Abstract
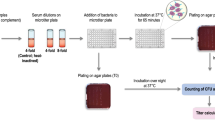
Serum bactericidal antibody (SBA) assays measure functional antibody titers against Neisseria meningitidis in sera. Induction of complement-dependent SBA after vaccination with meningococcal polysaccharide or conjugate or protein based vaccines is regarded as the surrogate of protection and thus acceptable evidence of the potential efficacy of these vaccines. This chapter discusses and details SBA assay protocols for measuring the complement-mediated lysis of serogroup A, B, C, W, X, and Y meningococci by human sera, for example, following vaccination or disease.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Chen WH, Neuzil KM, Boyce CR et al (2018) Safety and immunogenicity of a pentavalent meningococcal conjugate vaccine containing serogroups A, C, Y, W, and X in healthy adults: a phase 1, single-centre, double-blind, randomised, controlled study. Lancet Infect Dis 18:1088–1096
Feavers IM, Maiden MCJ (2017) Recent progress in the prevention of serogroup B meningococcal disease. Clin Vaccine Immunol 24. https://doi.org/10.1128/CVI.00566-16. pii: e00566-16
Goldschneider I, Gotschlich EC, Artenstein MS (1969) Human immunity to the meningococcus. I. The role of humoral antibodies. J Exp Med 129:1307–1326
Goldschneider I, Gotschlich EC, Artenstein MS (1969) Human immunity to the meningococcus II. Development of natural immunity. J Exp Med 129:1327–1348
Borrow R, Miller E (2006) Surrogates of protection. In: Frosch M, Maiden M (eds) Handbook of meningococcal disease. Wiley-VCH, Weinheim, pp 323–351
World Health Organization (1976) Requirements for meningococcal polysaccharide vaccine. World Health Organization technical report series, no. 594. World Health Organization, Geneva
Maslanka SE, Gheesling LL, LiButti DE et al (1997) Standardization and a multilaboratory comparison of Neisseria meningitidis serogroup A and C serum bactericidal assays. Clin Diag Lab Immunol 4:156–167
Borrow R, Aaberge IS, Santos GF et al (2005) Interlaboratory standardization of the measurement of serum bactericidal activity using human complement against meningococcal serogroup B, strain 44/76-SL before and after vaccination with the Norwegian MenBvac OMV vaccine. Clin Diag Lab Immunol 12:970–976
Zollinger WD, Mandrell RE (1983) Importance of complement source in bactericidal activity of human antibody and murine monoclonal antibody to meningococcal group B polysaccharide. Infect Immun 40:257–264
Borrow R, Findlow J, Gray S, Taylor S, Kaczmarski E (2014) Practical aspects and experience of safe working with Neisseria meningitidis. J Infect 68:305–312
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Lucidarme, J., Louth, J., Townsend-Payne, K., Borrow, R. (2019). Meningococcal Serogroup A, B, C, W, X, and Y Serum Bactericidal Antibody Assays. In: Seib, K., Peak, I. (eds) Neisseria meningitidis. Methods in Molecular Biology, vol 1969. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-9202-7_12
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9202-7_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-9201-0
Online ISBN: 978-1-4939-9202-7
eBook Packages: Springer Protocols