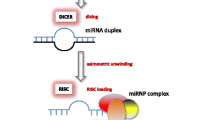
Abstract
In late years, a substantial advance has been made in the study of the role of microRNAs (miRNAs) in the pathogenesis of diseases. New evidence shows that dysregulation of miRNAs represents an etiologic factor of a variety of disorders, including cancer. Besides, miRNAs have also emerged as fundamental regulators of metabolic processes taking part in maintaining energy balance and metabolic homeostasis. Dysregulation of miRNAs, by directly affecting the status and functions of adipose tissue, pancreas, liver, and muscle, contributes to metabolic abnormalities being fully implicated in body fat accumulation, obesity, and obesity-related diseases. The discovery of circulating miRNAs has highlighted their potential as endocrine signaling molecules and disease indicators. Nevertheless, the study of the involvement of miRNAs in metabolic dysfunctions is still a young field of research and information on their role is nearly limited to date. As well, the contemporary rising in childhood obesity rates creates a need for tools that quantify metabolic changes in obese children and adolescents for the early detection or prevention of comorbidities. This chapter aims to provide current insights into the role of miRNAs as biological markers focusing on their link to obesity and obesity-related morbid conditions.
Availability of Data: We thoroughly explored PubMed using different combinations of keywords. Articles published until September 2021 were included. Whenever possible, the most recent and complete reviews on the research area were included. Due to length constraints, bibliographic references relevant to the topic from a variety of groups were excluded, but supplementary documents can be found within the references.
Similar content being viewed by others
Abbreviations
- AGO:
-
Argonaute
- BMI:
-
Body Mass Index
- BMPs:
-
bone morphogenetic proteins
- C/EBPs:
-
CCAAT/enhancer-binding proteins
- cmiRNAs:
-
circulating miRNAs
- DGCR8:
-
DiGeorge Syndrome Critical Region 8
- GWA:
-
genome-wide association (studies)
- HbA1c:
-
glycated hemoglobin A1c
- HNF:
-
hepatocyte nuclear factor
- HOMA-IR score:
-
homeostatic model assessment for insulin resistance
- LETFs:
-
liver-enriched transcription factors
- miRNAs:
-
microRNAs
- ncRNAs:
-
non-coding RNAs
- NGS :
-
next-generation sequencing
- NPM-1:
-
nucleophosmin-1
- PPAR-γ:
-
proliferator-activated receptor-γ
- RISCs:
-
RNA-induced silencing complexes
- SFA :
-
saturated fatty acids
- SREBP1:
-
sterol regulatory element-binding protein
- T1DM:
-
type 1 diabetes mellitus
- T2DM:
-
type-2 diabetes mellitus
- TGF-β:
-
transforming growth factor beta
- Wnt:
-
Wingless/Integrated
References
Alkandari A, Ashrafian H, Sathyapalan T, et al. Improved physiology and metabolic flux after Roux-en-Y gastric bypass is associated with temporal changes in the circulating microRNAome: a longitudinal study in humans. BMC Obes. 2018;5:20.
Andersen DC, Jensen CH, Schneider M, et al. MicroRNA-15a fine-tunes the level of Delta-like 1 homolog (DLK1) in proliferating 3T3-L1 preadipocytes. Exp Cell Res. 2010;316(10):1681–91.
Avnit-Sagi T, Kantorovich L, Kredo-Russo S, et al. The promoter of the pri-miR-375 gene directs expression selectively to the endocrine pancreas. PLoS One. 2009;4(4):e5033.
Balasubramanyam M, Aravind S, Gokulakrishnan K, et al. Impaired miR-146a expression links subclinical inflammation and insulin resistance in type 2 diabetes. Mol Cell Biochem. 2011;351(1–2):197–205.
Bao F, Slusher AL, Whitehurst M, et al. Circulating microRNAs are upregulated following acute aerobic exercise in obese individuals. Physiol Behav. 2018;197:15–21.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–97.
Bartel DP. Metazoan MicroRNAs. Cell. 2018;173(1):20–51.
Belarbi Y, Mejhert N, Lorente-Cebrian S, et al. MicroRNA-193b controls adiponectin production in human white adipose tissue. J Clin Endocrinol Metab. 2015;100(8):E1084–8.
Bork S, Horn P, Castoldi M, et al. Adipogenic differentiation of human mesenchymal stromal cells is down-regulated by microRNA-369-5p and up-regulated by microRNA-371. J Cell Physiol. 2011;226(9):2226–34.
Cai S, Pataillot-Meakin T, Shibakawa A, et al. Single-molecule amplification-free multiplexed detection of circulating microRNA cancer biomarkers from serum. Nat Commun. 2021;12(1):3515.
Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6(11):857–66.
Can U, Buyukinan M, Yerlikaya FH. The investigation of circulating microRNAs associated with lipid metabolism in childhood obesity. Pediatr Obes. 2016;11(3):228–34.
Carreras-Badosa G, Bonmati A, Ortega FJ, et al. Altered circulating miRNA expression profile in pregestational and gestational obesity. J Clin Endocrinol Metab. 2015;100(11):E1446–56.
Cermelli S, Ruggieri A, Marrero JA, et al. Circulating microRNAs in patients with chronic hepatitis C and non-alcoholic fatty liver disease. PLoS One. 2011;6(8):e23937.
Chakraborty C, Sharma AR, Sharma G, et al. Therapeutic advances of miRNAs: a preclinical and clinical update. J Adv Res. 2021;28:127–38.
Chen H, Mo D, Li M, et al. miR-709 inhibits 3T3-L1 cell differentiation by targeting GSK3beta of Wnt/beta-catenin signaling. Cell Signal. 2014a;26(11):2583–9.
Chen L, Cui J, Hou J, et al. A novel negative regulator of adipogenesis: microRNA-363. Stem Cells. 2014b;32(2):510–20.
Chen L, Dai YM, Ji CB, et al. MiR-146b is a regulator of human visceral preadipocyte proliferation and differentiation and its expression is altered in human obesity. Mol Cell Endocrinol. 2014c;393(1–2):65–74.
Cioffi M, Vallespinos-Serrano M, Trabulo SM, et al. MiR-93 controls adiposity via inhibition of Sirt7 and Tbx3. Cell Rep. 2015;12(10):1594–605.
Condrat CE, Thompson DC, Barbu MG, et al. miRNAs as biomarkers in disease: latest findings regarding their role in diagnosis and prognosis. Cell. 2020;9(2):276.
Deiuliis JA. MicroRNAs as regulators of metabolic disease: pathophysiologic significance and emerging role as biomarkers and therapeutics. Int J Obes. 2016;40(1):88–101.
Dong H, Lei J, Ding L, et al. MicroRNA: function, detection, and bioanalysis. Chem Rev. 2013;113(8):6207–33.
El Ouaamari A, Baroukh N, Martens GA, et al. miR-375 targets 3′-phosphoinositide-dependent protein kinase-1 and regulates glucose-induced biological responses in pancreatic beta-cells. Diabetes. 2008;57(10):2708–17.
Esau C, Kang X, Peralta E, et al. MicroRNA-143 regulates adipocyte differentiation. J Biol Chem. 2004;279(50):52361–5.
Finnegan EF, Pasquinelli AE. MicroRNA biogenesis: regulating the regulators. Crit Rev Biochem Mol Biol. 2013;48(1):51–68.
Flowers E, Gadgil M, Aouizerat BE, et al. Circulating micrornas associated with glycemic impairment and progression in Asian Indians. Biomark Res. 2015;3:22.
Galvao-Lima LJ, Morais AHF, Valentim RAM, et al. miRNAs as biomarkers for early cancer detection and their application in the development of new diagnostic tools. Biomed Eng Online. 2021;20(1):21.
Garcia-Jacobo RE, Uresti-Rivera EE, Portales-Perez DP, et al. Circulating miR-146a, miR-34a and miR-375 in type 2 diabetes patients, pre-diabetic and normal-glycaemic individuals in relation to beta-cell function, insulin resistance and metabolic parameters. Clin Exp Pharmacol Physiol. 2019;46(12):1092–100.
Gerin I, Bommer GT, McCoin CS, et al. Roles for miRNA-378/378* in adipocyte gene expression and lipogenesis. Am J Physiol Endocrinol Metab. 2010;299(2):E198–206.
Ghai V, Kim TK, Etheridge A, et al. Extracellular vesicle encapsulated MicroRNAs in patients with type 2 diabetes are affected by metformin treatment. J Clin Med. 2019;8(5):617.
Giardina S, Hernandez-Alonso P, Diaz-Lopez A, et al. Changes in circulating miRNAs in healthy overweight and obese subjects: effect of diet composition and weight loss. Clin Nutr. 2019;38(1):438–43.
Grueter CE, van Rooij E, Johnson BA, et al. A cardiac microRNA governs systemic energy homeostasis by regulation of MED13. Cell. 2012;149(3):671–83.
Guo Y, Chen Y, Zhang Y, et al. Up-regulated miR-145 expression inhibits porcine preadipocytes differentiation by targeting IRS1. Int J Biol Sci. 2012;8(10):1408–17.
Hamam D, Ali D, Vishnubalaji R, et al. microRNA-320/RUNX2 axis regulates adipocytic differentiation of human mesenchymal (skeletal) stem cells. Cell Death Dis. 2014;5:e1499.
Heneghan HM, Miller N, McAnena OJ, et al. Differential miRNA expression in omental adipose tissue and in the circulation of obese patients identifies novel metabolic biomarkers. J Clin Endocrinol Metab. 2011;96(5):E846–50.
Huang J, Zhao L, Xing L, et al. MicroRNA-204 regulates Runx2 protein expression and mesenchymal progenitor cell differentiation. Stem Cells. 2010;28(2):357–64.
Huang S, Wang S, Bian C, et al. Upregulation of miR-22 promotes osteogenic differentiation and inhibits adipogenic differentiation of human adipose tissue-derived mesenchymal stem cells by repressing HDAC6 protein expression. Stem Cells Dev. 2012;21(13):2531–40.
Huang HY, Lin YC, Li J, et al. miRTarBase 2020: updates to the experimentally validated microRNA-target interaction database. Nucleic Acids Res. 2020;48(D1):D148–54.
Huls A, Wright MN, Bogl LH, et al. Polygenic risk for obesity and its interaction with lifestyle and sociodemographic factors in European children and adolescents. Int J Obes. 2021;45(6):1321–30.
Hulsmans M, De Keyzer D, Holvoet P. MicroRNAs regulating oxidative stress and inflammation in relation to obesity and atherosclerosis. FASEB J. 2011;25(8):2515–27.
Iacomino G, Siani A. Role of microRNAs in obesity and obesity-related diseases. Genes Nutr. 2017;12:23.
Iacomino G, Russo P, Stillitano I, et al. Circulating microRNAs are deregulated in overweight/obese children: preliminary results of the I.Family study. Genes Nutr. 2016;11:7.
Iacomino G, Russo P, Marena P, et al. Circulating microRNAs are associated with early childhood obesity: results of the I.Family study. Genes Nutr. 2019;14:2.
Iacomino G, et al. Obesity and diabetes: scientific advances and best practice. SPRINGER NATURE; Cham, Switzerland; 2020. Chapter 6
Iacomino G, Lauria F, Russo P, et al. The association of circulating miR-191 and miR-375 expression levels with markers of insulin resistance in overweight children: an exploratory analysis of the I.Family study. Genes Nutr. 2021;16(1):10.
Jaeger A, Zollinger L, Saely CH, et al. Circulating microRNAs −192 and −194 are associated with the presence and incidence of diabetes mellitus. Sci Rep. 2018;8(1):14274.
Jeong BC, Kang IH, Hwang YC, et al. MicroRNA-194 reciprocally stimulates osteogenesis and inhibits adipogenesis via regulating COUP-TFII expression. Cell Death Dis. 2014;5:e1532.
Ji HL, Song CC, Li YF, et al. miR-125a inhibits porcine preadipocytes differentiation by targeting ERRalpha. Mol Cell Biochem. 2014;395(1–2):155–65.
Jimenez-Lucena R, Rangel-Zuniga OA, Alcala-Diaz JF, et al. Circulating miRNAs as predictive biomarkers of type 2 diabetes mellitus development in coronary heart disease patients from the CORDIOPREV study. Mol Ther Nucleic Acids. 2018;12:146–57.
Karolina DS, Tavintharan S, Armugam A, et al. Circulating miRNA profiles in patients with metabolic syndrome. J Clin Endocrinol Metab. 2012;97(12):E2271–6.
Keller A, Meese E. Can circulating miRNAs live up to the promise of being minimal invasive biomarkers in clinical settings? Wiley Interdiscip Rev RNA. 2016;7(2):148–56.
Kern F, Backes C, Hirsch P, et al. What’s the target: understanding two decades of in silico microRNA-target prediction. Brief Bioinform. 2020;21(6):1999–2010.
Kern F, Krammes L, Danz K, et al. Validation of human microRNA target pathways enables evaluation of target prediction tools. Nucleic Acids Res. 2021;49(1):127–44.
Kim YJ, Hwang SJ, Bae YC, et al. MiR-21 regulates adipogenic differentiation through the modulation of TGF-beta signaling in mesenchymal stem cells derived from human adipose tissue. Stem Cells. 2009;27(12):3093–102.
Kinoshita M, Ono K, Horie T, et al. Regulation of adipocyte differentiation by activation of serotonin (5-HT) receptors 5-HT2AR and 5-HT2CR and involvement of microRNA-448-mediated repression of KLF5. Mol Endocrinol. 2010;24(10):1978–87.
Kong L, Zhu J, Han W, et al. Significance of serum microRNAs in pre-diabetes and newly diagnosed type 2 diabetes: a clinical study. Acta Diabetol. 2011;48(1):61–9.
Krause BJ, Carrasco-Wong I, Dominguez A, et al. Micro-RNAs Let7e and 126 in plasma as markers of metabolic dysfunction in 10 to 12 years old children. PLoS One. 2015;10(6):e0128140.
Kredo-Russo S, Mandelbaum AD, Ness A, et al. Pancreas-enriched miRNA refines endocrine cell differentiation. Development. 2012;139(16):3021–31.
Langi G, Szczerbinski L, Kretowski A. Meta-analysis of differential miRNA expression after bariatric surgery. J Clin Med. 2019;8(8):1220.
Lee RC, Feinbaum RL, Ambros V. The C-elegans heterochronic gene Lin-4 encodes small RNAs with antisense complementarity to Lin-14. Cell. 1993;75(5):843–54.
Lee EK, Lee MJ, Abdelmohsen K, et al. miR-130 suppresses adipogenesis by inhibiting peroxisome proliferator-activated receptor gamma expression. Mol Cell Biol. 2011;31(4):626–38.
Li S, Zhu J, Zhang W, et al. Signature microRNA expression profile of essential hypertension and its novel link to human cytomegalovirus infection. Circulation. 2011;124(2):175–84.
Li H, Chen X, Guan L, et al. MiRNA-181a regulates adipogenesis by targeting tumor necrosis factor-alpha (TNF-alpha) in the porcine model. PLoS One. 2013;8(10):e71568.
Li M, Liu Z, Zhang Z, et al. miR-103 promotes 3T3-L1 cell adipogenesis through AKT/mTOR signal pathway with its target being MEF2D. Biol Chem. 2015;396(3):235–44.
Li X, Ballantyne LL, Yu Y, et al. Perivascular adipose tissue-derived extracellular vesicle miR-221-3p mediates vascular remodeling. FASEB J. 2019;33(11):12704–22.
Liang G, Zhu Y, Sun B, et al. Assessing the survival of exogenous plant microRNA in mice. Food Sci Nutr. 2014;2(4):380–8.
Ling HY, Wen GB, Feng SD, et al. MicroRNA-375 promotes 3T3-L1 adipocyte differentiation through modulation of extracellular signal-regulated kinase signalling. Clin Exp Pharmacol Physiol. 2011;38(4):239–46.
Locke AE, Kahali B, Berndt SI, et al. Genetic studies of body mass index yield new insights for obesity biology. Nature. 2015;518(7538):197–U401.
Lu Z, Wang J, Wang X, et al. miR-375 promotes pancreatic differentiation in vitro by affecting different target genes at different stages. Stem Cells Int. 2021;2021:6642983.
Matsuzaki J, Ochiya T. Circulating microRNAs and extracellular vesicles as potential cancer biomarkers: a systematic review. Int J Clin Oncol. 2017;22:413–20.
McCarthy JJ. The MyomiR network in skeletal muscle plasticity. Exerc Sport Sci Rev. 2011;39(3):150–4.
Mendez-Mancilla A, Lima-Rogel V, Toro-Ortiz JC, et al. Differential expression profiles of circulating microRNAs in newborns associated to maternal pregestational overweight and obesity. Pediatr Obes. 2018;13(3):168–74.
Mestdagh P, Hartmann N, Baeriswyl L, et al. Evaluation of quantitative miRNA expression platforms in the microRNA quality control (miRQC) study. Nat Methods. 2014;11(8):809–15.
Mononen N, Lyytikainen LP, Seppala I, et al. Whole blood microRNA levels associate with glycemic status and correlate with target mRNAs in pathways important to type 2 diabetes. Sci Rep. 2019;9(1):8887.
Nigi L, Grieco GE, Ventriglia G, et al. MicroRNAs as regulators of insulin signaling: research updates and potential therapeutic perspectives in type 2 diabetes. Int J Mol Sci. 2018;19(12):3705.
Nunez Lopez YO, Coen PM, Goodpaster BH, et al. Gastric bypass surgery with exercise alters plasma microRNAs that predict improvements in cardiometabolic risk. Int J Obes. 2017;41(7):1121–30.
Ortega FJ, Mercader JM, Catalan V, et al. Targeting the circulating microRNA signature of obesity. Clin Chem. 2013;59(5):781–92.
Oses M, Margareto Sanchez J, Portillo MP, et al. Circulating miRNAs as biomarkers of obesity and obesity-associated comorbidities in children and adolescents: a systematic review. Nutrients. 2019;11(12):2890.
Parrizas M, Mundet X, Castano C, et al. miR-10b and miR-223-3p in serum microvesicles signal progression from prediabetes to type 2 diabetes. J Endocrinol Investig. 2020;43(4):451–9.
Peng Y, Xiang H, Chen C, et al. MiR-224 impairs adipocyte early differentiation and regulates fatty acid metabolism. Int J Biochem Cell Biol. 2013;45(8):1585–93.
Pordzik J, Pisarz K, De Rosa S, et al. The potential role of platelet-related microRNAs in the development of cardiovascular events in high-risk populations, including diabetic patients: a review. Front Endocrinol (Lausanne). 2018;9:74.
Pordzik J, Jakubik D, Jarosz-Popek J, et al. Significance of circulating microRNAs in diabetes mellitus type 2 and platelet reactivity: bioinformatic analysis and review. Cardiovasc Diabetol. 2019;18(1):113.
Poy MN, Eliasson L, Krutzfeldt J, et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature. 2004;432(7014):226–30.
Prats-Puig A, Ortega FJ, Mercader JM, et al. Changes in circulating microRNAs are associated with childhood obesity. J Clin Endocrinol Metab. 2013;98(10):E1655–60.
Price NL, Fernandez-Hernando C. miRNA regulation of white and brown adipose tissue differentiation and function. Biochim Biophys Acta. 2016;1861(12 Pt B):2104–10.
Qin L, Chen Y, Niu Y, et al. A deep investigation into the adipogenesis mechanism: profile of microRNAs regulating adipogenesis by modulating the canonical Wnt/beta-catenin signaling pathway. BMC Genomics. 2010;11:320.
Sedgeman LR, Beysen C, Ramirez Solano MA, et al. Beta cell secretion of miR-375 to HDL is inversely associated with insulin secretion. Sci Rep. 2019;9(1):3803.
Shi XE, Li YF, Jia L, et al. MicroRNA-199a-5p affects porcine preadipocyte proliferation and differentiation. Int J Mol Sci. 2014;15(5):8526–38.
Shi C, Zhang M, Tong M, et al. miR-148a is associated with obesity and modulates adipocyte differentiation of mesenchymal stem cells through Wnt signaling. Sci Rep. 2015;5:9930.
Sidorkiewicz I, Niemira M, Maliszewska K, et al. Circulating miRNAs as a predictive biomarker of the progression from prediabetes to diabetes: outcomes of a 5-year prospective observational study. J Clin Med. 2020;9(7):1–20.
Simmonds M, Burch J, Llewellyn A, et al. The use of measures of obesity in childhood for predicting obesity and the development of obesity-related diseases in adulthood: a systematic review and meta-analysis. Health Technol Assess. 2015;19(43):1–336.
Skarn M, Namlos HM, Noordhuis P, et al. Adipocyte differentiation of human bone marrow-derived stromal cells is modulated by microRNA-155, microRNA-221, and microRNA-222. Stem Cells Dev. 2012;21(6):873–83.
Slattery ML, Herrick JS, Mullany LE, et al. Diet and lifestyle factors associated with miRNA expression in colorectal tissue. Pharmacogenomics Pers Med. 2017;10:1–16.
Song G, Xu G, Ji C, et al. The role of microRNA-26b in human adipocyte differentiation and proliferation. Gene. 2014;533(2):481–7.
Stranger BE, Stahl EA, Raj T. Progress and promise of genome-wide association studies for human complex trait genetics. Genetics. 2011;187(2):367–83.
Stratz C, Nuhrenberg T, Fiebich BL, et al. Controlled type II diabetes mellitus has no major influence on platelet micro-RNA expression. Results from micro-array profiling in a cohort of 60 patients. Thromb Haemost. 2014;111(5):902–11.
Sun T, Fu M, Bookout AL, et al. MicroRNA let-7 regulates 3T3-L1 adipogenesis. Mol Endocrinol. 2009;23(6):925–31.
Sun L, Xie H, Mori MA, et al. Mir193b-365 is essential for brown fat differentiation. Nat Cell Biol. 2011;13(8):958–65.
Szabo G, Bala S. MicroRNAs in liver disease. Nat Rev Gastroenterol Hepatol. 2013;10(9):542–52.
Tang YF, Zhang Y, Li XY, et al. Expression of miR-31, miR-125b-5p, and miR-326 in the adipogenic differentiation process of adipose-derived stem cells. OMICS. 2009;13(4):331–6.
Taniguchi M, Nakajima I, Chikuni K, et al. MicroRNA-33b downregulates the differentiation and development of porcine preadipocytes. Mol Biol Rep. 2014;41(2):1081–90.
Thomou T, Mori MA, Dreyfuss JM, et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature. 2017;542(7642):450–5.
Thompson MD, Cismowski MJ, Serpico M, et al. Elevation of circulating microRNA levels in obese children compared to healthy controls. Clin Obes. 2017;7:216–21.
Torres R, Lang UE, Hejna M, et al. MicroRNA ratios distinguish melanomas from nevi. J Invest Dermatol. 2019;139:S136.
Tribolet L, Kerr E, Cowled C, et al. MicroRNA biomarkers for infectious diseases: from basic research to biosensing. Front Microbiol. 2020;11:1197.
Tsai WC, Hsu SD, Hsu CS, Lai TC, Chen SJ, Shen R, Huang Y, Chen HC, Lee CH, Tsai TF, et al. MicroRNA-122 plays a critical role in liver homeostasis and hepatocarcinogenesis. J Clin Invest. 2012;122:2884–2897. https://doi.org/10.1172/JCI63455
Vasu S, Kumano K, Darden CM, et al. MicroRNA signatures as future biomarkers for diagnosis of diabetes states. Cell. 2019;8(12):1533.
Villard A, Marchand L, Thivolet C, et al. Diagnostic value of cell-free circulating MicroRNAs for obesity and type 2 diabetes: a meta-analysis. J Mol Biomark Diagn. 2015;6(6):251.
Wander PL, Boyko EJ, Hevner K, et al. Circulating early- and mid-pregnancy microRNAs and risk of gestational diabetes. Diabetes Res Clin Pract. 2017;132:1–9.
Wang Q, Li YC, Wang J, et al. miR-17-92 cluster accelerates adipocyte differentiation by negatively regulating tumor-suppressor Rb2/p130. Proc Natl Acad Sci U S A. 2008;105(8):2889–94.
Wang YT, Tsai PC, Liao YC, et al. Circulating microRNAs have a sex-specific association with metabolic syndrome. J Biomed Sci. 2013;20:72.
Witwer KW. Circulating microRNA biomarker studies: pitfalls and potential solutions. Clin Chem. 2015;61(1):56–63.
Xu P, Vernooy SY, Guo M, et al. The Drosophila microRNA Mir-14 suppresses cell death and is required for normal fat metabolism. Curr Biol CB. 2003;13(9):790–5.
Yang Z, Bian C, Zhou H, et al. MicroRNA hsa-miR-138 inhibits adipogenic differentiation of human adipose tissue-derived mesenchymal stem cells through adenovirus EID-1. Stem Cells Dev. 2011;20(2):259–67.
Zamanillo R, Sanchez J, Serra F, et al. Breast milk supply of MicroRNA associated with leptin and adiponectin is affected by maternal overweight/obesity and influences infancy BMI. Nutrients. 2019;11(11):2589.
Zampetaki A, Kiechl S, Drozdov I, et al. Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circ Res. 2010;107(6):810–7.
Zaragosi LE, Wdziekonski B, Brigand KL, et al. Small RNA sequencing reveals miR-642a-3p as a novel adipocyte-specific microRNA and miR-30 as a key regulator of human adipogenesis. Genome Biol. 2011;12(7):R64.
Zhang JF, Fu WM, He ML, et al. MiR-637 maintains the balance between adipocytes and osteoblasts by directly targeting Osterix. Mol Biol Cell. 2011;22(21):3955–61.
Zhang T, Lv C, Li L, et al. Plasma miR-126 is a potential biomarker for early prediction of type 2 diabetes mellitus in susceptible individuals. Biomed Res Int. 2013;2013:761617.
Zhu H, Leung SW. Identification of microRNA biomarkers in type 2 diabetes: a meta-analysis of controlled profiling studies. Diabetologia. 2015;58(5):900–11.
Zile MR, Mehurg SM, Arroyo JE, et al. Relationship between the temporal profile of plasma microRNA and left ventricular remodeling in patients after myocardial infarction. Circ Cardiovasc Genet. 2011;4(6):614–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Lauria, F., Venezia, A., Iacomino, G. (2022). Circulating MicroRNA (miRNA)s as Biological Markers and Links with Obesity and Obesity-Related Morbid Conditions. In: Patel, V.B., Preedy, V.R. (eds) Biomarkers in Nutrition . Biomarkers in Disease: Methods, Discoveries and Applications. Springer, Cham. https://doi.org/10.1007/978-3-030-81304-8_29-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-81304-8_29-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-81304-8
Online ISBN: 978-3-030-81304-8
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences