Abstract
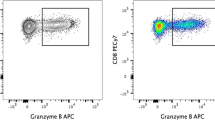
The detection and functional characterization of antigen-reactive T helper (Th) cells has been challenging due to their low frequency and functional heterogeneity. Antigen-reactive T cell enrichment (ARTE) allows the in-depth characterization of antigen-specific Th lymphocytes as a prerequisite for better understanding the role of adaptive immune responses in health and disease. ARTE is based on detection of the activation markers CD154 (CD40L) (expressed on all conventional Th cell subsets, Tcons) and CD137 (4-1BB) (expressed on regulatory T cells, Tregs), which are upregulated on the surface of CD4+ T cells upon short-term (7 h) in vitro stimulation with antigens in the presence of antigen-presenting cells (APCs). To substantially increase the sensitivity for the detection of antigen-specific Th cells, ARTE combines magnetic pre-enrichment of rare antigen-reactive T cells with multiparameter flow cytometry. Using CD154 and CD137 in combination allows the parallel detection of reactive Tcons and Tregs, after stimulation with the antigen. Thus, the ARTE technology now enables to characterize antigen-specific T cells with increased sensitivity of detection allowing even the investigation of antigen-specific Th cells in the naive T cell repertoire and regardless of prior knowledge of MHC alleles or antigenic epitopes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Geiger R, Duhen T, Lanzavecchia A, Sallusto F (2009) Human naive and memory CD4+ T cell repertoires specific for naturally processed antigens analyzed using libraries of amplified T cells. J Exp Med 206(7):1525–1534. https://doi.org/10.1084/jem.20090504
Nepom GT (2012) MHC class II tetramers. J Immunol 188(6):2477–2482. https://doi.org/10.4049/jimmunol.1102398
Letsch A, Scheibenbogen C (2003) Quantification and characterization of specific T-cells by antigen-specific cytokine production using ELISPOT assay or intracellular cytokine staining. Methods 31(2):143–149
Bacher P, Scheffold A (2013) Flow-cytometric analysis of rare antigen-specific T cells. Cytometry A 83(8):692–701. https://doi.org/10.1002/cyto.a.22317
Bacher P, Scheffold A (2015) New technologies for monitoring human antigen-specific T cells and regulatory T cells by flow-cytometry. Curr Opin Pharmacol 23:17–24. https://doi.org/10.1016/j.coph.2015.04.005
Kmieciak M, Gowda M, Graham L, Godder K et al (2009) Human T cells express CD25 and Foxp3 upon activation and exhibit effector/memory phenotypes without any regulatory/suppressor function. J Transl Med 7:89. https://doi.org/10.1186/1479-5876-7-89
Bremser A, Brack M, Izcue A (2015) Higher sensitivity of Foxp3+ Treg compared to Foxp3- conventional T cells to TCR-independent signals for CD69 induction. PLoS One 10(9):e0137393. https://doi.org/10.1371/journal.pone.0137393
Caruso A, Licenziati S, Corulli M, Canaris AD et al (1997) Flow cytometric analysis of activation markers on stimulated T cells and their correlation with cell proliferation. Cytometry 27(1):71–76
Mardiney M 3rd, Brown MR, Fleisher TA (1996) Measurement of T-cell CD69 expression: a rapid and efficient means to assess mitogen- or antigen-induced proliferative capacity in normals. Cytometry 26(4):305–310. https://doi.org/10.1002/(SICI)1097-0320(19961215)26:4<305::AID-CYTO11>3.0.CO;2-V
Bacher P, Schink C, Teutschbein J, Kniemeyer O et al (2013) Antigen-reactive T cell enrichment for direct, high-resolution analysis of the human naive and memory Th cell repertoire. J Immunol 190(8):3967–3976. https://doi.org/10.4049/jimmunol.1202221
Bacher P, Kniemeyer O, Schonbrunn A, Sawitzki B et al (2014) Antigen-specific expansion of human regulatory T cells as a major tolerance mechanism against mucosal fungi. Mucosal Immunol 7(4):916–928. https://doi.org/10.1038/mi.2013.107
Bacher P, Kniemeyer O, Teutschbein J, Thon M et al (2014) Identification of immunogenic antigens from Aspergillus fumigatus by direct multiparameter characterization of specific conventional and regulatory CD4+ T cells. J Immunol 193(7):3332–3343. https://doi.org/10.4049/jimmunol.1400776
Bacher P, Heinrich F, Stervbo U, Nienen M et al (2016) Regulatory T cell specificity directs tolerance versus allergy against aeroantigens in humans. Cell 167(4):1067–1078. e1016. https://doi.org/10.1016/j.cell.2016.09.050
Bacher P, Hohnstein T, Beerbaum E, Rocker M et al (2019) Human anti-fungal Th17 immunity and pathology rely on cross-reactivity against Candida albicans. Cell 176(6):1340–1355. e1315. https://doi.org/10.1016/j.cell.2019.01.041
Nowak A, Lock D, Bacher P, Hohnstein T et al (2018) CD137+CD154- expression as a regulatory T cell (Treg)-specific activation signature for identification and sorting of stable human Tregs from in vitro expansion cultures. Front Immunol 9:199. https://doi.org/10.3389/fimmu.2018.00199
Chattopadhyay PK, Yu J, Roederer M (2006) Live-cell assay to detect antigen-specific CD4+ T-cell responses by CD154 expression. Nat Protoc 1(1):1–6. https://doi.org/10.1038/nprot.2006.1
Kirchhoff D, Frentsch M, Leclerk P, Bumann D et al (2007) Identification and isolation of murine antigen-reactive T cells according to CD154 expression. Eur J Immunol 37(9):2370–2377. https://doi.org/10.1002/eji.200737322
Elgueta R, Benson MJ, de Vries VC, Wasiuk A et al (2009) Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol Rev 229(1):152–172. https://doi.org/10.1111/j.1600-065X.2009.00782.x
Yellin MJ, Sippel K, Inghirami G, Covey LR et al (1994) CD40 molecules induce down-modulation and endocytosis of T cell surface T cell-B cell activating molecule/CD40-L. Potential role in regulating helper effector function. J Immunol 152(2):598–608
Frentsch M, Arbach O, Kirchhoff D, Moewes B et al (2005) Direct access to CD4+ T cells specific for defined antigens according to CD154 expression. Nat Med 11(10):1118–1124. https://doi.org/10.1038/nm1292
Watts TH (2005) TNF/TNFR family members in costimulation of T cell responses. Annu Rev Immunol 23:23–68. https://doi.org/10.1146/annurev.immunol.23.021704.115839
Schoenbrunn A, Frentsch M, Kohler S, Keye J et al (2012) A converse 4-1BB and CD40 ligand expression pattern delineates activated regulatory T cells (Treg) and conventional T cells enabling direct isolation of alloantigen-reactive natural Foxp3+ Treg. J Immunol 189(12):5985–5994. https://doi.org/10.4049/jimmunol.1201090
Campion SL, Brodie TM, Fischer W, Korber BT et al (2014) Proteome-wide analysis of HIV-specific naive and memory CD4(+) T cells in unexposed blood donors. J Exp Med 211(7):1273–1280. https://doi.org/10.1084/jem.20130555
Su LF, Kidd BA, Han A, Kotzin JJ, Davis MM (2013) Virus-specific CD4(+) memory-phenotype T cells are abundant in unexposed adults. Immunity 38(2):373–383. https://doi.org/10.1016/j.immuni.2012.10.021
Nauerth M, Stemberger C, Mohr F, Weissbrich B et al (2016) Flow cytometry-based TCR-ligand Koff -rate assay for fast avidity screening of even very small antigen-specific T cell populations ex vivo. Cytometry A 89(9):816–825. https://doi.org/10.1002/cyto.a.22933
Acknowledgements
This work was supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy (EXC 2167-390884018, Excellence Cluster Precision Medicine in Chronic Inflammation) to P.B., A.S.; DFG grant n. 433038070 to P.B., A.S.; RU5042 – miTarget to P.B., and CRC/TR 241 to A.S. The German Federal Ministry of Education and Science (BMBF)—Project InfectControl 2020 AnDiPath (Fkz 03ZZ0838B) to A.S., the European Crohn and Colitis Organisation (ECCO) to P.B.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Saggau, C., Scheffold, A., Bacher, P. (2021). Flow Cytometric Characterization of Human Antigen-Reactive T-Helper Cells. In: Annunziato, F., Maggi, L., Mazzoni, A. (eds) T-Helper Cells. Methods in Molecular Biology, vol 2285. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1311-5_12
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1311-5_12
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1310-8
Online ISBN: 978-1-0716-1311-5
eBook Packages: Springer Protocols