Abstract
Purpose
Supplementation of growth hormone (GH) during controlled ovarian stimulation (COS) has been suggested to improve ovarian response. Despite potential benefits in poor responders, multiple injections of GH during COS are inconvenient. We conducted a randomized controlled study to evaluate the efficacy and safety of sustained-release human GH in poor responders undergoing in vitro fertilization (IVF).
Methods
This was a single-center, randomized, open-label, parallel study. Infertile women who satisfied the Bologna criteria for poor responders were randomized into GH treatment and control groups. The treatment group received a sustained-release GH (Eutropin Plus® 20 mg) three times before and during COS (mid-luteal, late luteal, and menstrual cycle day 2). The baseline characteristics and IVF outcomes were compared between the two groups.
Results
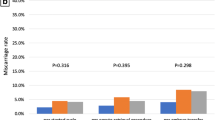
A total of 127 patients were included in the analysis. The mean age was 39.6 years and mean anti-Müllerian hormone level was 0.6 ng/ml. There was no significant difference in the baseline characteristics between GH treatment and control groups. The number of follicles on the human chorionic gonadotropin triggering day (3.1 ± 2.3 vs. 2.4 ± 1.6, P = 0.043) and the proportion of metaphase II oocytes (67.5 vs. 52.3%, P = 0.030) were higher in the GH group than in controls. The percentage of clinical and ongoing pregnancy and miscarriage was not different between the two groups.
Conclusion
Supplementation of sustained-release GH before and during COS improved ovarian response, with an increase in mature oocytes in poor responders. Further studies are needed to ensure this benefit in general infertility patients.

Similar content being viewed by others
References
Practice Committee of the American Society for Reproductive Medicine (2015) Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril 103:e9–e17
Cooper GS, Baird DD, Hulka BS, Weinberg CR, Savitz DA, Hughes CL (1995) Follicle-stimulating hormone concentrations in relation to active and passive smoking. Obstet Gynecol 85:407–411
Cooper GS, Thorp JM (1999) FSH levels in relation to hysterectomy and to unilateral oophorectomy. Obstet Gynecol 94:969–972
van Dorp W, Blijdorp K, Laven JS, Pieters R, Visser JA, van der Lely AJ, Neggers SJ, van den Heuvel-Eibrink MM (2013) Decreased ovarian function is associated with obesity in very long-term female survivors of childhood cancer. Eur J Endocrinol 168:905–912
Pandian Z, McTavish AR, Aucott L, Hamilton MP, Bhattacharya S (2010) Interventions for ‘poor responders’ to controlled ovarian hyper stimulation (COH) in in vitro fertilisation (IVF). Cochrane Database Syst Rev 1:CD004379
Tarlatzis BC, Zepiridis L, Grimbizis G, Bontis J (2003) Clinical management of low ovarian response to stimulation for IVF: a systematic review. Hum Reprod Update 9:61–76
Duffy JM, Ahmad G, Mohiyiddeen L, Nardo LG, Watson A (2010) Growth hormone for in vitro fertilization. Cochrane Database Syst Rev 1:CD000099
Li XL, Wang L, Lv F, Huang XM, Wang LP, Pan Y, Zhang XM (2017) The influence of different growth hormone addition protocols to poor ovarian responders on clinical outcomes in controlled ovary stimulation cycles: a systematic review and meta-analysis. Medicine 96:e6443
Saenger PH, Mejia-Corletto J (2016) Long-acting growth hormone: an update. Endocr Dev 30:79–97
Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L, EwgoPOR Definition (2011) ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod 26:1616–1624
Hwang JS, Lee HS, Chung WY, Han HS, Jin DK, Kim HS, Ko CW, Lee BC, Lee DY, Lee KH, Shin JH, Suh BK, Yoo HW, Ji HJ, Lee JH, Bae YJ, Kim DH, Yang SW (2013) Efficacy and safety of LB03002, a once-weekly sustained-release human GH for 12-month treatment in Korean children with GH deficiency. Eur J Endocrinol 169:179–185
Bassiouny YA, Dakhly DMR, Bayoumi YA, Hashish NM (2016) Does the addition of growth hormone to the in vitro fertilization/intracytoplasmic sperm injection antagonist protocol improve outcomes in poor responders? A randomized, controlled trial. Fertil Steril 105:697–702
Kim C-H, Park H, Moon J-W, Ahn J-W, Kim S-H, Chae H-D (2015) Efficacy of long-acting growth hormone, eutropin plus single injection on controlled ovarian stimulation and IVF outcomes in poor responders undergoing IVF. In: Abstract book. annual meeting of Korean Society of Obstetrics and Gynecology, vol 101, p 355
Weall BM, Al-Samerria S, Conceicao J, Yovich JL, Almahbobi G (2015) A direct action for GH in improvement of oocyte quality in poor-responder patients. Reproduction 149:147–154
Hull KL, Harvey S (2014) Growth hormone and reproduction: a review of endocrine and autocrine/paracrine interactions. Int J Endocrinol 2014:234014
Webb R, Garnsworthy PC, Gong JG, Armstrong DG (2004) Control of follicular growth: local interactions and nutritional influences. J Anim Sci 82 E-Suppl:E63–E74
Yoshimura Y, Nakamura Y, Koyama N, Iwashita M, Adachi T, Takeda Y (1993) Effects of growth hormone on follicle growth, oocyte maturation, and ovarian steroidogenesis. Fertil Steril 59:917–923
Yoshimura Y, Ando M, Nagamatsu S, Iwashita M, Adachi T, Sueoka K, Miyazaki T, Kuji N, Tanaka M (1996) Effects of insulin-like growth factor-I on follicle growth, oocyte maturation, and ovarian steroidogenesis and plasminogen activator activity in the rabbit. Biol Reprod 55:152–160
Qiao J, Feng HL (2011) Extra- and intra-ovarian factors in polycystic ovary syndrome: impact on oocyte maturation and embryo developmental competence. Hum Reprod Update 17:17–33
Schoyer KD, Liu HC, Witkin S, Rosenwaks Z, Spandorfer SD (2007) Serum insulin-like growth factor I (IGF-I) and IGF-binding protein 3 (IGFBP-3) in IVF patients with polycystic ovary syndrome: correlations with outcome. Fertil Steril 88:139–144
Karamouti M, Kollia P, Kallitsaris A, Vamvakopoulos N, Kollios G, Messinis IE (2008) Growth hormone, insulin-like growth factor I, and leptin interaction in human cultured lutein granulosa cells steroidogenesis. Fertil Steril 90:1444–1450
Kolibianakis EM, Venetis CA, Diedrich K, Tarlatzis BC, Griesinger G (2009) Addition of growth hormone to gonadotrophins in ovarian stimulation of poor responders treated by in vitro fertilization: a systematic review and meta-analysis. Hum Reprod Update 15:613–622
Du XF, Yang XH, Li J, Hao M, Guo YH (2016) Growth hormone co-treatment within a GnRH agonist long protocol improves implantation and pregnancy rates in patients undergoing IVF-ET. Arch Gynecol Obstet 294:877–883
Yovich JL, Stanger JD (2010) Growth hormone supplementation improves implantation and pregnancy productivity rates for poor-prognosis patients undertaking IVF. Reprod Biomed Online 21:37–49
de Ziegler D, Streuli I, Meldrum DR, Chapron C (2011) The value of growth hormone supplements in ART for poor ovarian responders. Fertil Steril 96:1069–1076
Tesarik J, Hazout A, Mendoza C (2005) Improvement of delivery and live birth rates after ICSI in women aged > 40 years by ovarian co-stimulation with growth hormone. Hum Reprod 20:2536–2541
Bayoumi YA, Dakhly DM, Bassiouny YA, Hashish NM (2015) Addition of growth hormone to the microflare stimulation protocol among women with poor ovarian response. Int J Gynecol Obstet 131:305–308
Eftekhar M, Aflatoonian A, Mohammadian F, Eftekhar T (2013) Adjuvant growth hormone therapy in antagonist protocol in poor responders undergoing assisted reproductive technology. Arch Gynecol Obstet 287:1017–1021
Kucuk T, Kozinoglu H, Kaba A (2008) Growth hormone co-treatment within a GnRH agonist long protocol in patients with poor ovarian response: a prospective, randomized, clinical trial. J Assist Reprod Genet 25:123–127
Yoon TK, Kang IS, Koong MK, Kim YS, Kim JY, Kim MJ (2017) Effect of supplementary grow hormone in poor responders. In: Poster presentation. The 72th biannual meeting of Korean Society of Obstetrics and Gynecology
Huang ZH, Baxter RC, Hughes SM, Matson PL, Lieberman BA, Morris ID (1993) Supplementary growth hormone treatment of women with poor ovarian response to exogenous gonadotrophins: changes in serum and follicular fluid insulin-like growth factor-1 (IGF-1) and IGF binding protein-3 (IGFBP-3). Hum Reprod 8:850–857
Author information
Authors and Affiliations
Contributions
YSK: Project development, data collection, and supervision of the entire study process; SAC: data analysis and manuscript writing; MJK: data collection and manuscript review; HJK: data analysis; JK: data collection and manuscript review; EMC: data collection and project administration; JYK: data collection and analysis; HMP: data collection; SWL: project development and manuscript review; WSL: data collection and project administration; TKY: project administration and data collection.
Corresponding author
Ethics declarations
Funding
This study was supported by a research Grant from LG Life Sciences, Seoul, Korea.
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
All procedures in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study protocol was approved by the institutional review board (IRB number: GCI-13-37). Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Choe, SA., Kim, M.J., Lee, H.J. et al. Increased proportion of mature oocytes with sustained-release growth hormone treatment in poor responders: a prospective randomized controlled study. Arch Gynecol Obstet 297, 791–796 (2018). https://doi.org/10.1007/s00404-017-4613-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-017-4613-4