Abstract
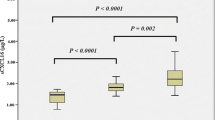
The pathological effects of thrombospondin-1 (TSP-1) have been studied in many preclinical tumor models and rheumatoid arthritis. However, the role of TSP-1 and anti-thrombospondin-1 autoantibodies (ATSA) in systemic lupus erythematosus (SLE) has not been specifically defined. In this study, we investigated the clinical relevance and functional significance of TSP-1 and ATSA in SLE patients. Serum levels of TSP-1 and ATSA were measured by ELISA in 138 adult SLE patients and 60 healthy controls. Blood cell counts, rheumatoid factor (RF), immunoglobulins, erythrocyte sedimentation rate (ESR), complements, and SLE-related autoantibodies were measured by standard laboratory techniques. Disease activity was assessed by systemic lupus erythematosus disease activity index (SLEDAI). TSP-1 concentrations were significantly lower in SLE patients compared with those in healthy controls. A significant difference of TSP-1 was observed in the patients with serositis, C3 decrease, RF positive, leukocytopenia, and thrombocytopenia. The levels of TSP-1 showed a positive correlation with the number of leukocyte and thrombocyte, while a negative correlation with anti-dsDNA antibody, IgG, ESR, and SLEDAI. ATSA was observed in 58.7% (81/138) of SLE patients, which was significantly higher than that in healthy controls (7/60, p < 0.05). Patients with active SLE showed higher ATSA positivity (67.1%) than those with inactive disease (47.1%, p < 0.05). ATSA was positively correlated with anti-rRNP antibody, IgG, total protein, and C4. This study revealed the opposite clinical relevance of TSP-1 and its autoantibody in SLE for the first time. TSP-1 may play an anti-inflammatory and immunoregulatory role in SLE autoimmunity. ATSA increased more frequently in disease-active patients and was associated with more severe clinical manifestations, which implicated its antagonistic role on TSP-1 and its involvement in the pathogenesis of SLE.

Similar content being viewed by others
References
Lawler J, Slayter H, Coligan JE (1978) Isolation and characterization of a high molecular weight glycoprotein from human blood platelets. J Biol Chem 253:8609–8616
Kuznetsova SA, Roberts DD (2004) Functional regulation of T lymphocytes by modulatory extracellular matrix proteins. Int J Biochem Cell Biol 36:1126–1134
Sarfati M, Fortin G, Raymond M, Susin S (2008) CD47 in the immune response: role of thrombospondin and SIRP-alpha reverse signaling. Curr Drug Targets 9:842–850
Wight TN, Raugi G, Mumby SM, Bornstein P (1985) Light microscopic immunolocation of thrombospondin in human tissues. J Histochem Cytochem 33:295–302
Naganuma H, Satoh H, Asahara T et al (2004) Quantification of thrombospondin-1 secretion and expression of αvβ3 and α3β1 integrins and syndecan-1 as cell-surface receptors for thrombospondin-1 in malignant glioma cells. J Neuro-Oncol 70:309–317
Lopez-Dee Z, Pidcock K, Gutierrez LS (2011) Trombospondin-1: multiple paths to inflammation. Mediat Inflamm 2011:296069
Grimbert P, Bouguermouh S, Baba N et al (2006) Thrombospondin/CD47 interaction: a pathway to generate regulatory T cells-from human CD4+ CD25- T cells in response to inflammation. J Immunol 177(6):3534–3541
Doyen V, Rubio M, Braun D, Nakajima T, Abe J, Saito H, Delespesse G, Sarfati M (2003) Thrombospondin 1 is an autocrine negative regulator of human dendritic cell activation. J Exp Med 198(8):1277–1283
Moulton VR, Tsokos GC (2015) T cell signaling abnormalities contribute to aberrant immune cell function and autoimmunity. J Clin Invest 125:2220–2227
Crow MK (2014) Advances in understanding the role of type I interferons in systemic lupus erythematosus. Curr Opin Rheumatol 26:467–474
Jung JY, Suh CH (2015) Incomplete clearance of apoptotic cells in systemic lupus erythematosus: pathogenic role and potential biomarker. Int J Rheum Dis 18:294–303
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40:1725
Bombardier C, Gladman D, Urowitz MB, Caron D, Chang CH (1992) Derivation of the SLEDAI: a disease activity index for lupus patients. The committee on prognosis studies in SLE. Arthritis Rheum 35:630–640
Xia YP, Li B, Hylton D, Detmar M, Yancopoulos GD, Rudge JS (2003) Transgenic delivery of VEGF to mouse skin leads to an inflammatory condition resembling human psoriasis. Blood 102:161–168
Isenberg JS, Ridnour LA, Dimitry J, Frazier WA, Wink DA, Roberts DD (2006) CD47 is necessary for inhibition of nitric oxide-stimulated vascular cell responses by thrombospondin-1. J Biol Chem 281:26069–26080
Isenberg JS, Ridnour L, Thomas DD, Wink DA, Roberts DD, Espey MG (2006) Guanylyl cyclase-dependent chemotaxis of endothelial cells in response to nitric oxide gradients. Free Radic Biol Med 40:1028–1033
Isenberg JS, Frazier WA, Roberts DD (2008) Thrombospondin-1: a physiological regulator of nitric oxide signaling. Cell Mol Life Sci 65:728–742
Crawford SE, Stellmach V, Murphy-Ullrich JE, Ribeiro SMF, Lawler J, Hynes RO, Boivin GP, Bouck N (1998) Thrombospondin-1 is a major activator of TGF-β1 in vivo. Cell 93:1159–1170
Young GD, Murphy-Ullrich J (2004) The tryptophan-rich motifs of the thrombospondin type 1 repeats bind VLAL motifs in the latent transforming growth factor-β complex. J Biol Chem 279:47633–47642
Schultz-Cherry S, Chen F, Mosher DF et al (1995) Regulation of transforming growth factor-β activation by discrete sequences of thrombospondin 1. J Biol Chem 270:7304–7310
Contreras-Ruiz L, Masli S (2015) Immunomodulatory cross-talk between conjunctival goblet cells and dendritic cells. PLoS One 10(3):e0120284
Mir F, Contreras-Ruiz L, Masli S (2015) Thrombospondin-1 dependent immune regulation by TGFβ2-exposed antigen presenting cells. Immunology 146(4):547–556
Cazac BB, Roes J (2000) TGF-β receptor controls B cell responsiveness and induction of IgA in vivo. Immunity 13:443–451
Lievens D, Habets KL, Robertson AK, Laouar Y, Winkels H, Rademakers T, Beckers L, Wijnands E, Boon L, Mosaheb M, Ait-Oufella H, Mallat Z, Flavell RA, Rudling M, Binder CJ, Gerdes N, Biessen EAL, Weber C, Daemen MJAP, Kuiper J, Lutgens E (2013) Abrogated transforming growth factor β receptor II (TGFβRII) signalling in dendritic cells promotes immune reactivity of T cells resulting in enhanced atherosclerosis. Eur Heart J 34:3717–3727
Ramalingam R, Larmonier CB, Thurston RD, Midura-Kiela MT, Zheng SG, Ghishan FK, Kiela PR (2012) Dendritic cell-specific disruption of TGF-β receptor II leads to altered regulatory T cell phenotype and spontaneous multiorgan autoimmunity. J Immunol 189:3878–3893
Avice MN, Rubio M, Sergerie M, Delespesse G, Sarfati M (2001) Role of CD47 in theinduction of human naive T cell anergy. J Immunol 167(5):2459–2468
Boilard E, Blanco P, Nigrovic PA (2012) Platelets: active players in the pathogenesis of arthritis and SLE. Nat Rev Rheumatol 8(9):534–542
Lood C, Tydén H, Gullstrand B, Nielsen C.T., Heegaard N.H.H., Linge P., Jönsen A., Hesselstrand R., Kahn R., Bengtsson A.A. (2016) Decreased platelet size is associated with platelet activation and anti-phospholipid syndrome in systemic lupus erythematosus. Rheumatology (Oxford)
Lood C, Tydén H, Gullstrand B, Sturfelt G, Jönsen A, Truedsson L, Bengtsson AA (2014) Platelet activation and anti-phospholipid antibodies collaborate in the activation of the complement system on platelets in systemic lupus erythematosus. PLoS One 9(6):e99386
Ohyama K, Kawakami A, Tamai M, Baba M, Kishikawa N, Kuroda N (2012) Serum immune complex containing thrombospondin-1: a novel biomarker for early rheumatoid arthritis. Ann Rheum Dis 71(11):1916–1917
Funding
This work was supported by National Natural Science Foundation of China (31530020, 81471601, 81671602), Beijing Municipal Science and Technology Commission (Z171100000417007), Peking University Clinical Medicine + X Project for Joint Investigation of Youth and Beijing Nova Program (Z171100001117025).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
None.
Electronic supplementary material
ESM 1
(DOC 2548 kb)
Rights and permissions
About this article
Cite this article
Meng, Y., Zhang, M., Zhao, X. et al. Decreased serum thrombospondin-1 and elevation of its autoantibody are associated with multiple exacerbated clinical manifestations in systemic lupus erythematosus. Clin Rheumatol 37, 2707–2714 (2018). https://doi.org/10.1007/s10067-018-4188-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-018-4188-x