Abstract
Background
Indocyanine green (ICG) fluorescence imaging has been used to detect many types of tumors during surgery; however, there are few studies on thymic masses and the dose and time of ICG injection have not been optimized.
Objective
We aimed to evaluate the optimal ICG injection dose and timing for detecting thymic masses during surgery.
Method
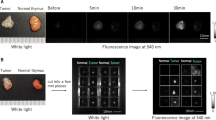
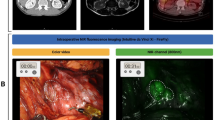
Forty-nine consecutive patients diagnosed with thymic masses on preoperative computed tomography (CT) and scheduled to undergo thymic cystectomy or thymectomy were included. Patients were administered 1, 2, or 5 mg/kg of ICG at different times. Thymic masses were observed during and after surgery using a near-infrared fluorescence imaging system, and the fluorescence signal tumor-to-normal ratio (TNR) was analyzed.
Results
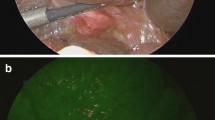
Among the 49 patients, 14 patients with thymic cysts showed negative fluorescence signals, 33 patients with thymoma or thymic carcinoma showed positive fluorescence signals, and 2 patients showed insufficient fluorescence signals. The diagnosis of thymic masses based on CT was correct in 32 (65%) of 49 cases; however, the differential diagnosis of thymic masses based on NIR signals was correct in 47 of 49 cases (96%), including 14 cases of thymic cysts (100%) and 33 cases of thymomas or thymic carcinomas (94%). In addition, TNR was not affected by the time or dose of ICG injection, histological type, stage, or tumor size.
Conclusions
Low-dose intravenous injection of ICG at flexible time can detect thymic tumors. In addition, thymic cysts can be distinguished from thymomas or thymic carcinomas during surgery by the absence of ICG fluorescence signals.





Similar content being viewed by others
Change history
18 March 2022
A Correction to this paper has been published: https://doi.org/10.1245/s10434-022-11663-5
References
Tomiyama N, Honda O, Tsubamoto M, et al. Anterior mediastinal tumors: diagnostic accuracy of CT and MRI. Eur J Radiol. 2009;69(2):280–8.
Kurup A, Loehrer PJ Sr. Thymoma and thymic carcinoma: therapeutic approaches. Clinical Lung Cancer. 2004;6(1):28–32.
Venuta F, Anile M, Diso D, et al. Thymoma and thymic carcinoma. Eur J Cardio-thoracic Surg. 2010;37(1):13–25.
Eng TY, Fuller CD, Jagirdar J, Bains Y, Thomas CR. Thymic carcinoma: state of the art review. Int J Radiation Oncology Biology Physics. 2004;59(3):654–64.
Weiss GJ. Thymic carcinoma: current and future therapeutic interventions. Exp Opinion on Investigat Drugs. 2010;19(8):1007–16.
Ettinger DS, Riely GJ, Akerley W, et al. Thymomas and thymic carcinomas: clinical practice guidelines in oncology. J Natl Comprehensive Cancer Netw. 2013;11(5):562–76.
Kent MS, Wang T, Gangadharan SP, Whyte RI. What is the prevalence of a “nontherapeutic” thymectomy? Ann Thorac Surg. 2014;97(1):276–82.
Kondo K. Optimal therapy for thymoma. The J Med Investig. 2008;55(1–2):17–28.
Kondo K, Monden Y. Therapy for thymic epithelial tumors: a clinical study of 1,320 patients from Japan. Ann Thorac Surg. 2003;76(3):878–84.
Masaoka A. Staging system of thymoma. J Thoracic Oncol. 2010;5(10 Suppl 4):S304-312.
Venuta F, Rendina EA, Anile M, de Giacomo T, Vitolo D, Coloni GF. Thymoma and thymic carcinoma. General Thoracic and Cardiovascular Surg. 2012;60(1):1–12.
Girard N, Ruffini E, Marx A, Faivre-Finn C, Peters S, Committee EG. Thymicepithelial tumours: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26(Suppl 5):v40-55.
Suster S, Rosai J. Multilocular thymic cyst an acquired reactive process Study of 18 cases. Am J Surg Pathol. 1991;15(4):388–98.
Vargas D, Suby-Long T, Restrepo CS. Cystic Lesions of the Mediastinum. Seminars in ultrasound, CT, and MR. 2016;37(3):212–22.
Wang X, Chen K, Li X, et al. Clinical features, diagnosis and thoracoscopic surgical treatment of thymic cysts. J Thoracic Dis. 2017;9(12):5203–11.
Keating J, Judy R, Newton A, Singhal S. Near-infrared operating lamp for intraoperative molecular imaging of a mediastinal tumor. BMC Med Imaging. 2016;16(1):15.
Predina JD, Keating J, Newton A, et al. A clinical trial of intraoperative near-infrared imaging to assess tumor extent and identify residual disease during anterior mediastinal tumor resection. Cancer. 2019;125(5):807–17.
Vaja R, Joshi V, Dawson AG, Waller DA. Is a diagnostic video-assisted thoracoscopic thymectomy an acceptable first-line approach to the suspicious anterior mediastinal mass? J Minimal Access Surg. 2017;13(4):286–90.
Vahrmeijer AL, Hutteman M, Van Der Vorst JR, Van De Velde CJ, Frangioni JV. Image-guided cancer surgery using near-infrared fluorescence. Nature Rev Clin Oncol. 2013;10(9):507–18.
Ishizawa T, Fukushima N, Shibahara J, et al. Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer. 2009;115(11):2491–504.
Manny TB, Krane LS, Hemal AK. Indocyanine green cannot predict malignancy in partial nephrectomy: histopathologic correlation with fluorescence pattern in 100 patients. J Endourol. 2013;27(7):918–21.
Altinoglu EI, Russin TJ, Kaiser JM, et al. Near-infrared emitting fluorophore-doped calcium phosphate nanoparticles for in vivo imaging of human breast cancer. ACS Nano. 2008;2(10):2075–84.
Keating J, Tchou J, Okusanya O, et al. Identification of breast cancer margins using intraoperative near-infrared imaging. J Surg Oncol. 2016;113(5):508–14.
Keating JJ, Nims S, Venegas O, et al. Intraoperative imaging identifies thymoma margins following neoadjuvant chemotherapy. Oncotarget. 2016;7(3):3059.
Kennedy GT, Newton A, Predina J, Singhal S. Intraoperative near-infrared imaging of mesothelioma. Translational Lung Cancer Res. 2017;6(3):279–84.
Jiang JX, Keating JJ, Jesus EM, et al. Optimization of the enhanced permeability and retention effect for near-infrared imaging of solid tumors with indocyanine green. Am J Nuclear Med Molecular Imaging. 2015;5(4):390–400.
Safieddine N, Keshavjee S. Anatomy of the thymus gland. Thoracic Surg Clinics. 2011;21(2):191–5.
Han KN, Kim HK, Choi YH. Transthoracic single-port video-assisted thoracoscopic thymectomy. J Vis Surg. 2018;4:164–164.
Oh Y, Quan YH, Choi Y, et al. Intraoperative combined color and fluorescent images-based sentinel node mapping in the porcine lung: comparison of indocyanine green with or without albumin premixing. J Thorac Cardiovasc Surg. 2013;146(6):1509–15.
Oh Y, Lee YS, Quan YH, et al. Thoracoscopic color and fluorescence imaging system for sentinel lymph node mapping in porcine lung using indocyanine green-neomannosyl human serum albumin: intraoperative image-guided sentinel nodes navigation. Ann Surg Oncol. 2014;21(4):1182–8.
Oh Y, Quan YH, Kim M, Kim BM, Kim HK. Intraoperative fluorescence image-guided pulmonary segmentectomy. J Surg Res. 2015;199(2):287–93.
Kim HK, Quan YH, Oh Y, et al. Macrophage-targeted indocyanine green-neomannosyl human serum albumin for intraoperative sentinel lymph node mapping in porcine esophagus. Ann Thoracic Surg. 2016;102(4):1149–55.
Quan YH, Oh CH, Jung D, et al. Evaluation of intraoperative near-infrared fluorescence visualization of the lung tumor margin with indocyanine green inhalation. JAMA Surg. 2020;155(8):732–40.
Jeong YJ, Lee KS, Kim J, Shim YM, Han J, Kwon O. Does CT of thymic epithelial tumors enable us to differentiate histologic subtypes and predict prognosis? AJR Am J Roentgenol. 2004;183(2):283–9.
Suster S. Diagnosis of thymoma. J Clin Pathol. 2006;59(12):1238–44.
Li H, Zhou J, Chi C, et al. Clinical application of near-infrared thoracoscope with indocyanine green in video-assisted thoracoscopic bullectomy. J Thoracic Dis. 2016;8(7):1841–5.
Jurado J, Javidfar J, Newmark A, et al. Minimally invasive thymectomy and open thymectomy: outcome analysis of 263 patients. Ann Thorac Surg. 2012;94(3):974–81.
Kondo K, Monden Y. Lymphogenous and hematogenous metastasis of thymic epithelial tumors. Ann Thorac Surg. 2003;76(6):1859–64.
Kim HK, Quan YH, Choi BH, et al. Intraoperative pulmonary neoplasm identification using near-infrared fluorescence imaging. Eur J Cardio-Thoracic Surg. 2015;49(5):1497–502.
Choi HS, Kim HK. Multispectral image-guided surgery in patients. Nature Biomed Eng. 2020;4(3):245–6.
Predina JD, Keating J, Newton A, et al. A clinical trial of intraoperative near-infrared imaging to assess tumor extent and identify residual disease during anterior mediastinal tumor resection. Cancer. 2019;125(5):807–17.
Predina JD, Newton A, Kennedy G, Lee MK, Singhal S. Near-Infrared Intraoperative Imaging Can Successfully Identify Malignant Pleural Mesothelioma After Neoadjuvant Chemotherapy. Mol Imaging Jan-Dec. 2017;16:1536012117723785.
Keating JJ, Kennedy GT, Singhal S. Identification of a subcentimeter pulmonary adenocarcinoma using intraoperative near-infrared imaging during video-assisted thoracoscopic surgery. J Thorac Cardiovasc Surg. 2015;149(3):e51-53.
James NS, Chen Y, Joshi P, et al. Evaluation of polymethine dyes as potential probes for near infrared fluorescence imaging of tumors: part - 1. Theranostics. 2013;3(9):692–702.
Mahalingam SM, Kularatne SA, Myers CH, et al. Evaluation of novel tumor-targeted near-infrared probe for fluorescence-guided surgery of cancer. J Med Chem. 2018;61(21):9637–46.
de Valk KS, Deken MM, Handgraaf HJM, et al. First-in-Human Assessment ofcRGD-ZW800-1, a Zwitterionic, Integrin-Targeted, Near-Infrared Fluorescent Peptide in Colon Carcinoma. Clin Cancer Res. 2020;26(15):3990–8.
Funding
This work was supported by a grant from the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (No: HI19C0166).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
YHQ, RX, BHC, JR, JHL, KNH, YHC, B-MK, and HKK declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Funding information was added.
Supplementary Information
Below is the link to the electronic supplementary material.
Intraoperative differentiation of thymic cyst from thymoma based on the NIR fluorescence signal. (MP4 24546 kb)
10434_2022_11466_MOESM2_ESM.jpg
Comparison of TNR in simple cysts, multilocular cysts, and sticky fluid density cysts. Error bars represent the SD value of TNR (**P < 0.001). TNR, tumor-to-normal ratio; CT, computed tomography; SD, standard deviation. (JPG 147 kb)
Rights and permissions
About this article
Cite this article
Quan, Y.H., Xu, R., Choi, B.H. et al. Fluorescence Imaging-Guided Identification of Thymic Masses Using Low-Dose Indocyanine Green. Ann Surg Oncol 29, 4476–4485 (2022). https://doi.org/10.1245/s10434-022-11466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11466-8