Abstract
Mesenchymal stromal/stem cell (MSC) therapy with the goal of restoring salivary function following irradiation injury or in Sjögren’s syndrome (SS) has made significant advances within the past 5 years. The majority of studies used MSCs obtained from the bone marrow or adipose tissue, but MSCs isolated from the salivary gland, dental pulp, and umbilical cord also demonstrated a therapeutic efficacy in reestablishing salivary function. Based on the amount of stimulated saliva secretion as a functional quantitative measure, irradiated mice/rats that received MSC therapy restored their salivary flow rate (SFR) to 60–90 % of normal age-matched animals, while SFR of irradiated animals without treatment remained at 35–50 % of secretory function. Thus, there was 25–40 % therapeutic improvement in animals receiving MSC therapy versus those that did not. This would be clinically significant because patients with severe salivary hypofunction (dry mouth) due to head and neck irradiation have no improvement in SFR, if left untreated. In the SS-like disease mouse model, MSC therapy restored SFR 80–100 % when treatment was given at an initial phase of SS-like disease, while its effectiveness decreased to 50–60 % when given at an advanced stage of disease. In SS patients, MSC therapy improved SFR by 40–50 %. When tested in the rodent model, MSC therapy was successful in restoring/maintaining the gland normal weights and histology (acinar cells, blood vessels) and upregulated the expression of genes favorable for salivary gland development and regeneration while downregulating inflammation and cell apoptosis, promising positive effects of MSC therapy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aframian DJ, Palmon A. Current status of the development of an artificial salivary gland. Tissue Eng Part B Rev. 2008;14:187–98.
Alajbeg I, Falcao DP, Tran SD, Martin-Granizo R, Lafaurie GI, Matranga D, Pejda S, Vuletic L, Mantilla R, Leal SC, Bezerra AC, Menard HA, Kimoto S, Pan S, Maniegas L, Krushinski CA, Melilli D, Campisi G, Paderni C, Mendoza GR, Yepes JF, Lindh L, Koray M, Mumcu G, Elad S, Zeevi I, Barrios BC, Lopez Sanchez RM, Lassauzay C, Fromentin O, Beiski BZ, Strietzel FP, Konttinen YT, Wolff A, Zunt SL. Intraoral electrostimulator for xerostomia relief: a long-term, multicenter, open-label, uncontrolled, clinical trial. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113:773–81.
Baum BJ, Kok M, Tran SD, Yamano S. The impact of gene therapy on dentistry: a revisiting after six years. J Am Dent Assoc. 2002;133:35–44.
Baum BJ, Tran SD. Synergy between genetic and tissue engineering: creating an artificial salivary gland. Periodontol 2000. 2006;2000(41):218–23.
Bhide SA, Miah AB, Harrington KJ, Newbold KL, Nutting CM. Radiation-induced xerostomia: pathophysiology, prevention and treatment. Clin Oncol (R Coll Radiol). 2009;21:737–44.
Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy. 2013;15:641–8.
Coppes R, Stokman M. Stem cells and the repair of radiation-induced salivary gland damage. Oral Dis. 2011;17:143–53.
Fang D, Hu S, Liu Y, Quan VH, Seuntjens J, Tran SD. Identification of the active components in bone marrow soup: a mitigator against irradiation-injury to salivary glands. Sci Rep. 2015;5:16017.
Feng J, van der Zwaag M, Stokman MA, van Os R, Coppes RP. Isolation and characterization of human salivary gland cells for stem cell transplantation to reduce radiation-induced hyposalivation. Radiother Oncol. 2009;92:466–71.
Fox PC. Acquired salivary dysfunction. drugs and radiation. Ann N Y Acad Sci. 1998;842:132–7.
Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ, Krause DS, Keating A. Clarification of the nomenclature for MSC: the international society for cellular therapy position statement. Cytotherapy. 2005;7:393–5.
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300.
Jensen DH, Oliveri RS, Trojahn Kolle SF, Fischer-Nielsen A, Specht L, Bardow A, Buchwald C. Mesenchymal stem cell therapy for salivary gland dysfunction and xerostomia: a systematic review of preclinical studies. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117:335–42. e1
Jeong J, Baek H, Kim YJ, Choi Y, Lee H, Lee E, Kim ES, Hah JH, Kwon TK, Choi IJ, Kwon H. Human salivary gland stem cells ameliorate hyposalivation of radiation-damaged rat salivary glands. Exp Mol Med. 2013;45:e58.
Kagami H. The potential use of cell-based therapies in the treatment of oral diseases. Oral Dis. 2015;21:545–9.
Khalili S, Faustman DL, Liu Y, Sumita Y, Blank D, Peterson A, Kodama S, Tran SD. Treatment for salivary gland hypofunction at both initial and advanced stages of Sjogren-like disease: a comparative study of bone marrow therapy versus spleen cell therapy with a 1-year monitoring period. Cytotherapy. 2014;16:412–23.
Khalili S, Liu Y, Kornete M, Roescher N, Kodama S, Peterson A, Piccirillo CA, Tran SD. Mesenchymal stromal cells improve salivary function and reduce lymphocytic infiltrates in mice with Sjogren’s-like disease. PLoS One. 2012;7:e38615.
Khalili S, Liu Y, Sumita Y, Maria OM, Blank D, Key S, Mezey E, Tran SD. Bone marrow cells are a source of undifferentiated cells to prevent Sjogren’s syndrome and to preserve salivary glands function in the non-obese diabetic mice. Int J Biochem Cell Biol. 2010;42:1893–9.
Kodama S, Kuhtreiber W, Fujimura S, Dale EA, Faustman DL. Islet regeneration during the reversal of autoimmune diabetes in NOD mice. Science. 2003;302:1223–7.
Kojima T, Kanemaru S, Hirano S, Tateya I, Ohno S, Nakamura T, Ito J. Regeneration of radiation damaged salivary glands with adipose-derived stromal cells. Laryngoscope. 2011;121:1864–9.
Lee J, Park S, Roh S. Transdifferentiation of mouse adipose-derived stromal cells into acinar cells of the submandibular gland using a co-culture system. Exp Cell Res. 2015;334:160–72.
Li Z, Wang Y, Xing H, Wang Z, Hu H, An R, Xu H, Liu Y, Liu B. Protective efficacy of intravenous transplantation of adipose-derived stem cells for the prevention of radiation-induced salivary gland damage. Arch Oral Biol. 2015;60:1488–96.
Lim JY, Ra JC, Shin IS, Jang YH, An HY, Choi JS, Kim WC, Kim YM. Systemic transplantation of human adipose tissue-derived mesenchymal stem cells for the regeneration of irradiation-induced salivary gland damage. PLoS One. 2013a;8:e71167.
Lim JY, Yi T, Choi JS, Jang YH, Lee S, Kim HJ, Song SU, Kim YM. Intraglandular transplantation of bone marrow-derived clonal mesenchymal stem cells for amelioration of post-irradiation salivary gland damage. Oral Oncol. 2013b;49:136–43.
Lin CY, Chang FH, Chen CY, Huang CY, Hu FC, Huang WK, Ju SS, Chen MH. Cell therapy for salivary gland regeneration. J Dent Res. 2011;90:341–6.
Lombaert IM, Brunsting JF, Wierenga PK, Faber H, Stokman MA, Kok T, Visser WH, Kampinga HH, de Haan G, Coppes RP. Rescue of salivary gland function after stem cell transplantation in irradiated glands. PLoS One. 2008;3:e2063.
Maimets M, Bron R, de Haan G, van Os R, Coppes RP. Similar ex vivo expansion and post-irradiation regenerative potential of juvenile and aged salivary gland stem cells. Radiother Oncol. 2015;16:443–8.
Maria OM, Maria O, Liu Y, Komarova SV, Tran SD. Matrigel improves functional properties of human submandibular salivary gland cell line. Int J Biochem Cell Biol. 2011a;43:622–31.
Maria OM, Tran SD. Human mesenchymal stem cells cultured with salivary gland biopsies adopt an epithelial phenotype. Stem Cells Dev. 2011;20:959–67.
Maria OM, Zeitouni A, Gologan O, Tran SD. Matrigel improves functional properties of primary human salivary gland cells. Tissue Eng Part A. 2011b;17:1229–38.
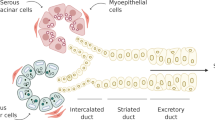
Melvin JE, Yule D, Shuttleworth T, Begenisich T. Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Annu Rev Physiol. 2005;67:445–69.
Misuno K, Tran SD, Khalili S, Huang J, Liu Y, Hu S. Quantitative analysis of protein and gene expression in salivary glands of Sjogren’s-like disease NOD mice treated by bone marrow soup. PLoS One. 2014;9:e87158.
Nanduri LS, Baanstra M, Faber H, Rocchi C, Zwart E, de Haan G, van Os R, Coppes RP. Purification and ex vivo expansion of fully functional salivary gland stem cells. Stem Cell Reports. 2014;3:957–64.
Nanduri LS, Lombaert IM, van der Zwaag M, Faber H, Brunsting JF, van Os RP, Coppes RP. Salisphere derived c-Kit+ cell transplantation restores tissue homeostasis in irradiated salivary gland. Radiother Oncol. 2013;108:458–63.
Neumann Y, David R, Stiubea-Cohen R, Orbach Y, Aframian DJ, Palmon A. Long-term cryopreservation model of rat salivary gland stem cells for future therapy in irradiated head and neck cancer patients. Tissue Eng Part C Methods. 2012;18:710–8.
Nguyen TT, Mui B, Mehrabzadeh M, Chea Y, Chaudhry Z, Chaudhry K, Tran SD. Regeneration of tissues of the oral complex: current clinical trends and research advances. J Can Dent Assoc. 2013;79:d1.
Palmon A, David R, Neumann Y, Stiubea-Cohen R, Krief G, Aframian DJ. High-efficiency immunomagnetic isolation of solid tissue-originated integrin-expressing adult stem cells. Methods. 2012;56:305–9.
Park YJ, Koh J, Gauna AE, Chen S, Cha S. Identification of regulatory factors for mesenchymal stem cell-derived salivary epithelial cells in a co-culture system. PLoS One. 2014;9:e112158.
Patel VN, Hoffman MP. Salivary gland development: a template for regeneration. Semin Cell Dev Biol. 2014;25-26:52–60.
Pringle S, Maimets M, van der Zwaag M, Stokman MA, van Gosliga D, Zwart E, Witjes MJ, de Haan G, van Os R, Coppes RP. Human salivary gland stem cells functionally restore radiation damaged salivary glands. Stem Cells. 2016;34:640–52.
Pringle S, van Os R, Coppes RP. Concise review: adult salivary gland stem cells and a potential therapy for xerostomia. Stem Cells. 2013;31:613–9.
Souza LN, Faria DR, Dutra WO, Gomes CC, Gomez RS. Microchimerism in labial salivary glands of hematopoietic stem cell transplanted patients. Oral Dis. 2011;17:484–8.
Sumita Y, Liu Y, Khalili S, Maria OM, Xia D, Key S, Cotrim AP, Mezey E, Tran SD. Bone marrow-derived cells rescue salivary gland function in mice with head and neck irradiation. Int J Biochem Cell Biol. 2011;43:80–7.
Tran SD, Kodama S, Lodde BM, Szalayova I, Key S, Khalili S, Faustman DL, Mezey E. Reversal of Sjogren’s-like syndrome in non-obese diabetic mice. Ann Rheum Dis. 2007;66:812–4.
Tran SD, Liu Y, Xia D, Maria OM, Khalili S, Wang RW, Quan VH, Hu S, Seuntjens J. Paracrine effects of bone marrow soup restore organ function, regeneration, and repair in salivary glands damaged by irradiation. PLoS One. 2013;8:e61632.
Tran SD, Redman RS, Barrett AJ, Pavletic SZ, Key S, Liu Y, Carpenter A, Nguyen HM, Sumita Y, Baum BJ, Pillemer SR, Mezey E. Microchimerism in salivary glands after blood- and marrow-derived stem cell transplantation. Biol Blood Marrow Transplant. 2011a;17:429–33.
Tran SD, Sugito T, Dipasquale G, Cotrim AP, Bandyopadhyay BC, Riddle K, Mooney D, Kok MR, Chiorini JA, Baum BJ. Re-engineering primary epithelial cells from rhesus monkey parotid glands for use in developing an artificial salivary gland. Tissue Eng. 2006;12:2939–48.
Tran SD, Sumita Y, Khalili S. Bone marrow-derived cells: a potential approach for the treatment of xerostomia. Int J Biochem Cell Biol. 2011b;43:5–9.
Vissink A, Burlage FR, Spijkervet FK, Jansma J, Coppes RP. Prevention and treatment of the consequences of head and neck radiotherapy. Crit Rev Oral Biol Med. 2003a;14:213–25.
Vissink A, Jansma J, Spijkervet FK, Burlage FR, Coppes RP. Oral sequelae of head and neck radiotherapy. Crit Rev Oral Biol Med. 2003b;14:199–212.
Xiao N, Lin Y, Cao H, Sirjani D, Giaccia AJ, Koong AC, Kong CS, Diehn M, LE QT. Neurotrophic factor GDNF promotes survival of salivary stem cells. J Clin Invest. 2014;124:3364–77.
Xiong X, Shi X, Chen F. Human adipose tissue derived stem cells alleviate radiationinduced xerostomia. Int J Mol Med. 2014;34:749–55.
Xu J, Wang D, Liu D, Fan Z, Zhang H, Liu O, Ding G, Gao R, Zhang C, Ding Y, Bromberg JS, Chen W, Sun L, Wang S. Allogeneic mesenchymal stem cell treatment alleviates experimental and clinical Sjogren syndrome. Blood. 2012;120:3142–51.
Yamamura Y, Yamada H, Sakurai T, Ide F, Inoue H, Muramatsu T, Mishima K, Hamada Y, Saito I. Treatment of salivary gland hypofunction by transplantation with dental pulp cells. Arch Oral Biol. 2013;58:935–42.
Acknowledgments
We thank Professor Bruce J Baum for his invaluable advice, input, and encouragements during these past several years on the research pertaining to bone marrow cells and its cell soup in the repair of salivary glands. These studies were supported in part by the Canadian Institutes of Health Research (CIHR).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Tran, S.D., Sumita, Y., Fang, D., Hu, S. (2017). Adult Stem Cell Therapy for Salivary Glands, with a Special Emphasis on Mesenchymal Stem Cells. In: Cha, S. (eds) Salivary Gland Development and Regeneration. Springer, Cham. https://doi.org/10.1007/978-3-319-43513-8_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-43513-8_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-43511-4
Online ISBN: 978-3-319-43513-8
eBook Packages: MedicineMedicine (R0)