/10.1159/000364913
Chang, E.F.: Towards large-scale, human-based, mesoscopic neurotechnologies. Neuron. 86, 68–78 (2015). https://doi.org/10.1016/j.neuron.2015.03.037
Muller, L., Hamilton, L.S., Edwards, E., et al.: Spatial resolution dependence on spectral frequency in human speech cortex electrocorticography. J. Neural Eng. 13, 056013 (2016). https://doi.org/10.1088/1741-2560/13/5/056013
Hermiz, J., Rogers, N., Kaestner, E., et al.: Sub-millimeter ECoG pitch in human enables higher fidelity cognitive neural state estimation. NeuroImage. 176, 454–464 (2018). https://doi.org/10.1016/j.neuroimage.2018.04.027
Khodagholy, D., Gelinas, J.N., Zhao, Z., et al.: Organic electronics for high-resolution electrocorticography of the human brain. Sci. Adv. 2, e1601027 (2016). https://doi.org/10.1126/sciadv.1601027
Khodagholy, D., Gelinas, J.N., Thesen, T., et al.: NeuroGrid: recording action potentials from the surface of the brain. Nat. Neurosci. 18, 310–315 (2015). https://doi.org/10.1038/nn.3905
Katz, J.S., Abel, T.J.: Stereoelectroencephalography versus subdural electrodes for localization of the epileptogenic zone: what is the evidence? Neurotherapeutics. 16, 59–66 (2019). https://doi.org/10.1007/s13311-018-00703-2
Bronstein, J.M., Tagliati, M., Alterman, R.L., et al.: Deep brain stimulation for Parkinson disease. Arch. Neurol. 68, 165–171 (2011). https://doi.org/10.1001/archneurol.2010.260
Della Flora, E., Perera, C.L., Cameron, A.L., Maddern, G.J.: Deep brain stimulation for essential tremor: a systematic review. Mov. Disord. 25, 1550–1559 (2010). https://doi.org/10.1002/mds.23195
Lee, D.J., Lozano, C.S., Dallapiazza, R.F., Lozano, A.M.: Current and future directions of deep brain stimulation for neurological and psychiatric disorders. J. Neurosurg. 131, 333–342 (2019). https://doi.org/10.3171/2019.4.JNS181761
Schrock, L.E., Mink, J.W., Woods, D.W., et al.: Tourette syndrome deep brain stimulation: a review and updated recommendations. Mov. Disord. 30, 448–471 (2015). https://doi.org/10.1002/mds.26094
Rasche, D., Rinaldi, P.C., Young, R.F., Tronnier, V.M.: Deep brain stimulation for the treatment of various chronic pain syndromes. Neurosurg. Focus. 21, 1–8 (2006). https://doi.org/10.3171/foc.2006.21.6.10
Lozano, A.M., Mayberg, H.S., Giacobbe, P., et al.: Subcallosal cingulate gyrus deep brain stimulation for treatment-resistant depression. Biol. Psychiatry. 64, 461–467 (2008). https://doi.org/10.1016/j.biopsych.2008.05.034
Anderson, D.N., Osting, B., Vorwerk, J., et al.: Optimized programming algorithm for cylindrical and directional deep brain stimulation electrodes. J. Neural Eng. 15, 026005 (2018). https://doi.org/10.1088/1741-2552/aaa14b
Montgomery, E.B., Gale, J.T.: Mechanisms of action of deep brain stimulation (DBS). Neurosci. Biobehav. Rev. 32, 388–407 (2008). https://doi.org/10.1016/j.neubiorev.2007.06.003
Ho, C.H., Triolo, R.J., Elias, A.L., et al.: Functional electrical stimulation and spinal cord injury. Phys. Med. Rehabil. Clin. N. Am. 25, 631–654 (2014). https://doi.org/10.1016/j.pmr.2014.05.001
Auchstaetter, N., Luc, J., Lukye, S., et al.: Physical therapists’ use of functional electrical stimulation for clients with stroke: frequency, barriers, and facilitators. Phys. Ther. 96, 995–1005 (2016). https://doi.org/10.2522/ptj.20150464
Thomas, C.K., Griffin, L., Godfrey, S., et al.: Fatigue of paralyzed and control thenar muscles induced by variable or constant frequency stimulation. J. Neurophysiol. 89, 2055–2064 (2003). https://doi.org/10.1152/jn.01002.2002
Ajiboye, A.B., Willett, F.R., Young, D.R., et al.: Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration. Lancet. 389, 1821–1830 (2017). https://doi.org/10.1016/S0140-6736(17)30601-3
Bouton, C.E., Shaikhouni, A., Annetta, N.V., et al.: Restoring cortical control of functional movement in a human with quadriplegia. Nature. 533, 1–13 (2016). https://doi.org/10.1038/nature17435
Capogrosso, M., Milekovic, T., Borton, D., et al.: A brain-spine interface alleviating gait deficits after spinal cord injury in primates. Nature. 539, 284–288 (2016). https://doi.org/10.1038/nature20118
Sohal, V.S., Sun, F.T.: Responsive neurostimulation suppresses synchronized cortical rhythms in patients with epilepsy. Neurosurg. Clin. N. Am. 22, 481–488 (2011). https://doi.org/10.1016/j.nec.2011.07.007
Morrell, M.J.: Responsive cortical stimulation for the treatment of medically intractable partial epilepsy. Neurology. 77, 1295–1304 (2011). https://doi.org/10.1212/WNL.0b013e3182302056
Lee, B., Zubair, M.N., Marquez, Y.D., et al.: A single-center experience with the NeuroPace RNS system: a review of techniques and potential problems. World Neurosurg. 84, 719–726 (2015). https://doi.org/10.1016/j.wneu.2015.04.050
Sun, F.T., Morrell, M.J.: Closed-loop neurostimulation: the clinical experience. Neurotherapeutics. 11, 553–563 (2014). https://doi.org/10.1007/s13311-014-0280-3
Herron, J.A., Thompson, M.C., Brown, T., et al.: Chronic electrocorticography for sensing movement intention and closed-loop deep brain stimulation with wearable sensors in an essential tremor patient. J. Neurosurg. 127, 580–587 (2017). https://doi.org/10.3171/2016.8.JNS16536
Herron, J.A., Thompson, M.C., Brown, T., et al.: Cortical brain–computer interface for closed-loop deep brain stimulation. IEEE Trans. Neural Syst. Rehabil. Eng. 25, 2180–2187 (2017). https://doi.org/10.1109/TNSRE.2017.2705661
Herron, J., Stanslaski, S., Chouinard, T., et al.: Bi-directional brain interfacing instrumentation. In: 2018 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), pp. 1–6. IEEE (2018).
Starr, P.A.: Totally implantable bidirectional neural prostheses: a flexible platform for innovation in neuromodulation. Front. Neurosci. 12, 1–5 (2018). https://doi.org/10.3389/fnins.2018.00619
Gilron, R., Little, S., Perrone, R., et al.: Long-term wireless streaming of neural recordings for circuit discovery and adaptive stimulation in individuals with Parkinson’s disease. Nat. Biotechnol. (2021). https://doi.org/10.1038/s41587-021-00897-5
Wang, W., Collinger, J.L., Degenhart, A.D., et al.: An electrocorticographic brain interface in an individual with tetraplegia. PLoS One. 8, 1–8 (2013). https://doi.org/10.1371/journal.pone.0055344
Degenhart, A.D., Hiremath, S.V., Yang, Y., et al.: Remapping cortical modulation for electrocorticographic brain–computer interfaces: a somatotopy-based approach in individuals with upper-limb paralysis. J. Neural Eng. 15, 026021 (2018). https://doi.org/10.1088/1741-2552/aa9bfb
Vansteensel, M.J., Pels, E.G.M., Bleichner, M.G., et al.: Fully implanted brain–computer interface in a locked-in patient with ALS. N. Engl. J. Med. 375, 2060–2066 (2016). https://doi.org/10.1056/NEJMoa1608085
Benabid, A.L., Costecalde, T., Eliseyev, A., et al.: An exoskeleton controlled by an epidural wireless brain–machine interface in a tetraplegic patient: a proof-of-concept demonstration. Lancet Neurol. 4422, 1–11 (2019). https://doi.org/10.1016/s1474-4422(19)30321-7
Khanna, P., Swann, N.C., De Hemptinne, C., et al.: Neurofeedback control in parkinsonian patients using electrocorticography signals accessed wirelessly with a chronic, fully implanted device. IEEE Trans. Neural Syst. Rehabil. Eng. 25, 1715–1724 (2017). https://doi.org/10.1109/TNSRE.2016.2597243
Subramanian, L., Hindle, J.V., Johnston, S., et al.: Real-time functional magnetic resonance imaging neurofeedback for treatment of Parkinson’s disease. J. Neurosci. 31, 16309–16317 (2011). https://doi.org/10.1523/JNEUROSCI.3498-11.2011
Kuo, C., White-Dzuro, GA., Ko, AL.: Approaches to closed-loop deep brain stimulation for movement disorders. Neurosurgical Focus. 45(2), E2 (2018). https://doi.org/10.3171/2018.5.FOCUS18173
Little, S., Pogosyan, A., Neal, S., et al.: Adaptive deep brain stimulation in advanced Parkinson disease. Ann. Neurol. 74, 449–457 (2013). https://doi.org/10.1002/ana.23951
Tinkhauser, G., Pogosyan, A., Little, S., et al.: The modulatory effect of adaptive deep brain stimulation on beta bursts in Parkinson’s disease. Brain. 140, 1053–1067 (2017). https://doi.org/10.1093/brain/awx010
Moraud, E.M., Tinkhauser, G., Agrawal, M., et al.: Predicting beta bursts from local field potentials to improve closed-loop DBS paradigms in Parkinson’s patients. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2018, 3766–3769 (2018). https://doi.org/10.1109/EMBC.2018.8513348
Velisar, A., Syrkin-Nikolau, J., Blumenfeld, Z., et al.: Dual threshold neural closed loop deep brain stimulation in Parkinson disease patients. Brain Stimul. 12, 868–876 (2019). https://doi.org/10.1016/j.brs.2019.02.020
Shah, S.A., Tinkhauser, G., Chen, C.C., et al.: Parkinsonian tremor detection from subthalamic nucleus local field potentials for closed-loop deep brain stimulation. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2018, 2320–2324 (2018). https://doi.org/10.1109/EMBC.2018.8512741
Kühn, A.A., Tsui, A., Aziz, T., et al.: Pathological synchronisation in the subthalamic nucleus of patients with Parkinson’s disease relates to both bradykinesia and rigidity. Exp. Neurol. 215, 380–387 (2009). https://doi.org/10.1016/j.expneurol.2008.11.008
Honey, C.R., Hamani, C., Kalia, S.K., et al.: Deep brain stimulation target selection for Parkinson’s disease. Can. J. Neurol. Sci. 44, 3–8 (2017). https://doi.org/10.1017/cjn.2016.22
Little, S., Beudel, M., Zrinzo, L., et al.: Bilateral adaptive deep brain stimulation is effective in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry. 87, 717–721 (2016). https://doi.org/10.1136/jnnp-2015-310972
Little, S., Tan, H., Anzak, A., et al.: Bilateral functional connectivity of the basal ganglia in patients with Parkinson’s disease and its modulation by dopaminergic treatment. PLoS One. 8, e82762 (2013). https://doi.org/10.1371/journal.pone.0082762
Malekmohammadi, M., Herron, J., Velisar, A., et al.: Kinematic adaptive deep brain stimulation for resting tremor in Parkinson’s disease. Mov. Disord. 31, 426–428 (2016). https://doi.org/10.1002/mds.26482
Swann, N.C., De Hemptinne, C., Miocinovic, S., et al.: Gamma oscillations in the hyperkinetic state detected with chronic human brain recordings in Parkinson’s disease. J. Neurosci. 36, 6445–6458 (2016). https://doi.org/10.1523/JNEUROSCI.1128-16.2016
Swann, N.C., De Hemptinne, C., Thompson, M.C., et al.: Adaptive deep brain stimulation for Parkinson’s disease using motor cortex sensing. J. Neural Eng. 15, 046006 (2018). https://doi.org/10.1088/1741-2552/aabc9b
Little, S., Tripoliti, E., Beudel, M., et al.: Adaptive deep brain stimulation for Parkinson’s disease demonstrates reduced speech side effects compared to conventional stimulation in the acute setting. J. Neurol. Neurosurg. Psychiatry. 87, 1388–1389 (2016). https://doi.org/10.1136/jnnp-2016-313518
Rosa, M., Arlotti, M., Marceglia, S., et al.: Adaptive deep brain stimulation controls levodopa-induced side effects in parkinsonian patients. Mov. Disord. 32, 628–629 (2017). https://doi.org/10.1002/mds.26953
Tan, H., Debarros, J., He, S., et al.: Decoding voluntary movements and postural tremor based on thalamic LFPs as a basis for closed-loop stimulation for essential tremor. Brain Stimul. 12, 858–867 (2019). https://doi.org/10.1016/j.brs.2019.02.011
Loza, C.A., Shute, J.B., Principe, J.C., et al.: A marked point process approach for identifying neural correlates of tics in Tourette Syndrome. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2017, 4375–4378 (2017). https://doi.org/10.1109/EMBC.2017.8037825
Cagle, J.N., Okun, M.S., Opri, E., et al.: Differentiating tic electrophysiology from voluntary movement in the human thalamocortical circuit. J. Neurol. Neurosurg. Psychiatry. 91, 533–539 (2020). https://doi.org/10.1136/jnnp-2019-321973
Cagnan, H., Pedrosa, D., Little, S., et al.: Stimulating at the right time: phase-specific deep brain stimulation. Brain. 140, 132–145 (2017). https://doi.org/10.1093/brain/aww286
Molina, R., Okun, M.S., Shute, J.B., et al.: Report of a patient undergoing chronic responsive deep brain stimulation for Tourette syndrome: proof of concept. J. Neurosurg. 129, 308–314 (2018). https://doi.org/10.3171/2017.6.JNS17626
Meidahl, A.C., Tinkhauser, G., Herz, D.M., et al.: Adaptive deep brain stimulation for movement disorders: the long road to clinical therapy. Mov. Disord. 32, 810–819 (2017). https://doi.org/10.1002/mds.27022
Mestre, T.A., Lang, A.E., Okun, M.S.: Factors influencing the outcome of deep brain stimulation: placebo, nocebo, lessebo, and lesion effects. Mov. Disord. 31, 290–298 (2016). https://doi.org/10.1002/mds.26500
Arlotti, M., Marceglia, S., Foffani, G., et al.: Eight-hours adaptive deep brain stimulation in patients with Parkinson disease. Neurology. 90, e971–e976 (2018). https://doi.org/10.1212/WNL.0000000000005121
Coffey, R.J.: Deep brain stimulation devices: a brief technical history and review. Artif. Organs. 33, 208–220 (2009). https://doi.org/10.1111/j.1525-1594.2008.00620.x
Boon, P., Vonck, K., Vandekerckhove, T., et al.: Vagus nerve stimulation for medically refractory epilepsy; efficacy and cost-benefit analysis. Acta Neurochir. 141, 447–453 (1999). https://doi.org/10.1007/s007010050324
Pereira, E.A., Green, A.L., Nandi, D., Aziz, T.Z.: Deep brain stimulation: indications and evidence. Expert Rev. Med. Devices. 4, 591–603 (2007). https://doi.org/10.1586/17434440.4.5.591
Eskandar, E.N., Flaherty, A., Cosgrove, G.R., et al.: Surgery for Parkinson disease in the United States, 1996 to 2000: practice patterns, short-term outcomes, and hospital charges in a nationwide sample. J. Neurosurg. 99, 863–871 (2009). https://doi.org/10.3171/jns.2003.99.5.0863
Lázaro-Muñoz, G., Yoshor, D., Beauchamp, M.S., et al.: Continued access to investigational brain implants. Nat. Rev. Neurosci. 19, 317–318 (2018). https://doi.org/10.1038/s41583-018-0004-5
Rossi, P.J., Giordano, J., Okun, M.S.: The problem of funding off-label deep brain stimulation. JAMA Neurol. 74, 9 (2017). https://doi.org/10.1001/jamaneurol.2016.2530
Hendriks, S., Grady, C., Ramos, K.M., et al.: Ethical challenges of risk, informed consent, and posttrial responsibilities in human research with neural devices: a review. JAMA Neurol. 76, 1506–1514 (2019). https://doi.org/10.1001/jamaneurol.2019.3523
Wang, X., Gkogkidis, A., Iljina, O., et al.: Mapping the fine structure of cortical activity with different micro-ECoG electrode array geometries. J. Neural Eng. 265, 197–212 (2017). https://doi.org/10.1088/1741-2552/aa785e
Wang, P.T., King, C.E., McCrimmon, C.M., et al.: Comparison of decoding resolution of standard and high-density electrocorticogram electrodes. J. Neural Eng. 13, 026016 (2016). https://doi.org/10.1088/1741-2560/13/2/026016
Muller, L., Felix, S., Shah, K.G., et al.: Thin-film, high-density micro-electrocorticographic decoding of a human cortical gyrus. In: 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), pp. 1528–1531. IEEE (2016).
Sadtler, P.T., Quick, K.M., Golub, M.D., et al.: Neural constraints on learning. Nature. 512, 423–426 (2014). https://doi.org/10.1038/nature13665
Golub, M.D., Sadtler, P.T., Oby, E.R., et al.: Learning by neural reassociation. Nat. Neurosci. 21, 607–616 (2018). https://doi.org/10.1038/s41593-018-0095-3
Sakellaridi, S., Christopoulos, V.N., Aflalo, T., et al.: Intrinsic variable learning for brain-machine interface control by human anterior intraparietal cortex. Neuron. 102, 694–705.e3 (2019). https://doi.org/10.1016/j.neuron.2019.02.012
Bashford, L., Wu, J., Sarma, D., et al.: Concurrent control of a brain-computer interface and natural overt movements. J. Neural Eng. 15, 066021 (2018). https://doi.org/10.1088/1741-2552/aadf3d
Makin, J.G., Moses, D.A., Chang, E.F.: Machine translation of cortical activity to text with an encoder–decoder framework. Nat. Neurosci. 23, 575–582 (2020). https://doi.org/10.1038/s41593-020-0608-8
Xie, Z., Schwartz, O., Prasad, A.: Decoding of finger trajectory from ECoG using deep learning. J. Neural Eng. 15, 036009 (2018). https://doi.org/10.1088/1741-2552/aa9dbe
Wang, N.X.R., Farhadi, A., Rao, R., Brunton, B.: AJILE movement prediction: multimodal deep learning for natural human neural recordings and video. In: AAAI Conference on Artificial Intelligence (2018).
Caldwell, D.J., Ojemann, J.G., Rao, R.P.N.: Direct electrical stimulation in electrocorticographic brain–computer interfaces: enabling technologies for input to cortex. Front. Neurosci. 13, 1–16 (2019). https://doi.org/10.3389/fnins.2019.00804
Bensmaia, S.J., Miller, L.E.: Restoring sensorimotor function through intracortical interfaces: progress and looming challenges. Nat. Rev. Neurosci. 15, 313–325 (2014). https://doi.org/10.1038/nrn3724
Delhaye, B.P., Saal, H.P., Bensmaia, S.J.: Key considerations in designing a somatosensory neuroprosthesis. J. Physiol. Paris. 110, 1–7 (2016). https://doi.org/10.1016/j.jphysparis.2016.11.001
Suminski, A.J., Tkach, D.C., Fagg, A.H., Hatsopoulos, N.G.: Incorporating feedback from multiple sensory modalities enhances brain-machine interface control. J. Neurosci. 30, 16777–16787 (2010). https://doi.org/10.1523/JNEUROSCI.3967-10.2010
Johnson, L.A., Wander, J.D., Sarma, D., et al.: Direct electrical stimulation of the somatosensory cortex in humans using electrocorticography electrodes: a qualitative and quantitative report. J. Neural Eng. 10, 036021 (2013). https://doi.org/10.1088/1741-2560/10/3/036021
Caldwell, D.J., Cronin, J.A., Wu, J., et al.: Direct stimulation of somatosensory cortex results in slower reaction times compared to peripheral touch in humans. Sci. Rep. 9, 3292 (2019). https://doi.org/10.1038/s41598-019-38619-2
Collins, K.L., Guterstam, A., Cronin, J., et al.: Ownership of an artificial limb induced by electrical brain stimulation. Proc. Natl. Acad. Sci. U. S. A. 114, 166–171 (2017). https://doi.org/10.1073/pnas.1616305114
Cronin, J.A., Wu, J., Collins, K.L., et al.: Task-specific somatosensory feedback via cortical stimulation in humans. IEEE Trans. Haptics. 9, 515–522 (2016). https://doi.org/10.1109/TOH.2016.2591952
Hiremath, S.V., Tyler-Kabara, E.C., Wheeler, J.J., et al.: Human perception of electrical stimulation on the surface of somatosensory cortex. PLoS One. 12, e0176020 (2017). https://doi.org/10.1371/journal.pone.0176020
Kramer, D.R., Lee, M.B., Barbaro, M., et al.: Mapping of primary somatosensory cortex of the hand area using a high-density electrocorticography grid for closed-loop brain computer interface. J. Neural Eng. (2020). https://doi.org/10.1088/1741-2552/ab7c8e
Lee, B., Kramer, D., Armenta Salas, M., et al.: Engineering artificial somatosensation through cortical stimulation in humans. Front. Syst. Neurosci. 12, 1–11 (2018). https://doi.org/10.3389/fnsys.2018.00024
Ramirez-Zamora, A., Giordano, J., Gunduz, A., et al.: Proceedings of the seventh annual deep brain stimulation think tank: advances in neurophysiology, adaptive DBS, virtual reality, neuroethics and technology. Front. Hum. Neurosci. 14, 54 (2020). https://doi.org/10.3389/FNHUM.2020.00054
Tinkhauser, G., Pogosyan, A., Debove, I., et al.: Directional local field potentials: a tool to optimize deep brain stimulation. Mov. Disord. 33, 159–164 (2018). https://doi.org/10.1002/mds.27215
Metman, L.V., Vesper, J., Mir, P., et al.: Directional versus omnidirectional deep brain stimulation: results of a multi-center prospective blinded crossover study (1375). Neurology. 94, 42 (2020)
Connolly, A.T., Vetter, R.J., Hetke, J.F., et al.: A novel lead design for modulation and sensing of deep brain structures. IEEE Trans. Biomed. Eng. 63, 148–157 (2016). https://doi.org/10.1109/TBME.2015.2492921
Butson, C.R., Cooper, S.E., Henderson, J.M., McIntyre, C.C.: Patient-specific analysis of the volume of tissue activated during deep brain stimulation. NeuroImage. 34, 661–670 (2007). https://doi.org/10.1016/j.neuroimage.2006.09.034
Peña, E., Zhang, S., Patriat, R., et al.: Multi-objective particle swarm optimization for postoperative deep brain stimulation targeting of subthalamic nucleus pathways. J. Neural Eng. 15, 066020 (2018). https://doi.org/10.1088/1741-2552/aae12f
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Caldwell, D.J., Herron, J.A., Ko, A.L., Ojemann, J.G. (2022). Motor BMIs Have Entered the Clinical Realm. In: Thakor, N.V. (eds) Handbook of Neuroengineering. Springer, Singapore. https://doi.org/10.1007/978-981-15-2848-4_108-1
Download citation
DOI: https://doi.org/10.1007/978-981-15-2848-4_108-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-2848-4
Online ISBN: 978-981-15-2848-4
eBook Packages: Springer Reference EngineeringReference Module Computer Science and Engineering