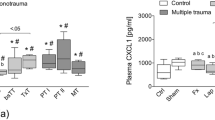
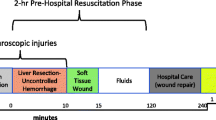
Abstract
Severe injury and shock remain major sources of morbidity and mortality worldwide. Immunologic dysregulation following trauma contributes to these poor outcomes. Few, if any, therapeutic interventions have benefited these patients, and this is due to our limited understanding of the host response to injury and shock. The Food and Drug Administration requires preclinical animal studies prior to any interventional trials in humans; thus, animal models of injury and shock will remain the mainstay for trauma research. However, adequate animal models that reflect the severe response to trauma in both the acute and subacute phases have been limited. Here we describe a novel murine model of polytrauma and shock that combines hemorrhagic shock, cecectomy, long bone fracture, and soft-tissue damage. This model produces an equivalent Injury Severity Score associated with adverse outcomes in humans, and may better recapitulate the human leukocyte, cytokine, transcriptomic, and overall inflammatory response following injury and hemorrhagic shock.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Pfeifer R, Tarkin IS, Rocos B, Pape HC (2009) Patterns of mortality and causes of death in polytrauma patients—has anything changed? Injury 40:907–911
Probst C, Pape HC, Hildebrand F, Regel G, Mahlke L, Giannoudis P, Krettek C, Grotz MR (2009) 30 years of polytrauma care: an analysis of the change in strategies and results of 4849 cases treated at a single institution. Injury 40:77–83
Probst C, Zelle BA, Sittaro NA, Lohse R, Krettek C, Pape HC (2009) Late death after multiple severe trauma: when does it occur and what are the causes? J Trauma 66(4):1212–1217
Sasser SM, Varghese M, Joshipura M, Kellermann A (2006) Preventing death and disability through the timely provision of prehospital trauma care. Bull World Health Organ 84:507
Gentile LF, Cuenca AG, Efron PA, Ang D, Bihorac A, McKinley BA, Moldawer LL, Moore FA (2012) Persistent inflammation and immunosuppression: a common syndrome and new horizon for surgical intensive care. J Trauma Acute Care Surg 72:1491–1501
Robertson CM, Coopersmith CM (2006) The systemic inflammatory response syndrome. Microbes Infect 8:1382–1389
Ward NS, Casserly B, Ayala A (2008) The compensatory anti-inflammatory response syndrome (CARS) in critically ill patients. Clin Chest Med 29:617–625. viii
Rosenthal MD, Moore FA (2015) Persistent inflammatory, immunosuppressed, catabolic syndrome (PICS): a new phenotype of multiple organ failure. J Adv Nutr Hum Metab 1:e874
Mira JC, Gentile LF, Mathias BJ, Efron PA, Brakenridge SC, Mohr AM, Moore FA, Moldawer LL (2017) Sepsis pathophysiology, chronic critical illness, and persistent inflammation-immunosuppression and catabolism syndrome. Crit Care Med 45:253–262
Mathias B, Delmas AL, Ozrazgat-Baslanti T, Vanzant EL, Szpila BE, Mohr AM, Moore FA, Brakenridge SC, Brumback BA, Moldawer LL et al (2016) Human myeloid-derived suppressor cells are associated with chronic immune suppression after severe sepsis/septic shock. Ann Surg 265:827–834
Stahel PF, Smith WR, Moore EE (2007) Role of biological modifiers regulating the immune response after trauma. Injury 38:1409–1422
Artenstein AW, Higgins TL, Opal SM (2013) Sepsis and scientific revolutions. Crit Care Med 41:2770–2772
Seok J, Warren HS, Cuenca AG, Mindrinos MN, Baker HV, Xu W, Richards DR, McDonald-Smith GP, Gao H, Hennessy L et al (2013) Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc Natl Acad Sci U S A 110:3507–3512
Takao K, Miyakawa T (2015) Genomic responses in mouse models greatly mimic human inflammatory diseases. Proc Natl Acad Sci U S A 112:1167–1172
Deitch EA (1998) Animal models of sepsis and shock: a review and lessons learned. Shock 9:1–11
Efron PA, Mohr AM, Moore FA, Moldawer LL (2015) The future of murine sepsis and trauma research models. J Leukoc Biol 98:945–952
Pratt D (1980) Alternatives to pain in experiments on animals. Argus Archives, New York
Tsukamoto T, Pape HC (2009) Animal models for trauma research: what are the options? Shock 31:3–10
Frink M, Andruszkow H, Zeckey C, Krettek C, Hildebrand F (2011) Experimental trauma models, an update. J Biomed Biotechnol 2011:797383
Noble RL, Collip JB (1942) A quantitative method for the production of experimental traumatic shock without haemorrhage in unanaesthetized animals. Q J Exp Physiol 31:187–199
Gill R, Ruan X, Menzel CL, Namkoong S, Loughran P, Hackam DJ, Billiar TR (2011) Systemic inflammation and liver injury following hemorrhagic shock and peripheral tissue trauma involve functional TLR9 signaling on bone marrow-derived cells and parenchymal cells. Shock 35:164–170
Kang SC, Matsutani T, Choudhry MA, Schwacha MG, Rue LW, Bland KI, Chaudry IH (2004) Are the immune responses different in middle-aged and young mice following bone fracture, tissue trauma and hemorrhage? Cytokine 26:223–230
Matsutani T, Kang SC, Miyashita M, Sasajima K, Choudhry MA, Bland KI, Chaudry IH (2007) Young and middle-age associated differences in cytokeratin expression after bone fracture, tissue trauma, and hemorrhage. Am J Surg 193:61–68
Matsutani T, Kang SC, Miyashita M, Sasajima K, Choudhry MA, Bland KI, Chaudry IH (2007) Liver cytokine production and ICAM-1 expression following bone fracture, tissue trauma, and hemorrhage in middle-aged mice. Am J Physiol Gastrointest Liver Physiol 292:G268–G274
Venet F, Chung CS, Huang X, Lomas-Neira J, Chen Y, Ayala A (2009) Lymphocytes in the development of lung inflammation: a role for regulatory CD4+ T cells in indirect pulmonary lung injury. J Immunol 183:3472–3480
Wichmann MW, Ayala A, Chaudry IH (1998) Severe depression of host immune functions following closed-bone fracture, soft-tissue trauma, and hemorrhagic shock. Crit Care Med 26:1372–1378
Wang P, Ba ZF, Burkhardt J, Chaudry IH (1993) Trauma-hemorrhage and resuscitation in the mouse—effects on cardiac-output and organ blood-flow. Am J Physiol 264:H1166–H1173
Hollenberg SM (2005) Mouse models of resuscitated shock. Shock 24(Suppl 1):58–63
Marshall JC, Deitch E, Moldawer LL, Opal S, Redl H, van der Poll T (2005) Preclinical models of shock and sepsis: what can they tell us? Shock 24(Suppl 1):1–6
Xiao W, Mindrinos MN, Seok J, Cuschieri J, Cuenca AG, Gao H, Hayden DL, Hennessy L, Moore EE, Minei JP et al (2011) A genomic storm in critically injured humans. J Exp Med 208:2581–2590
Gentile LF, Nacionales DC, Lopez MC, Vanzant E, Cuenca A, Cuenca AG, Ungaro R, Baslanti TO, McKinley BA, Bihorac A et al (2014) A better understanding of why murine models of trauma do not recapitulate the human syndrome. Crit Care Med 42:1406–1413
Yue F, Cheng Y, Breschi A, Vierstra J, Wu W, Ryba T, Sandstrom R, Ma Z, Davis C, Pope BD et al (2014) A comparative encyclopedia of DNA elements in the mouse genome. Nature 515:355–364
Cuschieri J, Johnson JL, Sperry J, West MA, Moore EE, Minei JP, Bankey PE, Nathens AB, Cuenca AG, Efron PA et al (2012) Benchmarking outcomes in the critically injured trauma patient and the effect of implementing standard operating procedures. Ann Surg 255:993–999
Chute CG, Ullman-Cullere M, Wood GM, Lin SM, He M, Pathak J (2013) Some experiences and opportunities for big data in translational research. Genet Med 15:802–809
Kannry JL, Williams MS (2013) Integration of genomics into the electronic health record: mapping terra incognita. Genet Med 15:757–760
Dyson A, Singer M (2009) Animal models of sepsis: why does preclinical efficacy fail to translate to the clinical setting? Crit Care Med 37:S30–S37
Keel M, Trentz O (2005) Pathophysiology of polytrauma. Injury 36:691–709
Gauthier C, Griffin G (2005) Using animals in research, testing and teaching. Rev Sci Tech 24:735–745
Gentile LF, Nacionales DC, Cuenca AG, Armbruster M, Ungaro RF, Abouhamze AS, Lopez C, Baker HV, Moore FA, Ang DN et al (2013) Identification and description of a novel murine model for polytrauma and shock. Crit Care Med 41:1075–1085
Mira JC, Szpila BE, Nacionales DC, Lopez MC, Gentile LF, Mathias BJ, Vanzant EL, Ungaro R, Holden D, Rosenthal MD et al (2016) Patterns of gene expression among murine models of hemorrhagic shock/trauma and sepsis. Physiol Genomics 48:135–144
Baker SP, O’Neill B, Haddon W Jr, Long WB (1974) The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 14:187–196
Ayala A, Chung CS, Lomas JL, Song GY, Doughty LA, Gregory SH, Cioffi WG, LeBlanc BW, Reichner J, Simms HH et al (2002) Shock-induced neutrophil mediated priming for acute lung injury in mice: divergent effects of TLR-4 and TLR-4/FasL deficiency. Am J Pathol 161:2283–2294
Lomas JL, Chung CS, Grutkoski PS, LeBlanc BW, Lavigne L, Reichner J, Gregory SH, Doughty LA, Cioffi WG, Ayala A (2003) Differential effects of macrophage inflammatory chemokine-2 and keratinocyte-derived chemokine on hemorrhage-induced neutrophil priming for lung inflammation: assessment by adoptive cells transfer in mice. Shock 19:358–365
Council NR (2011) Guide for the care and use of laboratory animals, 8th edn. National Academies Press, Washington, DC
Cuenca AG, Delano MJ, Kelly-Scumpia KM, Moldawer LL, Efron PA (2010) Cecal ligation and puncture. In: Coligan JE et al (eds) Current protocols in immunology, Chapter 19, Unit 19 13. Wiley, New York
Stephan RN, Kuppr TS, Geha AS, Baue AE, Chaudry IH (1987) Hemorrhage without tissue trauma produces immunosuppression and enhances susceptibility to sepsis. Arch Surg 122:62–68
Monaghan SF, Thakkar RK, Heffernan DS, Huang X, Chung CS, Lomas-Neira J, Cioffi WG, Ayala A (2012) Mechanisms of indirect acute lung injury a novel role for the coinhibitory receptor, programmed death-1. Ann Surg 255:158–164
Bible LE, Pasupuleti LV, Gore AV, Sifri ZC, Kannan KB, Mohr AM (2015) Chronic restraint stress after injury and shock is associated with persistent anemia despite prolonged elevation in erythropoietin levels. J Trauma Acute Care Surg 79:91–97
Darwiche SS, Kobbe P, Pfeifer R, Kohut L, Pape HC, Billiar T (2011) Pseudofracture: an acute peripheral tissue trauma model. J Vis Exp (50):2074
Schwulst SJ, Trahanas DM, Saber R, Perlman H (2013) Traumatic brain injury-induced alterations in peripheral immunity. J Trauma Acute Care 75:780–788
Lpaktchi K, Mattar A, Niederbichler AD, Kim J, Hoesel LM, Hemmila MR, GL S, Remick DG, Wang SC, Arbabi S (2007) Attenuating burn wound inflammation improves pulmonary function and survival in a burn-pneumonia model. Crit Care Med 35:2139–2144
Acknowledgment
J.C.M., T.L.M., and B.M. were supported by a training grant in burn and trauma research from the National Institute of General Medical Sciences (NIGMS) (T32 GM-008721). This work was also supported by NIH Grants R01 GM-040586 and R01 GM-081923, awarded by the NIGMS. A.M.M. was supported by R01 GM-105893. In addition, P.A.E. was supported by P30 AG-028740 from the NIH National Institute on Aging and R01 GM113945 (NIGMS). Finally, P.A.E. and L.L.M. were supported by P50 GM-111152 (NIGMS).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
1 Electronic Polytrauma Supplementary Material
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Mira, J.C. et al. (2018). Mouse Injury Model of Polytrauma and Shock. In: Tharakan, B. (eds) Traumatic and Ischemic Injury. Methods in Molecular Biology, vol 1717. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7526-6_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7526-6_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7524-2
Online ISBN: 978-1-4939-7526-6
eBook Packages: Springer Protocols