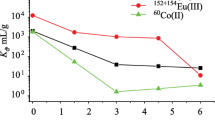
Abstract
Ion exchange technique is one of the widely applied conventional treatment options in nuclear industry, which is used to reduce the volume of aqueous radioactive waste streams, to separate isotopes and in decontamination operations. After being exhausted, spent exchangers are considered as radioactive waste and consequently they must be safely managed to minimize their environmental impacts. Exchangers’ selection and operating conditions optimization are affected by the waste and exchanger characteristics. In this chapter, the life cycle of the exchange materials will be traced starting from material preparation to disposal. Within this context, sources of radioactive wastes and their important characteristics will be introduced. Exchanger preparation and characterization methods will be overviewed. Chemical, mechanical, and radiological design criteria that affect performances of the process will be summarized. Treatment and immobilization options of spent ion exchangers will be overviewed and methods to assess immobilization sustainability will be highlighted.
Similar content being viewed by others
References
IAEA (1995) The principles of radioactive waste management. Safety series no. 111-F. International Atomic Energy Agency, Vienna
Abdel Rahman RO (2012) Planning and implementation of radioactive waste management system. In: Abdel Rahman RO (ed) Radioactive waste. InTech, Rijeka, pp 3–18. http://www.intechopen.com/books/radioactive-waste/planning-and-implementation-of-radioactive-waste-management-system
Abdel Rahman RO, El Kamash AM, Zaki AA, El Sourougy MR (2005) Disposal: a last step towards an integrated waste management system in Egypt. In: International conference on the safety of radioactive waste disposal, Tokyo, IAEA-CN-135/81, pp 317–324
IAEA (2003) Radioactive waste management glossary, 2003rd edn. International Atomic Energy Agency, Vienna
Abdel Rahman RO, Rakhimov RZ, Rakhimova NR, Ojovan MI (2014) Cementitious materials for nuclear waste immobilisation. Wiely, New York, ISBN 9781118512005
Abdel Rahman RO, Guskov A, Kozak M, Hung YT (2016) Recent evaluation of early radioactive disposal practice. In: Wang LK, Wang MS, Hung YT, Shammas NK (eds) Handbook of environmental engineering, vol 17, Natural resources and control processes. Springer,Switzerland, pp 371–400. https://doi.org/10.1007/978-3-319-26800-2_8
IAEA (2009) Classification of radioactive waste: general safety guide GSG-1. IAEA, Vienna
IAEA (2001) Handling and processing of radioactive waste from nuclear applications. TRS, 402. International Atomic Energy Agency, Vienna
Nasef MM, Ujang Z (2012) Introduction to ion exchange processes. In: Luqman IM (ed) Ion exchange technology: theory and principals. Springer, Netherlands
Metwally SS, Ahmed IM, Rizk HE (2017) Modification of hydroxyapatite for removal of cesium and strontium ions from aqueous solution. J Alloys Compd 709:438–444
Varshney D (2003) Synthetic ion exchange materials and their analytical applications: past, present and future. In: Sajgalík P, Drábik M, Varga S (eds) Solid state phenomena, vols 90–91. pp 445–450. Solid State Chemistry V, Trans Tech Publication LTD, 10.4028/www.scientific.net/SSP.90-91.445
Lehto J, Brodkin L, Harjula R, Tusa E (1999) Separation of radioactive strontium from alkaline nuclear waste solutions with the highly effective ion exchanger SrTreat. Nucl Technol 127:81–87
Clearfield A, Bortun LN, Bortun AI (2000) Alkali metal ion exchange by the framework titanium silicate M2Ti2O3SiO4·nH2O (M=H, Na). React Funct Polym 43:85–95
Clearfield A (2000) Inorganic ion exchangers, past, present, and future. Solvent Extr Ion Exch 18:655–678
Da Silva MLCP, Da Silva GLJP, Filho DNV (2002) Hydrous Tantalum Phosphates for Ion Exchange Purposes: A Systematic Study. Mater Res 5:71–75
Metwally SS, Ghaly M, El-Sherief EA (2017) Physicochemical properties of synthetic nano birnessite and its enhanced scavenging of Co2+ and Sr2+ ions from aqueous solutions. Mater Chem Phys 193:63–72
Ghaly M, El-Dars FMSE, Hegazy MM, Abdel Rahman RO (2016) Evaluation of synthetic Birnessite utilization as a sorbent for cobalt and strontium removal from aqueous solution. Chem Eng J 284:1373–1385
Harjula R, Lehto J, Paajanen A, Brodkin L (2001) Removal of Radioactive Cesium from Nuclear Waste Solutions with the Transition Metal Hexacyanoferrate Ion Exchanger CsTreat Nucl Sci Eng 137:206–214
El-Kamash AM (2008) Evaluation of zeolite A for the sorptive removal of Cs+ and Sr2+ ions from aqueous solutions using batch and fixed bed column operations. J Hazard Mater 151:432–445
Abdel Rahman RO, Ibrahim HA, Abdel Monem NM (2009) Long-term performance of zeolite Na A-X blend as backfill material in near surface disposal vault. Chem Eng J 149:143–152
Abdel Rahman RO, Ibrahim HA, Hanafy M, Abdel Monem NM (2010) Assessment of synthetic zeolite NaA-X as sorbing barrier for strontium in a radioactive disposal facility. Chem Eng J 157:100–112
El-Naggar MR, El-Kamash AM, El-Dessouky MI, Ghonaim AK (2008) Two-step method for preparation of NaA-X zeolite blend from fly ash for removal of cesium ions. J Hazard Mater 154:963–972
Abdel Rahman RO, Abdel Moamen OA, Hanafy M, Abdel Monem NM (2012) Preliminary investigation of zinc transport through zeolite-X barrier: linear isotherm assumption. Chem Eng J 185–186:61–70
Abdel Moamen OA, Ismail IM, Abdel Monem NM, Abdel Rahman RO (2015) Factorial design analysis for optimizing the removal of cesium and strontium ions on synthetic nano-sized zeolites. J Taiwan Inst Chem Eng 55:133–144
El-Kamash AM, Zaki AA, Abed El Geleel M (2005) Modeling batch kinetics and thermodynamics of zinc and cadmium ions removal from waste solution using synthetic zeolite A. J Hazard Mater 127:211–220
Abd El-Rahman KM, El-Kamash AM, El-Sourougy MR, Abdel Moniem NM (2006) Thermodynamic modeling for the removal of Cs+, Sr2+, Ca2+, and Mg2+ ions from aqueous waste solution using zeolite A. J Radioanal Nucl Chem 268(2):221–230
Sharma P, Sharma M, Tomar R (2013) Na HEU zeolite synthesis for the removal of Th(IV) and Eu(III) from aqueous waste by batch process. J Taiwan Inst Chem Eng 44:480–488
Flainigen EM (1991) In: van Bekkum H, Flainigen EM, Jansen JC (eds) Introduction to zeolite science and practice, vol 13. Elsevier, Amsterdam
Abdel Rahman RO, Ojovan MI(2017) Ch 15, Application of nano-materials in radioactive waste management. In: Zhang TC, Gurjar BR, Govil JN (eds) Environmental science and engineering, vol 10, Industrial processes & nanotechnology. Studium Press, LLC, pp 361–378, ISBN 10: 1-62699-098-0
Sahu BB, Mishra HK, Parida K (2000) Cation exchange and sorption properties of TIN(IV) phosphate. J Colloid Interface Sci 225:511–519
Jiang P, Pan B, Pan B, Zhang W, Zhang Q (2008) A comparative study on lead sorption by amorphous and crystalline zirconium phosphates. Colloids Surf A Physicochem Eng Asp 322:108–112
Michel C, Barré Y, De Windt L, de Dieuleveult C, Brackx E, Grandjean A (2017) Ion exchange and structural properties of a new cyanoferrate mesoporous silica material for Cs removal from natural saline waters. J Environ Chem Eng 5:810–817
El-Kamash AM, El-Gammal B, El-Sayed AA (2007) Preparation and evaluation of cerium(IV) tungstate powder as inorganic exchanger in sorption of cobalt and europium ions from aqueous solutions. J Hazard Mater 141:719
Abdel Rahman RO, Ibrahium HA, Hung YT (2011) Liquid radioactive wastes treatment: a review. Water 3:551–565
Cundy CS, Cox PA (2003) The hydrothermal synthesis of zeolites: history and development from the earliest days to the present time. Chem Rev 103:663–701
Byrappa K, Yoshimura M (2001) Handbook of hydrothermal technology. Noyes Publications, Park Ridge
Ames LL Jr (1963) Synthesis of a clinoptilolite-like zeolite. Am Mineral 48:1374–1381
Sinha PK, Amalraj RV, Krishnasamy V (1993) Flocculation studies on freshly precipitated copper ferrocyanide for the removal of cesium from radioactive liquid waste. Waste Manag 13:341–350
Metwally E, Abd El-Rahman RO, Ayoub RR (2007) Modeling batch kinetics of cesium, cobalt and strontium ions adsorption from aqueous solutions using hydrous titanium oxide. Radiochim Acta 95:409–416
Gasser MS, Mekhamer HS, Abdel Rahman RO (2016) Optimization of the utilization of Mg/Fe hydrotalcite like compounds in the removal of Sr(II) from aqueous solution. J Environ Chem Eng 4:4619–4630
Gasser MS, El Sherif E, Abdel Rahman RO (2017) Modification of Mg-Fe hydrotalcite using Cyanex 272 for lanthanides separation. Chem Eng J 316:758–769
Lee CG, Alvarez PJJ, Nam A, Park SJ, Do T, Choi US, Lee SH (2017) Arsenic(V) removal using an amine-doped acrylic ion exchange fiber: kinetic, equilibrium, and regeneration studies. J Hazard Mater 325:223–229
Young GK, Ung SC, Tai YK, Dong JA, Yong JC (2002) FT-IR and isotherm study on anion adsorption onto novel chelating fiber. Macromol Rapid Commun 23:535–539
Bajpai SK, Susamma J (2005) Superabsorbent hydrogels for removal of divalent toxic ions. Part I: synthesis and swelling characterization. React Funct Polym 62:271–283
Metwally SS, El-gammal B, Aly HF, Abo-el-enein SA (2011) Removal and separation of some radionuclides by poly-acrylamide based Ce(IV) phosphate from radioactive waste solutions. Sep Sci Technol 46:1808–1821
El-Gammal B, Metwally SS, Aly HF, Abo-El-Enein SA (2012) Verification of double-shell model for sorption of cesium, cobalt, and europium ions on poly-acrylonitrile-based Ce(IV) phosphate from aqueous solutions. Desalin Water Treat 46:1–15
Metwally SS, Ayoub RR, Aly HF (2013) Amidoximation of cyano group for chelating ion exchange of some heavy metal ions from wastewater. Sep Sci Technol 48:1829–1839
Mohamed WR, Metwally SS, Ibrahim HA, El-Sherief EA, Mekhamer HS, Moustafa IMI, Mabrouk EM (2017) Impregnation of task-specific ionic liquid into a solid support for removal of neodymium and gadolinium ions from aqueous solution. J Mol Liq 236:9–17
Naser AA, Sharaf El-deen GE, Bhran AA, Metwally SS, El-Kamash AM (2015) Elaboration of impregnated composite for sorption of europium and neodymium ions from aqueous solutions. J Ind Eng Chem 32:264–272
Kim YK, Kim Y, Kim S, Harbottle D, Lee JW (2017) Solvent-assisted synthesis of potassium copper hexacyanoferrate embedded 3D-interconnected porous hydrogel for highly selective and rapid cesium ion removal. J Environ Chem Eng 5:975–986
Borai EH, Hamed MG, El-Kamash AM, Siyam T, El-Sayed GO (2015) Template polymerization synthesis of hydrogel and silica composite for sorption of some rare earth elements. J Colloid Interface Sci 456:228–240
IAEA (2002) Application of ion exchange process for treatment of radioactive waste. Technical report series 408. International Atomic Energy Agency, Vienna
Dounreay Site Restoration Ltd (2009) A review of national and international best practice on waste minimisation, DEC(09)P175, Mar 2009, pp 1–60
Chapter 1 ref 0.8 Resin and FILTER handbook primers and product information
Abdel Rahman RO (2011) Preliminary evaluation of the technical feasibility of using different soils in waste disposal cover system. Environ Prog Sustain Energy 30(1):19–28
Tariqul Islam MD, Dafader NC, Poddar P, Khan NMDS, Chowdhury AMS (2016) Studies on swelling and absorption properties of the γ– irradiated polyvinyl alcohol (PVA)/kappa-carrageenan blend hydrogels. Adv Chem Eng 6:2. https://doi.org/10.4172/2090-4568.1000153
IAEA (1986) Ion exchange technology. tecdoc 365. International Atomic Energy Agency, Vienna
Simon GP (1991) Ion exchange training manual. Springer science, Netherlands
Gauthier MA, Luo J, Calvet D, Ni C, Zhu XX, Garon M, Buschmann MD (2004) Degree of crosslinking and mechanical properties of crosslinked poly(vinyl alcohol) beads for use in solid-phase organic synthesis. Polymer 45:8201–8210
Richardson JF, Harker JH, Backhurst JB(2002) Flow of fluids through granular beds and packed columns. In: Harker JH, Backhurst JR, Richardson JF(eds) Chemical engineering, 5th edn, vol 2. pp 191–236, Butterworth Heinemann, Oxford
Westinghouse Technology Systems Manual. Chemical and volume control system, USNRC HRTD, Rev 1208
Power plants energized by Lanxess, Lewatit. Ion exchange resin for the power industry: focus on internal water circuits
Fredric S, Marsh K, Pillay KS(1993) Effects of ionizing radiation on modern ion exchange materials, LA-12655-MS, Oct 1993-Los Alamos report
Traboulsi A, Labed V, Dauvois V, Dupuy N, Rebufa C (2013) Gamma radiation effect on gas production in anion exchange resins. Nucl Inst Methods Phy Res B 312:7–14
Hiser MA, Pulvirenti AL, Al-Sheikhly M(2013) Monitoring degradation of phenolic resin-based neutron absorbers in spent nuclear fuel pools, June 2013. U.S. Nuclear Regulatory Commission
Gangwer TE, Goldstein M, Pillay KKS (1977) Radiation effects on ion exchange materials, Nov 1977, under contract NO. EY-76-C-02-0016 US DOE, BNL 50781
Abdel Rahman RO, Kozak MW, Hung YT (2014) Radioactive pollution and control. In: Hung YT, Wang LK, Shammas NK (eds) Handbook of environment and waste management. World Scientific, Singapore, pp 949–1027. https://doi.org/10.1142/9789814449175_0016
Drace Z, Mele I, Ojovan MI, Abdel Rahman RO (2012) An overview of research activities on cementitious materials for radioactive waste management. Mater Res Soc Symp Proc 1475:253–264
Luca V, Bianchi HL, Manzini AC (2012) Cation immobilization in pyrolyzed simulated spent ion exchange resins. J Nucl Mater 424:1–11
Abdel Rahman RO, Ojovan MI (2016) Recent trends in the evaluation of cementitious material in radioactive waste disposal. In: Wang LK, Wang MS, Hung YT, Shammas NK (eds) Handbook of environmental engineering, vol 17, Natural resources and control processes. Springer, pp 401–448. https://doi.org/10.1007/978-3-319-26800-2_9. http://link.springer.com/chapter/10.1007/978-3-319-26800-2_9/fulltext.html
Abdel Rahman RO, El-Kamash AM, Zaki AA (2007) Modeling the long term leaching behavior of 137Cs, 60Co, and152,154Eu radionuclides from cement- clay matrices. Hazard Mater 145:372–380
Abdel Rahman RO, Zaki AA (2011) Comparative study of leaching conceptual models: Cs leaching from different ILW cement based matrices. Chem Eng J 173:722–736
Abdel Rahman RO, Zein DH, Abo Shadi H (2014) Cesium binding and leaching from single and binary contaminant cement-bentonite matrices. Chem Eng J 245:276–287
Abdel Rahman RO, Ojovan MI (2014) Leaching tests and modelling of cementitious wasteforms corrosion. Innov Corros Mater Sci 4(2):90–95. https://doi.org/10.2174/2352094904666141126221626
Ojovan MI, Varlackov GA, Golubev ZI, Burlak ON (2011) Long-term field and laboratory leaching tests of cemented radioactive wastes. J Hazard Mater 187:296–302
Mattigod SV, Wellman DM, Bovaird CC, Parker KE, Recknagle KP, Clayton L, Wood MI (2012) Diffusion of radionuclides in concrete and soil. In: Abdel Rahman RO (ed) Ratioactive waste. INTECH, pp 331–350. http://www.intechopen.com/books/radioactive-waste
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this entry
Cite this entry
Rahman, R.O.A., Metwally, S.S., El-Kamash, A.M. (2018). Life Cycle of Ion Exchangers in Nuclear Industry: Application and Management of Spent Exchangers. In: Martínez, L., Kharissova, O., Kharisov, B. (eds) Handbook of Ecomaterials. Springer, Cham. https://doi.org/10.1007/978-3-319-48281-1_108-1
Download citation
DOI: https://doi.org/10.1007/978-3-319-48281-1_108-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48281-1
Online ISBN: 978-3-319-48281-1
eBook Packages: Springer Reference EngineeringReference Module Computer Science and Engineering