Abstract
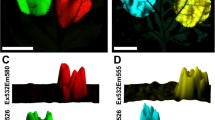
The effect of the type of leaf tissue selected for the study of green fluorescent protein (GFP) fluorescence intensity was investigated here using the T1 generation of transgenic tobacco expressing the m-gfp5-ER gene. The fluorescence of GFP was detected by fluorescence binocular microscope coupled with the CCD camera and quantified by means of image analyses using the Lucia® software. Mean brightness values from various leaf tissues were compared. First, an original data revealing the significant differences in the fluorescence intensity between the abaxial and adaxial surfaces are given. Stronger signal was detected on the abaxial side. Subsequently, the effect of the tissue location within the leaf surface was investigated and higher fluorescence was detected on the samples detached from leaf tips. Finally, the effect of the physiological age of leaves was studied using the in vitro clonally propagated plants. Leaves from the analogous positions within the plant body of three clones were investigated. The decrease in the fluorescence towards the plant top (youngest leaves) was observed in all studied plants. Surprisingly, the variability of the fluorescence within the clones of studied genotype was high enough to conclude, that the fluorescence of each individual is unique and affected by particular genotype and environment. Our study showed that the origin of leaf tissue selected for the GFP quantification is crucial and that the fluctuations in the fluorescence intensity should be taken into account when comparing the GFP fluorescence patterns of different plants. Moreover, the degree of fluorescence variability seems to be individually affected.







Similar content being viewed by others
References
Benfey PN, Chua N-H (1989) Regulated genes in transgenic plants. Science 244:174–181. doi:10.1126/science.244.4901.174
Blumenthal A, Kuznetzova L, Edelbaum O, Raskin V, Levy M, Sela I (1999) Measurement of green fluorescence protein in plants: quantification, correlation to expression, rapid screening and differential gene expression. Plant Sci 142:93–99. doi:10.1016/S0168-9452(98)00249-0
Cho M-J, Choi H-W, Jiang W, Ha ChD, Lemaux PG (2002) Endosperm-specific expression of green fluorescent protein driven by the hordein promoter is stably inherited in transgenic barley (Hordeum vulgare) plants. Physiol Plant 115:144–154. doi:10.1034/j.1399-3054.2002.1150117.x
Doyle JJ, Doyle JL (1990) A rapid total DNA preparation procedure for fresh plant tissue. Focus 12:13–15
Eady CC, Weld RJ, Lister CE (2000) Agrobacterium tumefaciens-mediated transformation and transgenic-plant regeneration of onion (Allium cepa L.). Plant Cell Rep 19:376–381. doi:10.1007/s002990050743
Halfhill MD, Millwood RJ, Rufty TW, Weissinger AK, Stewart CN (2003) Spatial and temporal patterns of green fluorescent protein (GFP) in transgenic oilseed rape, Brassica napus L. Plant Cell Rep 22:338–343. doi:10.1007/s00299-003-0696-4
Halfhill MD, Richards HA, Mabon SA, Stewart CN Jr (2001) Expression of GFP and Bt transgenes in Brassica napus and hybridization with Brassica rapa. Theor Appl Genet 103:659–667. doi:10.1007/s001220100613
Harper BK, Stewart CN Jr (2000) Patterns of green fluorescenct protein expression in transgenic plants. Plant Mol Biol Rep 18:141a–141i
Horsch RB, Fry JE, Hoffmann NL, Eicholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Hraška M, Rakouský S, Kocábek T (2005) Use of simple semiquantitative method for appraisal of green fluorescent protein gene expression in transgenic tobacco plants. Biol Plant 49:313–316.doi:10.1007/s10535-005-3316-z
Hraška M, Rakouský S, Čurn V (2006) Green fluorescent protein as a vital marker for non-destructive detection of transformation events in transgenic plants. Plant Cell Tissue Organ Cult 86:303–318. doi:10.1007/s11240-006-9131-1
Laemmli UK (1973) Cleavage of structural proteins during assembly of head bacterophage-T4. Nature 227: 680–685. doi:10.1038/227680a0
Ponappa T, Brzozowski AE, Finer JJ (1999) Transient expression and stable transformation of soybean using the jellyfish green fluorescent protein. Plant Cell Rep 19:6–12. doi:10.1007/s002990050702
Preťová A, Obert B, Wetzstein HY (2001) Leaf developmental stage and tissue location affect the detection of β-glucuronidase in transgenic tobacco plants. Biotech Lett 23:555–558. doi:10.1023/A:1010311524048
Rooke L, Byrne D, Salgueiro S (2000) Marker gene expression driven by the maize ubiquitin promoter in transgenic wheat. Ann Appl Biol 136:167–172. doi:10.1111/j.1744-7348.2000.tb00022.x
Sunilkumar G, Mohr L-A, Lopata-Finch E, Emani Ch, Rathore KS (2002) Developmental and tissue specific expression of CaMV 35S promoter in cotton as revealed by GFP. Plant Mol Biol 50:463–474. doi:10.1023/A:1019832123444
Šesták Z (1971) Determination of chlorophyll a and b. In: Šesták Z, Čatský J, Jarvis PG (eds) Plant photosynthetis production. Manual and methods. Dr. W. Jung N. V. Publ., The hague 1971, pp 672–701
Zhou X, Carranco R., Vitha S, Hall TC (2005) The dark side of green fluorescent protein. New Phytol 168:313–322. doi:10.1111/j.1469-8137.2005.01489.x
Acknowledgments
The authors gratefully acknowledge the financial support received from the Ministry of Education, Youth and Sport of the Czech Republic (grants 1M06030, 1PO5ME800 and MSM 60076658–06). M. H. and V. H. received the support from grant GA ČR 31/H160 provided by the Grant Agency of the Czech Republic and both contributed to this work equally. The authors thank Dr. Jan Bárta (Department Plant Production, Faculty of Agriculture, University of South Bohemia) for his valuable advice and guidance in the part of this work concerning the proteins. Whole manuscript was kindly revised by Mentewab Ayalew.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Atanassov.
Rights and permissions
About this article
Cite this article
Hraška, M., Heřmanová, V., Rakouský, S. et al. Sample topography and position within plant body influence the detection of the intensity of green fluorescent protein fluorescence in the leaves of transgenic tobacco plants. Plant Cell Rep 27, 67–77 (2008). https://doi.org/10.1007/s00299-007-0431-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-007-0431-7