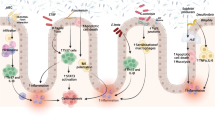
Abstract
Inflammatory bowel disease (IBD) is a set of chronic inflammatory disorders affecting the gastrointestinal tract, occurring in a relapsing-remitting fashion, and requiring lifelong treatments.
Although great advancements in the IBD research field have been made, its exact etiology remains uncertain. It has been widely acknowledged that IBD represents a multifactorial disorder resulting from a complex interplay between genetic, host immune system, and environmental factors. Emerging evidence points at diet as a crucial factor in the pathogenesis and progression of IBD. A common feature of people suffering from IBD is intestinal dysbiosis, characterized by a reduction of Firmicutes, Bacteroidetes, Actinobacteria, and other beneficial commensal species and a concomitant increase of Proteobacteria and pathobionts, all together leading to loss of intestinal homeostasis. The consumption of industrialized and processed food, rich in saturated fatty acids, typical of a Western-like type of diet, has been shown to contribute to the onset of an aberrant hyperactivated mucosal immune response to commensal bacteria and impairment of the intestinal barrier integrity, thereby triggering intestinal inflammation. Conversely, the Mediterranean diet, rich in anti-inflammatory bioactive compounds, appears to be one of the most efficient dietetic regimen able to restore the host intestinal physiology. Given IBD molecular and clinical heterogeneity, we are far from establishing a universal nutritional protocol for IBD patients; however, data demonstrate that specific dietetic protocols and GM manipulation are useful as adjuvant therapies to prevent nutritional deficiencies, promote eubiosis, alleviate inflammatory symptoms, and ameliorate the clinical progression of IBD.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Alatab S, et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(1):17–30. https://doi.org/10.1016/S2468-1253(19)30333-4.
Alfadhli AA, McDonald JW, Feagan BG. Methotrexate for induction of remission in refractory Crohn’s disease. In: The Cochrane Collaboration, editor. Cochrane database of systematic reviews. Chichester: Wiley; 2004. p. CD003459.pub2. https://doi.org/10.1002/14651858.CD003459.pub2.
Amersfoort J, Eelen G, Carmeliet P. Immunomodulation by endothelial cells – partnering up with the immune system? Nat Rev Immunol. 2022;22(9):576–88. https://doi.org/10.1038/s41577-022-00694-4.
Andersen V, et al. Fibre intake and the development of inflammatory bowel disease: a European prospective multi-centre cohort study (EPIC-IBD). J Crohn’s Colitis. 2018;12(2):129–36. https://doi.org/10.1093/ecco-jcc/jjx136.
Barberio B, Facchin S, et al. A specific microbiota signature is associated to various degrees of ulcerative colitis as assessed by a machine learning approach. Gut Microbes. 2022a;14(1):2028366. https://doi.org/10.1080/19490976.2022.2028366.
Barberio B, Savarino EV, et al. Incidence comparison of adverse events in patients with inflammatory bowel disease receiving different biologic agents: retrospective long-term evaluation. Intest Res. 2022b;20(1):114–23. https://doi.org/10.5217/ir.2021.00037.
Barnes EL, et al. High dietary intake of specific fatty acids increases risk of flares in patients with ulcerative colitis in remission during treatment with aminosalicylates. Clin Gastroenterol Hepatol. 2017;15(9):1390–1396.e1. https://doi.org/10.1016/j.cgh.2016.12.036.
Birchenough G, et al. Dietary destabilisation of the balance between the microbiota and the colonic mucus barrier. Gut Microbes. 2019;10(2):246–50. https://doi.org/10.1080/19490976.2018.1513765.
Bischoff SC, et al. ESPEN practical guideline: clinical nutrition in inflammatory bowel disease. Clin Nutr. 2020;39(3):632–53. https://doi.org/10.1016/j.clnu.2019.11.002.
Bodini G, et al. A randomized, 6-wk trial of a low FODMAP diet in patients with inflammatory bowel disease. Nutrition. 2019;67–68:110542. https://doi.org/10.1016/j.nut.2019.06.023.
Bozkurt HS, Kara B. A new treatment for ulcerative colitis: intracolonic Bifidobacterium and xyloglucan application. Eur J Inflamm. 2020;18:205873922094262. https://doi.org/10.1177/2058739220942626.
Cariello M, et al. Extra-virgin olive oil from Apulian cultivars and intestinal inflammation. Nutrients. 2020;12(4):1084. https://doi.org/10.3390/nu12041084.
Cariello M, et al. Intestinal FXR activation via transgenic chimera or chemical agonism prevents colitis-associated and genetically-induced colon cancer. Cancers. 2022;14(13):3081. https://doi.org/10.3390/cancers14133081.
Charpentier C, et al. Dietary n-3 PUFA may attenuate experimental colitis. Mediat Inflamm. 2018;2018:1–10. https://doi.org/10.1155/2018/8430614.
Chassaing B, et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature. 2015;519(7541):92–6. https://doi.org/10.1038/nature14232.
Chiba M, et al. Relapse prevention by plant-based diet incorporated into induction therapy for ulcerative colitis: a single-group trial. Perm J. 2019;23(2):18–220. https://doi.org/10.7812/TPP/18-220.
Cohen LJ, et al. Genetic factors and the intestinal microbiome guide development of microbe-based therapies for inflammatory bowel diseases. Gastroenterology. 2019;156(8):2174–89. https://doi.org/10.1053/j.gastro.2019.03.017.
Comeche JM, et al. Parenteral nutrition in patients with inflammatory bowel disease systematic review, meta-analysis and meta-regression. Nutrients. 2019;11(12):2865. https://doi.org/10.3390/nu11122865.
De Sire R, et al. Exclusive enteral nutrition in adult Crohn’s disease: an overview of clinical practice and perceived barriers. Clin Exp Gastroenterol. 2021;14:493–501. https://doi.org/10.2147/CEG.S267172.
El Amrousy D, et al. Adherence to the Mediterranean diet improved clinical scores and inflammatory markers in children with active inflammatory bowel disease: a randomized trial. J Inflamm Res. 2022;15:2075–86. https://doi.org/10.2147/JIR.S349502.
Gadaleta RM, et al. Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut. 2011;60(4):463–72. https://doi.org/10.1136/gut.2010.212159.
Gadaleta RM, et al. Fibroblast growth factor 19 modulates intestinal microbiota and inflammation in presence of farnesoid X receptor. EBioMedicine. 2020;54:102719. https://doi.org/10.1016/j.ebiom.2020.102719.
Gadaleta RM, et al. Bile salt hydrolase-competent probiotics in the management of IBD: unlocking the “bile acid code”. Nutrients. 2022;14(15):3212. https://doi.org/10.3390/nu14153212.
Gibson GR, et al. Dietary prebiotics: current status and new definition. Food Sci Technol Bull Funct Foods. 2010;7(1):1–19. https://doi.org/10.1616/1476-2137.15880.
Guo Y, et al. Effects of oral vitamin D supplementation on inflammatory bowel disease: a systematic review and meta-analysis. Food Funct. 2021;12(17):7588–606. https://doi.org/10.1039/D1FO00613D.
He C, et al. Characteristics of mucosa-associated gut microbiota during treatment in Crohn’s disease. World J Gastroenterol. 2019;25(18):2204–16. https://doi.org/10.3748/wjg.v25.i18.2204.
Herman R, et al. Induction of remission in pediatric Crohn’s disease patients assessed by the mucosal inflammation noninvasive index. J Clin Med. 2021;10(23):5613. https://doi.org/10.3390/jcm10235613.
Jantchou P, et al. Animal protein intake and risk of inflammatory bowel disease: the E3N prospective study. Am J Gastroenterol. 2010;105(10):2195–201. https://doi.org/10.1038/ajg.2010.192.
Khademi Z, et al. Dietary intake of total carbohydrates, sugar and sugar-sweetened beverages, and risk of inflammatory bowel disease: a systematic review and meta-analysis of prospective cohort studies. Front Nutr. 2021;8:707795. https://doi.org/10.3389/fnut.2021.707795.
Kruis W. Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut. 2004;53(11):1617–23. https://doi.org/10.1136/gut.2003.037747.
Lee M, Chang EB. Inflammatory bowel diseases (IBD) and the microbiome – searching the crime scene for clues. Gastroenterology. 2021;160(2):524–37. https://doi.org/10.1053/j.gastro.2020.09.056.
Lee J-E, et al. Diet-induced host–microbe interactions: personalized diet strategies for improving inflammatory bowel disease. Curr Dev Nutr. 2022;6(8):nzac110. https://doi.org/10.1093/cdn/nzac110.
Lim W-C, et al. Aminosalicylates for induction of remission or response in Crohn’s disease. Cochrane Database Syst Rev. 2016;2016(7) Edited by Cochrane IBD Group. https://doi.org/10.1002/14651858.CD008870.pub2.
Limketkai BN, et al. Prevalence and factors associated with gluten sensitivity in inflammatory bowel disease. Scand J Gastroenterol. 2018;53(2):147–51. https://doi.org/10.1080/00365521.2017.1409364.
Lorentz A, Müller L. Probiotics in the treatment of inflammatory bowel disease in adulthood: a systematic review. J Gastrointestin Liver Dis. 2022;31(1):74–84. https://doi.org/10.15403/jgld-3936.
Lu Y, et al. Dietary polyphenols in the aetiology of Crohnʼs disease and ulcerative colitis – a multicenter European Prospective Cohort Study (EPIC). Inflamm Bowel Dis. 2017;23(12):2072–82. https://doi.org/10.1097/MIB.0000000000001108.
Ma C, Vasu R, Zhang H. The role of long-chain fatty acids in inflammatory bowel disease. Mediat Inflamm. 2019;2019:1–10. https://doi.org/10.1155/2019/8495913.
MacMaster MJ, et al. A prospective analysis of micronutrient status in quiescent inflammatory bowel disease. Clin Nutr. 2021;40(1):327–31. https://doi.org/10.1016/j.clnu.2020.05.010.
Marsal J, et al. Management of non-response and Loss of response to anti-tumor necrosis factor therapy in inflammatory bowel disease. Front Med. 2022;9:897936. https://doi.org/10.3389/fmed.2022.897936.
Masnadi Shirazi K, et al. Vitamin a supplementation decreases disease activity index in patients with ulcerative colitis: a randomized controlled clinical trial. Complement Ther Med. 2018;41:215–9. https://doi.org/10.1016/j.ctim.2018.09.026.
Mentella MC, et al. Nutrition, IBD and gut microbiota: a review. Nutrients. 2020;12(4):944. https://doi.org/10.3390/nu12040944.
Moustafa A, et al. Genetic risk, dysbiosis, and treatment stratification using host genome and gut microbiome in inflammatory bowel disease. Clin Transl Gastroenterol. 2018;9(1):e132. https://doi.org/10.1038/ctg.2017.58.
Nardone OM, et al. Lactose intolerance assessed by analysis of genetic polymorphism, breath test and symptoms in patients with inflammatory bowel disease. Nutrients. 2021;13(4):1290. https://doi.org/10.3390/nu13041290.
Nemoto H, et al. Reduced diversity and imbalance of fecal microbiota in patients with ulcerative colitis. Dig Dis Sci. 2012;57(11):2955–64. https://doi.org/10.1007/s10620-012-2236-y.
Opstelten JL, et al. Dietary intake of patients with inflammatory bowel disease: a comparison with individuals from a general population and associations with relapse. Clin Nutr. 2019;38(4):1892–8. https://doi.org/10.1016/j.clnu.2018.06.983.
Park YE, et al. Incidence and risk factors of micronutrient deficiency in patients with IBD and intestinal Behçet’s disease: folate, vitamin B12, 25-OH-vitamin D, and ferritin. BMC Gastroenterol. 2021;21(1):32. https://doi.org/10.1186/s12876-021-01609-8.
Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347(6):417–29. https://doi.org/10.1056/NEJMra020831.
Qiu X, et al. Characterization of fungal and bacterial dysbiosis in young adult Chinese patients with Crohn’s disease. Ther Adv Gastroenterol. 2020;13:175628482097120. https://doi.org/10.1177/1756284820971202.
Saez A, et al. Pathophysiology of inflammatory bowel disease: innate immune system. Int J Mol Sci. 2023;24(2):1526. https://doi.org/10.3390/ijms24021526.
Sandborn WJ, et al. Azathioprine or 6-mercaptopurine for induction of remission in Crohn’s disease. In: The Cochrane Collaboration, editor. Cochrane database of systematic reviews. Chichester: Wiley; 1998. p. CD000545. https://doi.org/10.1002/14651858.CD000545.
Serrano-Moreno C, et al. Diets for inflammatory bowel disease: what do we know so far? Eur J Clin Nutr. 2022;76(9):1222–33. https://doi.org/10.1038/s41430-021-01051-9.
Singh V, et al. Chronic inflammation in ulcerative colitis causes long-term changes in goblet cell function. Cell Mol Gastroenterol Hepatol. 2022;13(1):219–32. https://doi.org/10.1016/j.jcmgh.2021.08.010.
Sinha SR, et al. Dysbiosis-induced secondary bile acid deficiency promotes intestinal inflammation. Cell Host Microbe. 2020;27(4):659–670.e5. https://doi.org/10.1016/j.chom.2020.01.021.
Storz MA, et al. Bowel health in U.S. vegetarians: a 4-year data report from the National Health and Nutrition Examination Survey (NHANES). Nutrients. 2022;14(3):681. https://doi.org/10.3390/nu14030681.
Strauss J, et al. Invasive potential of gut mucosa-derived fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm Bowel Dis. 2011;17(9):1971–8. https://doi.org/10.1002/ibd.21606.
Sugihara K, Morhardt TL, Kamada N. The role of dietary nutrients in inflammatory bowel disease. Front Immunol. 2019;9:3183. https://doi.org/10.3389/fimmu.2018.03183.
Talebi S, et al. The association between total protein, animal protein, and animal protein sources with risk of inflammatory bowel diseases: a systematic review and meta-analysis of cohort studies. Adv Nutr. 2023:S2161831323003137. https://doi.org/10.1016/j.advnut.2023.05.008.
Tamaki H, et al. Efficacy of probiotic treatment with Bifidobacterium longum 536 for induction of remission in active ulcerative colitis: a randomized, double-blinded, placebo-controlled multicenter trial. Dig Endosc. 2016;28(1):67–74. https://doi.org/10.1111/den.12553.
Teofani A, et al. Intestinal taxa abundance and diversity in inflammatory bowel disease patients: an analysis including covariates and confounders. Nutrients. 2022;14(2):260. https://doi.org/10.3390/nu14020260.
Turpin W, et al. Determinants of IBD heritability: genes, bugs, and more. Inflamm Bowel Dis. 2018;24(6):1133–48. https://doi.org/10.1093/ibd/izy085.
Van Der Post S, et al. Structural weakening of the colonic mucus barrier is an early event in ulcerative colitis pathogenesis. Gut. 2019;68(12):2142–51. https://doi.org/10.1136/gutjnl-2018-317571.
Wlodarska M, et al. Indoleacrylic acid produced by commensal peptostreptococcus species suppresses inflammation. Cell Host Microbe. 2017;22(1):25–37.e6. https://doi.org/10.1016/j.chom.2017.06.007.
Yan J, et al. Dietary patterns and gut microbiota changes in inflammatory bowel disease: current insights and future challenges. Nutrients. 2022;14(19):4003. https://doi.org/10.3390/nu14194003.
Zamani S, et al. Detection of enterotoxigenic Bacteroides fragilis in patients with ulcerative colitis. Gut Pathogens. 2017;9(1):53. https://doi.org/10.1186/s13099-017-0202-0.
Zangara MT, et al. Maltodextrin consumption impairs the intestinal mucus barrier and accelerates colitis through direct actions on the epithelium. Front Immunol. 2022;13:841188. https://doi.org/10.3389/fimmu.2022.841188.
Zhang S-L, Wang S-N, Miao C-Y. Influence of microbiota on intestinal immune system in ulcerative colitis and its intervention. Front Immunol. 2017;8:1674. https://doi.org/10.3389/fimmu.2017.01674.
Zheng J, et al. Dietary inflammatory potential in relation to the gut microbiome: results from a cross-sectional study. Br J Nutr. 2020;124(9):931–42. https://doi.org/10.1017/S0007114520001853.
Zhou Y, et al. Increased Enterococcus faecalis infection is associated with clinically active Crohn disease. Medicine. 2016;95(39):e5019. https://doi.org/10.1097/MD.0000000000005019.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 Springer Nature Switzerland AG
About this entry
Cite this entry
Florio, M., Crudele, L., Moschetta, A., Gadaleta, R.M. (2024). Discovering the Nutrition-Microbiota Interplay in Inflammatory Bowel Disease: Are We There Yet?. In: Federici, M., Menghini, R. (eds) Gut Microbiome, Microbial Metabolites and Cardiometabolic Risk. Endocrinology. Springer, Cham. https://doi.org/10.1007/978-3-031-35064-1_14
Download citation
DOI: https://doi.org/10.1007/978-3-031-35064-1_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-35063-4
Online ISBN: 978-3-031-35064-1
eBook Packages: MedicineReference Module Medicine