Abstract
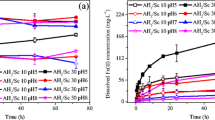
Scorodite (FeAsO4·2H2O) is suitable mineral carrier for immobilization of arsenic-rich wastes. Its stability is, however, highly pH dependent (typically at 4 ≤ pH ≤ 7) and satisfactory only under oxic disposal conditions. In this work an encapsulation process using mineralized gels of aluminum hydroxy-oxides is developed to enhance the stability of scorodite under wider pH and redox potential range conditions. The encapsulation involves blending and ageing of synthetic scorodite produced by McGill’s atmospheric scorodite process with aluminum hydroxyl gels derived from controlled hydrolysis of aluminum salts. The amorphous hydrolyzed Al-gel encapsulates the scorodite particles, which upon short-term aging transforms into crystalline Al(OH)3/AlOOH mineral phases providing a robust protection microscopic barrier. Long-term stability testing demonstrates the encapsulation system to be highly effective in suppressing arsenic release under either oxic or anoxic (100 mV < Eh < 600 mV) potential and neutral-alkaline pH (7 ≤ pH ≤ 9) ranges.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Riveros PA, Dutrizac JE, Spencer P (2001) Arsenic disposal practices in the metallurgical industry. Can Metall Q 40:395–420
Krause E, Ettel VA (1989) Solubilities and stabilities of ferric arsenate compounds. Hydrometallurgy 22(3):311–337
Langmuir D, Mahoney J, Rowson J (2006) Solubility products of amorphous ferric arsenate and crystalline scorodite (FeAsO4·2H2O) and their application to arsenic behavior in buried mine tailings. Geochim Cosmochim Acta 70(12):2942–2956
Bluteau MC, Demopoulos GP (2007) The incongruent dissolution of scorodite—solubility, kinitics and mechanism. Hydrometallurgy 87(3–4):163–177
Swash PM, Monhemius AJ (1994) Hydrothermal precipitation from aqueous solutions containing iron(III), arsenate and sulphate. Hydrometallurgy ’94. Springer, Dordrecht, pp 177–190
Gomez MA, Becze L, Cutler JN, Demopoulos GP (2011) Hydrothermal reaction chemistry and characterization of ferric arsenate phases precipitated from Fe2(SO4)3-As2O5-H2SO4 solutions. Hydrometallurgy 107(3–4):74–90
Droppert DJ, Demopoulos GP, Harris GB (1996) Ambient Pressure Production of Crystalline Scorodite from Arsenic-Rich Metallurgical Effluent Solutions. In: Warren GW (ed) EPD Congress 1996. TMS, Warrendale, PA, pp 227–239
Filippou D, Demopoulos GP (1997) Arsenic immobilization by controlled scorodite precipitation. JOM 49(12):52–55
Singhania S, Wang Q, Filippou D, Demopoulos GP (2005) Temperature and seeding effects on the precipitation of scorodite from sulfate solutions under atmospheric-pressure conditions. Metall Mater Trans B 36(3):327–333
Demopoulos GP, Lagno F, Wang Q, Singhania S (2003) The Atmospheric Scorodite Process. In: Riveros PA, Dixon D, Dreisinger DB, Menacho J (eds) Copper 2003- Hydrometallurgy of Copper. CIM, Montreal, QC, pp 597–616
Demopoulos GP (2005) Preparation and stability of scorodite. In: Reddy RG, Ramachandran V (eds) Arsenic metallurgy. TMS, Warrendale, PA, pp 25–50
Lagno F, Garcia I, Ledesma C, Reghezza A, Demopoulos GP, Becze L, Gomez M, Katsarou L (2009) Fixation of arsenic and antimony from copper smelter flue dust and electrorefinery bleed-off as crystalline scorodite. Paper presented at Proceedings of HYDROCOPPER 2009 conference, Antofagasta, Chile, 13–15 May 2009
Fujita T, Taguchi R, Abumiya M, Matsumoto M, Shibata E, Nakamura T (2008) Novel atmospheric scorodite synthesis by oxidation of ferrous sulfate solution. Part I. Hydrometallurgy 90(2–4):92–102
Fujita T, Taguchi R, Abumiya M, Matsumoto M, Shibata E, Nakamura T (2008) Novel atmospheric scorodite synthesis by oxidation of ferrous sulfate solution. Part II. Effect of temperature and air. Hydrometallurgy 9(2–4):85–91
Kubo H, Abumiya M, Matsumoto M (2010) Dowa mining scorodite process—application to copper hydrometallurgy. In: Proceedings of copper 2010, vol 7. Hamburg, Germany, pp 2947–2958
Demopoulos GP (2014) Arsenic immobilization research advances: past, present and future. In: Barnes A et al (ed), Proceedings of arsenic metallurgy & the environment symposium, COM2014. MetSoc of CIM, Vancouver, BC, p 13
Rochette EA, Li GC, Fendorf SE (1998) Stability of arsenate minerals in soil under biotically generated reducing conditions. Soil Sci Soc Am J 62(6):1530–1537
McCreadie H, Jambor JL, Blowes DW, Hiller D (1998) Geochemical behaviour of autoclave-produced ferric arsenates and jarosite in a gold-mine tailings impoundment. In: Petruk W (ed) Waste characterization and treatment. Society for Mining Metallurgy and Exploration, Littleton, CO, pp 61–78
Verdugo C, Lagos G, Becze L, Gómez M, Demopoulos GP (2012) Study of arsenic release of atmospheric scorodite in reductive environments. In: Casas JM et al (eds) Proceedings of HydroProcess 2012. Gecamin, Santiago, Chile, pp 86–94
Dutre V, Vandecasteele C (1998) Immobilization of arsenic in waste solidified using cement and lime. Environ Sci Technol 32:2782–2787
Lagno F, Rocha SDF, Chryssoulis S, Demopoulos GP (2010) Scorodite encapsulation by controlled deposition of aluminum phosphate coatings. J Hazard Mater 181:526–534
Rocha SDF, Katsarou L, Demopoulos GP (2012) Reduction of arsenic releasing in aqueous environment by scorodite encapsulation. In: Jia CQ et al (eds) Sustainable Issues in mineral and metal extraction (WALSIM II). MetSoc of CIM, Niagara Falls, ON, pp 215–224
Leetmaa K, Guo FG, Becze L, Gomez MA, Demopoulos GP (2016) Stabilization of iron arsenate solids by encapsulation with aluminum hydroxyl gels. J Chem Technol Biotechnol 91:408–415
Leetmaa K, Gomez MA, Becze L, Guo FG, Demopoulos GP (2014) Comparative molecular characterization of aluminum hydroxyl-gels derived from chloride and sulphate salts. J Chem Technol Biotechnol 89:206–213
Dabekaussen R, Droppert D, Demopoulos GP (2001) Ambient pressure hydrometallurgical conversion of arsenic trioxide to crystalline scorodite. CIM Bull 94:116–122
Wang QK, Demopoulos GP, Harris GB (2000) Arsenic fixation in metallurgical plant effluents in the form of crystalline scorodite via a non-autoclave oxidation-precipitation process. In: Young C (ed) Minor Elements 2000, Processing and environmental aspects of As, Sb, Se, Te, and Bi. Society for Mining, Metallurgy, and Exploration, Littleton, CO, pp 225–237
Acknowledgements
The support of NSERC and industrial partners is gratefully acknowledged. Similarly, the contributions of several past members of our group in this work is proudly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 The Minerals, Metals & Materials Society
About this paper
Cite this paper
Guo, F., Demopoulos, G.P. (2018). Development of an Encapsulation Process to Extend the Stability of Scorodite Under Wider pH and Redox Potential Range Conditions. In: Davis, B., et al. Extraction 2018. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-319-95022-8_115
Download citation
DOI: https://doi.org/10.1007/978-3-319-95022-8_115
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-95021-1
Online ISBN: 978-3-319-95022-8
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)