Abstract
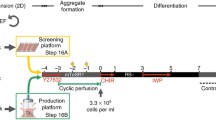
The formation of cells into more physiologically relevant three-dimensional multicellular aggregates is an important technique for the differentiation and manipulation of stem cells and their progeny. As industrial and clinical applications for these cells increase, it will be necessary to execute this procedure in a readily scalable format. We present here a method employing microwells to generate large numbers of human pluripotent stem cell aggregates and control their subsequent differentiation towards a cardiac fate.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wintermantel E, Mayer J, Blum J et al (1996) Tissue engineering scaffolds using superstructures. Biomaterials 17:83–91
Lazar A, Peshwa MV, Wu FJ et al (1995) Formation of porcine hepatocyte spheroids for use in a bioartificial liver. Cell Transplant 4:259–268
Sachlos E, Auguste DT (2008) Embryoid body morphology influences diffusive transport of inductive biochemicals: a strategy for stem cell differentiation. Biomaterials 29:4471–4480
Johnstone B, Hering TM, Caplan AI et al (1998) In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res 238:265–272
Steinberg MS (1970) Does differential adhesion govern self-assembly processes in histogenesis? Equilibrium configurations and the emergence of a hierarchy among populations of embryonic cells. J Exp Zool 173:395–433
Bauwens CL, Peerani R, Niebruegge S et al (2008) Control of human embryonic stem cell colony and aggregate size heterogeneity influences differentiation trajectories. Stem Cells 26:2300–2310
Koike M, Kurosawa H, Amano Y (2005) A round-bottom 96-well polystyrene plate coated with 2-methacryloyloxyethyl phosphorylcholine as an effective tool for embryoid body formation. Cytotechnology 47:3–10
Ng ES, Davis RP, Azzola L et al (2005) Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood 106:1601–1603
Ungrin MD, Joshi C, Nica A et al (2008) Reproducible, ultra high-throughput formation of multicellular organization from single cell suspension-derived human embryonic stem cell aggregates. PLoS One 3:e1565
Kozhich O, Hamilton R, Mallon B (2013) Standardized generation and differentiation of neural precursor cells from human pluripotent stem cells. Stem Cell Rev Rep 9:531–536
Ungrin MD, Clarke G, Yin T et al (2012) Rational bioprocess design for human pluripotent stem cell expansion and endoderm differentiation based on cellular dynamics. Biotechnol Bioeng 109:853–866
Bauwens CL, Song H, Thavandiran N et al (2011) Geometric control of cardiomyogenic induction in human pluripotent stem cells. Tissue Eng Part A 17:1901–1909
Golos TG, Giakoumopoulos M, Garthwaite MA (2010) Embryonic stem cells as models of trophoblast differentiation: progress, opportunities, and limitations. Reproduction 140:3–9
Markway B, Tan G-K, Brooke G et al (2010) Enhanced chondrogenic differentiation of human bone marrow-derived mesenchymal stem cells in low oxygen environment micropellet cultures. Cell Transplant 19(1):29–42
Fey SJ, Wrzesinski K (2012) Determination of drug toxicity using 3D spheroids constructed from an immortal human hepatocyte cell line. Toxicol Sci 127(2):403–411
Wallace L, Reichelt J (2013) Using 3D culture to investigate the role of mechanical signaling in keratinocyte stem cells. In: Turksen K (ed) Skin stem cells. Humana Press, Totowa, NJ, pp 153–164
Acknowledgements
We thank Dr. Peter Zandstra, in whose laboratory this protocol was developed, and Drs. Mark Gagliardi and Gordon Keller who provided assistance in establishing the initial methods on which this process was based. Protocol development was supported by an Ontario Graduate Scholarship in Science and Technology to C.B. and a grant from the Heart and Stroke Foundation of Ontario to Peter Zandstra.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Bauwens, C.L., Ungrin, M.D. (2014). Scalable Cardiac Differentiation of Human Pluripotent Stem Cells as Microwell-Generated, Size Controlled Three-Dimensional Aggregates. In: Radisic, M., Black III, L. (eds) Cardiac Tissue Engineering. Methods in Molecular Biology, vol 1181. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1047-2_2
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1047-2_2
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1046-5
Online ISBN: 978-1-4939-1047-2
eBook Packages: Springer Protocols