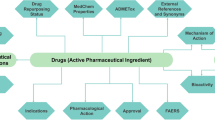
Abstract
Docking and quantifying the binding of small molecules to the 3D structure of a macromolecular bioregulator by computational techniques is a typical task in R&D aimed at the design and optimization of medically or otherwise active compounds. Much less known is the fact that these methods can be successfully applied for the purpose of toxicity prediction—for example, detecting a compound’s potential binding to so-called “off-targets” already at the preclinical stage. In this chapter, we provide an overview of such a computational approach, discuss its strengths and weaknesses, and include a case study—focused on natural compounds present in traditional medicines.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (1997) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 23:3–25
Veber DF, Johnson SR, Cheng H-Y, Smith BR, Ward KW, Kopple KD (2002) Molecular properties that influence the oral bioavailability of drug candidates. J Med Chem 45:2615–2623
Carrupt P-A, Testa B, Gaillard P (1997) Computational approaches to lipophilicity: methods and applications. Rev Comp Chem 11:241–315
Wang R, Fu Y, Lai L (1997) A new atom-additive method for calculating partition coefficients. J Chem Inf Comput Sci 37:615–621
Kellogg GE, Abraham DJ (2000) Hydrophobicity: is LogP o/w more than the sum of its parts? Eur J Med Chem 35:651–661
Tetko IV, Tanchuk VY, Villa AE (2001) Prediction of n-octanol/water partition coefficients from PHYSPROP database using artificial neural networks and E-state indices. J Chem Inf Comput Sci 41:1407–1421
Tetko IV, Tanchuk VY (2002) Application of associative neural networks for prediction of lipophilicity in ALOGPS 2.1 program. J Chem Inf Comput Sci 42:1136–1145, http://www.vcclab.org/lab/alogps/
Kalgutkar AS (2011) Handling reactive metabolite positives in drug discovery: what has retrospective structure–toxicity analyses taught us? Chem Biol Interact 192:46–55
Spyrakis F, Kellogg GE, Amadasi A, Cozzini P (2007) Scoring functions for virtual screening. Front Drug Des Discov 3:317–379, Bentham Science Publishers Ltd., eISBN: 978-1-60805-201-1
Polgár T, Keserü GM (2007) Structure-based virtual screening. Front Drug Des Discov 3:477–502, Bentham Science Publishers Ltd., eISBN: 978-1-60805-201-1
Vedani A, Dobler M, Hu Z, Smieško M (2015) OpenVirtualToxLab—a platform for generating and exchanging in silico toxicity data. Toxicol Lett 232:519–532, available at http://www.virtualtoxlab.org/
Kumar S, Pandey K (2013) Chemistry and biological activities of flavonoids: an overview. Sci World J 2013:162750. doi:10.1155/2013/162750
Allen FH (2002) The Cambridge Structural Database: a quarter of a million crystal structures and rising. Acta Crystallogr B 58:380–388
QikProp, version 4.0 (2014) Schrödinger, LLC, New York
Desmond Molecular Dynamics System, version 3.8 (2014) D. E. Shaw Research, New York. Maestro-Desmond Interoperability Tools, version 3.8 (2014) Schrödinger, New York
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this protocol
Cite this protocol
Smieško, M., Vedani, A. (2016). VirtualToxLab: Exploring the Toxic Potential of Rejuvenating Substances Found in Traditional Medicines. In: Benfenati, E. (eds) In Silico Methods for Predicting Drug Toxicity. Methods in Molecular Biology, vol 1425. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-3609-0_7
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3609-0_7
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-3607-6
Online ISBN: 978-1-4939-3609-0
eBook Packages: Springer Protocols