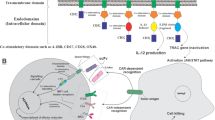
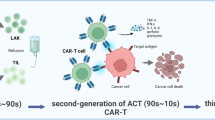
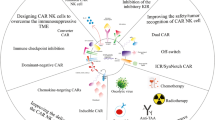
Abstract
Background. The review aims to present the major advances of adoptive cell immunotherapy from the emergence of antitumor immunotherapy to future perspectives. Methods. The search sources for the appropriate data included MEDLINE, PubMed, and references from the relevant articles. The keywords for the search process were as follows: adoptive cell immunotherapy (ACT), lymphokine-activated killers (LAK), cytokine-activated killers (CIK), dendritic cell (DC)-based vaccine, chimeric antigen receptor T-cells (CAR-T-cells), tumor-infiltrating lymphocytes (TILs), natural killer (NK) cells, CAR-T-cell therapy, and CAR-NK cells. The papers published in English between 1994 and 2022 were considered for the review. Results. The initial experimental research demonstrated the start of a novel and very promising antitumor therapy, though the first clinical trials with the combination of LAK cells and high-dose IL-2 therapy showed less effectiveness than expected. Further development of LAK/IL-2 therapy led to improved results in locoregional treatment, and CIK modifications enhanced effectiveness in adjuvant settings and relapse-free survival of patients with solid tumors. An important milestone in the cell-based therapy was the design of antitumor DC vaccines tailored to induce a specific immune response. Various approaches to improve adoptive cell immunotherapy resulted in designing a number of activation methods for immune effectors, such as TILs, NK cells, and NKT lymphocytes. However, the major breakthrough in cell immunotherapy was achieved with the introduction of genetically modified lymphocytes and implementation of CAR technologies. Although CAR-T-cell therapy has serious unfavorable effects, the experimental and clinical studies of CAR-T and CAR-NK treatment are currently of the highest interest in the field of adoptive cell immunotherapy, and their different modifications seem to be very promising. Conclusion. The perspectives of the adoptive cell immunotherapy are virtually immense. A variety of options for CAR structure modifications, genetic engineering, and selection of effector cells suggest further research potential. Moreover, combining adoptive cell therapy with other medications, such as immune checkpoint inhibitors, ensures improved clinical outcomes.
Similar content being viewed by others
References
Agliardi G, Liuzzi AR, Hotblack A et al (2021) Intratumoral IL-12 delivery empowers CAR-T cell immunotherapy in a pre-clinical model of glioblastoma. Nat Commun 12(1):444. https://doi.org/10.1038/s41467-020-20599-x. PMID: 33469002; PMCID: PMC7815781
Biederstädt A, Rezvani K (2021) Engineering the next generation of CAR-NK immunotherapies. Int J Hematol 114(5):554–571. https://doi.org/10.1007/s12185-021-03209-4
Bielamowicz K, Fousek K, Byrd TT et al (2017) Trivalent CAR T-cells overcome interpatient antigenic variability in glioblastoma. Neuro-Oncology 20:506. https://doi.org/10.1093/neuonc/nox182
Boyiadzis M, Agha M, Redner RL et al (2017) Phase 1 clinical trial of adoptive immunotherapy using ―off-the-shelf‖ activated natural killer cells in patients with refractory and relapsed acute myeloid leukemia. Cytotherapy 19(10):1225–1232. https://doi.org/10.1016/j.jcyt.2017.07.008
Brown CE, Alizadeh D, Starr R et al (2016) Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N Engl J Med 375(26):25619. https://doi.org/10.1056/NEJMoa1610497
Buchbinder EI, Desai A (2016) CTLA-4 and PD-1 pathways: similarities, differences, and implications of their inhibition. Am J Clin Oncol 39(1):98–106. https://doi.org/10.1097/COC.000000000000023
Chen Y, You F, Jiang L et al (2017) Gene-modified NK- 92MI cells expressing a chimeric CD16-BB-zeta or CD64-BB-zeta receptor exhibit enhanced cancer-killing ability in combination with therapeutic antibody. Oncotarget 8(23):37128–37139. https://doi.org/10.18632/oncotarget.1620
Chhabra N, Kennedy J (2022) A review of cancer immunotherapy toxicity II: adoptive cellular therapies, kinase inhibitors, monoclonal antibodies, and oncolytic viruses. J Med Toxicol 18(1):43–55. https://doi.org/10.1007/s13181-021-00835-6. PMID: 33821435; PMCID: PMC8021214
Cho S-F, Anderson KC, Tai Y-T (2018) Targeting B. Cell maturation antigen (BCMA) in multiple myeloma: potential uses of BCMA-based immunotherapy. Front Immunol 9:1821. https://doi.org/10.3389/fimmu.2018.01821
Chongsathidkiet P, Jackson C, Koyama S et al (2018) Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors. Nat Med 24:1459–1468. https://doi.org/10.1038/s41591-018-0135-2
Creagan ET, Rowland KM Jr, Suman VJ et al (1997) Phase II study of combined levamisole with recombinant interleukin-2 in patients with advanced malignant melanoma. Am J Clin Oncol 20(5):490–492. https://doi.org/10.1097/00000421-199710000-00011
Dafni U, Michielin O, Lluesma SM et al (2019) Efficacy of adoptive therapy with tumor-infiltrating lymphocytes and recombinant interleukin-2 in advanced cutaneous melanoma: a systematic review and meta-analysis. Ann Oncol 30(12):1902–1913. https://doi.org/10.1093/annonc/mdz398. PMID: 31566658
Davies JOJ, Stringaris K, Barrett AJ et al (2014) Opportunities and limitations of natural killer cells as adoptive therapy for malignant disease. Cytotherapy 16(11):1453–1466. https://doi.org/10.1016/j.jcyt.2014.03.009
Donovan LK, Delaidelli A, Joseph SK et al (2020) Locoregional delivery of CAR T cells to the cerebrospinal fluid for treatment of metastatic medulloblastoma and ependymoma. Nat Med 26(5):720–731. https://doi.org/10.1038/s41591-020-0827-2
Federico SM, McCarville MB, Shulkin BL et al (2017) A pilot trial of humanized anti-GD2 monoclonal antibody (hu14.18K322A) with chemotherapy and natural killer cells in children with recurrent/refractory neuroblastoma. Clin Cancer Res 23(21):6441–6449. https://doi.org/10.1158/1078-0432.CCR-17-037
Freeman AJ, Vervoort SJ, Ramsbottom KM et al (2019) Natural killer cells suppress T cell-associated tumor immune evasion. Cell Rep 28:2784–94.e5. https://doi.org/10.1016/j.celrep.2019.08.017
Golchin A, Farahany TZ (2019) Biological products: cellular therapy and FDA approved products. Stem Cell Rev Rep 15:166–175. https://doi.org/10.1007/s12015-018-9866-1
Gong JH, Maki G, Klingemann HG (1994) Characterization of a human cell line (NK-92) with phenotypical and functional characteristics of activated natural killer cells. Leukemia 8:652–658
Guo Z, Tu S, Yu S et al (2021) Preclinical and clinical advances in dual-target chimeric antigen receptor therapy for hematological malignancies. Cancer Sci 112(4):1357–1368. https://doi.org/10.1111/cas.14799
He X, Gao Y, Li Z et al (2021) Review on natural killer /T-cell lymphoma. Hematol Oncol. https://doi.org/10.1002/hon.2944. Epub ahead of print. PMID: 34731509
Hinrichs CS, Restifo NP (2013) Reassessing target antigens for adoptive T-cell therapy. Nat Biotechnol 31(11):999–1008. https://doi.org/10.1038/nbt.2725
Hirai I, Funakoshi T, Kamijuku H et al (2021) Adoptive cell therapy using tumor-infiltrating lymphocytes for melanoma refractory to immune-checkpoint inhibitors. Cancer Sci 112(8):3163–3172. https://doi.org/10.1111/cas.15009
Introna M, Lussana F, Algarotti A et al (2017) Phase II study of sequential infusion of donor lymphocyte infusion and cytokine-induced killer cells for patients relapsed after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 23(12):2070–2078. https://doi.org/10.1016/j.bbmt.2017.07.00
Kalos M, Levine BL, Porter DL et al (2011) T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci Transl Med 3(95):9–73
Kalos M, Nazimuddin F, Finklestein JM et al (2013) Long-term functional persistence, B cell aplasia and anti- leukemia efficacy in refractory B cell malignancies following T cell immunotherapy using CAR-redirected T cells targeting CD19. Blood 122:163
Kirtane K, Elmariah H, Chung CH, Abate-Daga D (2021) Adoptive cellular therapy in solid tumor malignancies: review of the literature and challenges ahead. J Immunother Cancer 9(7):e002723. https://doi.org/10.1136/jitc-2021-002723
Kiselevsky MV (2021) Malignant effusions. Springer
Kraehenbuehl L, Weng CH, Eghbali S et al (2022) Enhancing immunotherapy in cancer by targeting emerging immunomodulatory pathways. Nat Rev Clin Oncol 19(1):37–50. https://doi.org/10.1038/s41571-021-00552-7
Lanier LL (1998) NK cell receptors. Annu Rev Immunol 16:359–393. https://doi.org/10.1146/annurev.immunol.16.1.359
Levine DB (2008) The hospital for the ruptured and crippled: William Bradley Coley, third surgeon-in-chief 1925–1933. HSS J 4(1):1–9. https://doi.org/10.1007/s11420-007-9063-2
Lisi L, Lacal PM, Martire M et al (2022) Clinical experience with CTLA-4 blockade for cancer immunotherapy: from the monospecific monoclonal antibody ipilimumab to probodies and bispecific molecules targeting the tumor microenvironment. Pharmacol Res 175:105997. https://doi.org/10.1016/j.phrs.2021.105997
Liu E, Marin D, Banerjee P et al (2020) Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med 382:545–553. https://doi.org/10.1056/NEJMoa1910607
Long AH, Highfill SL, Cui Y et al (2016) Reduction of MDSCs with all-trans retinoic acid improves CAR therapy efficacy for sarcomas. Cancer Immunol Res 4(10):869–880. https://doi.org/10.1158/2326-6066.CIR-15-0230
MacLeod RA, Nagel S, Kaufmann M et al (2002) Multicolor-FISH analysis of a natural killer cell line (NK-92). Leuk Res 26(11):1027–1033. https://doi.org/10.1016/S0145-2126(02)00055-3
Maki G, Klingemann HG, Martinson JA et al (2001) Factors regulating the cytotoxic activity of the human natural killer cell line, NK-92. J Hematother Stem Cell Res 10(3):369–383. https://doi.org/10.1089/152581601750288975
Méndez R, Aptsiauri N, Del Campo A et al (2009) HLA and melanoma: multiple alterations in HLA class I and II expression in human melanoma cell lines from ESTDAB cell bank. Cancer Immunol Immunother 58:1507–1515. https://doi.org/10.1007/s00262-009-0701-z
Ni Z, Knorr DA, Bendzick L et al (2014) Expression of chimeric receptor CD4zeta by natural killer cells derived from human pluripotent stem cells improves in vitro activity but does not enhance suppression of HIV infection in vivo. Stem Cells 32(4):1021–1031. https://doi.org/10.1002/stem.1611
Oelsner S, Friede ME, Zhang C et al (2017) Continuously expanding CAR NK-92 cells display selective cytotoxicity against B-cell leukemia and lymphoma. Cytotherapy 19(2):235–249. https://doi.org/10.1016/j.jcyt.2016.10.009
Olson JA, Leveson-Gower DB, Gill S et al (2010) NK cells mediate reduction of GVHD by inhibiting activated, alloreactive T cells while retaining GVT effects. Blood 115(21):4293–4301. https://doi.org/10.1182/blood-2009-05-222190
O'Rourke DM, Nasrallah MP, Desai A et al (2017) A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med 9(399):eaaa0984. https://doi.org/10.1126/scitranslmed.aaa0984. PMID: 28724573; PMCID: PMC5762203
Palucka K, Ueno H, Roberts L, Fay J, Banchereau J (2010) Dendritic cells: are they clinically relevant? Cancer J 16(4):318–324. https://doi.org/10.1097/PPO.0b013e3181eaca83. PMID: 20693842; PMCID: PMC2919819
Park JH, Riviere I, Gonen M et al (2018) Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N Engl J Med 378(5):449–459
Rosenberg SA, Yang JC, Sherry RM et al (2011) Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res 17(13):4550–4557. https://doi.org/10.1158/1078-0432.CCR-11-0116. Epub 2011 Apr 15. PMID: 21498393; PMCID: PMC3131487
Rouce RH, Shaim H, Sekine T et al (2016) The TGF-beta/SMAD pathway is an important mechanism for NK cell immune evasion in childhood B-acute lymphoblastic leukemia. Leukemia 30(4):800–811. https://doi.org/10.1038/leu.2015.327
Sadeghzadeh M, Bornehdeli S, Mohahammadrezakhani H, Abolghasemi M, Poursaei E, Asadi M, Zafari V, Aghebati-Maleki L, Shanehbandi D (2020) Dendritic cell therapy in cancer treatment; the state-of-the-art. Life Sci 254:117580. https://doi.org/10.1016/j.lfs.2020.117580. PMID: 32205087
Santos PM, Butterfield LH (2018) Dendritic cell-based cancer vaccines. J Immunol 200(2):443–449. https://doi.org/10.4049/jimmunol.1701024. PMID: 29311386; PMCID: PMC5880540
Schönfeld K, Sahm C, Zhang C et al (2015) Selective inhibition of tumor growth by clonal NK cells expressing an ErbB2/HER2-specific chimeric antigen receptor. Mol Ther 23(2):330–338. https://doi.org/10.1038/mt.2014.219
Seitz CM, Flaadt T, Mezger M et al (2021) Immunomonitoring of stage IV relapsed neuroblastoma patients undergoing haploidentical hematopoietic stem cell transplantation and subsequent GD2 (ch14.18/CHO) antibody treatment. Front Immunol 12:690467. https://doi.org/10.3389/fimmu.2021.690467
Simonetta F, Alvarez M, Negrin RS (2017) Natural killer cells in graft-versus-host-disease after allogeneic hematopoietic cell transplantation. Front Immunol 8:465. https://doi.org/10.3389/fimmu.2017.00465
Theruvath J, Sotillo E, Mount CW et al (2020) Locoregionally administered B7-H3-targeted CAR T cells for treatment of atypical teratoid/rhabdoid tumors. Nat Med 26(5):712–719. https://doi.org/10.1038/s41591-020-0821-8. PMID: 32341579; PMCID: PMC7992505
Ueda T, Kaneko S (2021) Induced pluripotent stem cell-derived natural killer cells gene-modified to express chimeric antigen receptor-targeting solid tumors. Int J Hematol 114(5):572–579. https://doi.org/10.1007/s12185-020-02951-5
van Vliet AA, Georgoudaki AM, Raimo M et al (2021) Adoptive NK cell therapy: a promising treatment prospect for metastatic melanoma. J Cancers (Basel) 13(18):4722. https://doi.org/10.3390/cancers1318472
Waldman AD, Fritz JM, Lenardo MJ (2020) A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol 20(11):651–668. https://doi.org/10.1038/s41577-020-0306-5
Wang Z, Zhou G, Risu N et al (2020) Lenalidomide enhances CAR-T cell activity against solid tumor. Cell Transplant 29:963689720920825. https://doi.org/10.1177/0963689720920825
Wilkie S, van Schalkwyk MC, Hobbs S et al (2012) Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J Clin Immunol 32(5):1059–1070. https://doi.org/10.1007/s10875-012-9689-9
Yu S, Li A, Liu Q et al (2017) Chimeric antigen receptor T cells: a novel therapy for solid tumors. J Hematol Oncol 10(1):78. https://doi.org/10.1186/s13045-017-0444-9
Zhang C, Oberoi P, Oelsner S et al (2017) Chimeric antigen receptor-engineered NK-92 cells: an off-the-shelf cellular therapeutic for targeted elimination of cancer cells and induction of protective antitumor immunity. Front Immunol 8:533. https://doi.org/10.3389/fimmu.2017.00533
Zhang J, Zheng H, Diao Y (2019) Natural killer cells and current applications of chimeric antigen receptor-modified NK-92 cells in tumor immunotherapy. Int J Mol Sci 20(2):317. https://doi.org/10.3390/ijms20020317
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Shubina, I.Z., Chikileva, I.O., Kirgizov, K.I., Varfolomeeva, S.R., Kiselevskiy, M.V. (2022). Adoptive Immunotherapy Is a Successful Step into Bright Future. In: Rezaei, N. (eds) Handbook of Cancer and Immunology. Springer, Cham. https://doi.org/10.1007/978-3-030-80962-1_182-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80962-1_182-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80962-1
Online ISBN: 978-3-030-80962-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences