Abstract
Background
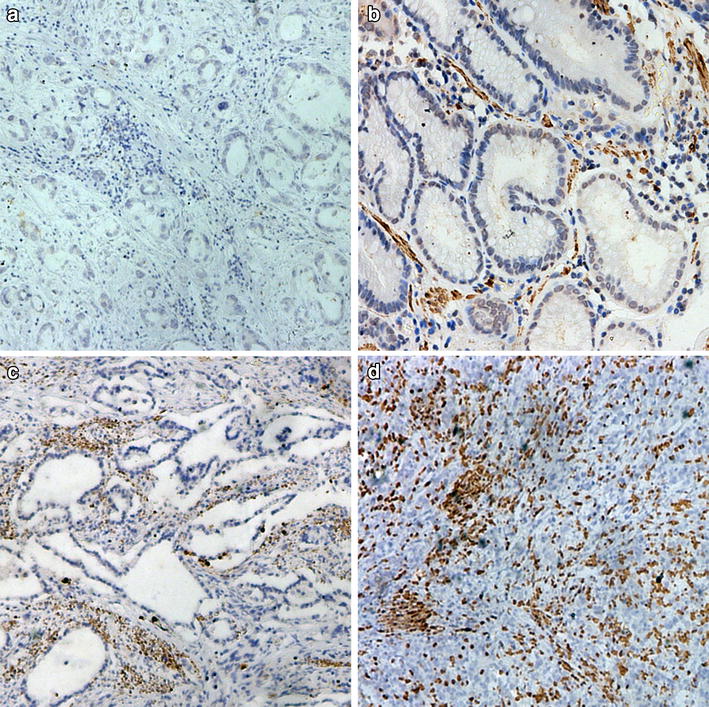
CXCL10, a member of the CXC chemokine family, is known to mediate chemotaxis, apoptosis, angiogenesis, and cell growth. It is also reportedly involved in tumor development and can affect prognosis in several cancers. However, the precise relationship between CXCL10 and the prognosis of patients with esophageal squamous cell carcinoma (ESCC) is not fully understood.
Methods
We used ESCC tissue microarrays containing samples from 177 patients to test whether the CXCL10 expression status, determined using immunohistochemical analysis, is predictive of prognosis. We also tested whether CXCL10 expression status could serve as a clinically useful marker for evaluating the need for adjuvant chemotherapy after surgery.
Results
We found that high CXCL10 expression in clinical samples was an independent prognostic factor and was predictive of a favorable 5-year overall survival and disease-specific survival (p = 0.0102 and 0.0332, respectively). Additionally, no significant difference was detected between patients in the CXCL10-high group treated with surgery alone and those treated with surgery followed by adjuvant chemotherapy. In the CXCL10-low group, on the other hand, patients treated with surgery followed by adjuvant chemotherapy had better 5-year overall survival than those treated with surgery alone.
Conclusions
High CXCL10 expression is an independent prognostic factor and has the potential to serve as a clinically useful marker of the need for adjuvant chemotherapy after surgery in patients with advanced thoracic ESCC.



Similar content being viewed by others
References
Jemal A, Bray F, Center M, et al. Global cancer statics. CA Cancer J Clin. 2011;61:69–90.
Ando N, Ozawa S, Kitagawa Y, et al. Improvement in the results of surgical treatment of advanced squamous esophageal carcinoma during 15 consecutive years. Ann Surg. 2002;232:225–32.
Law S, Kwong D, Wong KF, et al. Improvement in treatment results and long-term survival of patients with esophageal cancer: impact of chemoradiation and change in treatment strategy. Ann Surg. 2003;238:339–47.
Ando N, Iizuka T, Ide H, et al. Surgery plus chemotherapy compared with surgery alone for localized squamous cell carcinoma of the thoracic esophagus: a Japan Clinical Oncology Group Study—JCOG9204. J Clin Oncol. 2003;21:4592–6.
Kato K, Muro K, Minashi K, et al. Phase II study of chemoradiotherapy with 5-fluorouracil and cisplatin for stage II–III esophageal squamous cell carcinoma: JCOG trial (JCOG 9906). Int J Radiat Oncol Biol Phys. 2011;81:684–90.
Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19:68–74.
Sanuki N, Ishikura S, Shinoda M, et al. Radiotherapy quality assurance review for a multi-center randomized trial of locally advanced esophageal cancer: the Japan Clinical Oncology Group (JCOG) trial 0303. Int J Clin Oncol. 2012;17:105–11.
Liu M, Guo S, Stiles JK. The emerging role of CXCL10 in cancer. Oncol Lett. 2011;2:583–9.
Sun L, Hui AM, Su Q, et al. Neuronal and glioma-derived stem cell factor induces angiogenesis within the brain. Cancer Cell. 2006;9:287–300.
Karnoub AE, Dash AB, Vo AP, et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–63.
Ki DH, Jeung HC, Park CH, et al. Whole genome analysis for liver metastasis gene signatures in colorectal cancer. Int J Cancer. 2007;121:2005–12.
Ginos MA, Page GP, Michalowicz BS, et al. Identification of a gene expression signature associated with recurrent disease in squamous cell carcinoma of the head and neck. Cancer Res. 2004;64:55–63.
Talbot SG, Estilo C, Maghami E, et al. Gene expression profiling allows distinction between primary and metastatic squamous cell carcinomas in the lung. Cancer Res. 2005;65:3063–71.
Rosenwald A, Wright G, Chan WC, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:1937–47.
Lu L, Pan K, Zheng HX, et al. IL-17A promotes immune cell recruitment in human esophageal cancers and the infiltrating dendritic cells represent a positive prognostic marker for patient survival. J Immunother. 2013;36:451–8.
Yoo JY, Choi HK, Choi KC, et al. Nuclear hormone receptor corepressor promotes esophageal cancer cell invasion by transcriptional repression of interferon-γ-inducible protein 10 in a casein kinase 2–dependent manner. Mol Biol Cell. 2012;23:2943–54.
Sobin LH, Gospodarowicz MK, Wittekind C, International Union Against Cancer. TNM classification of malignant tumours. 7th ed. Chichester: Wiley; 2010.
Sato Y, Motoyama S, Maruyama K, et al. A second malignancy is the major cause of death among thoracic squamous cell esophageal cancer patients negative for lymph node involvement. J Am Coll Surg. 2005;201:188–93.
Sato Y, Motoyama S, Nanjo H, et al. REG1A expression status suggests chemosensitivity among advanced thoracic esophageal squamous cell carcinoma patients treated with esophagectomy followed by adjuvant chemotherapy. Ann Surg Oncol. 2013;20:3044–51.
Sato Y, Marzese DM, Ohta K, et al. Epigenetic regulation of REG1A and chemosensitivity of cutaneous melanoma. Epigenetics. 2013;8:1043–52.
Wolff AC, Hammond ME, Schwartz JN, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 2007;25:118–45.
Jiang Z, Xu Y, Cai S. CXCL10 expression and prognostic significance in stage II and III colorectal cancer. Mol Biol Rep. 2010;37:3029–36.
Persano L, Crescenzi M, Indraccolo S. Anti-angiogenic gene therapy of cancer: current status and future prospects. Mol Aspects Med. 2007;28:87–114.
Aronica SM, Raiber L, Hanzly M, et al. Antitumor/antiestrogenic effect of the chemokine interferon inducible protein 10 (IP-10) involves suppression of VEGF expression in mammary tissue. J Interferon Cytokine Res. 2009;29:83–92.
Fujita M, Zhu X, Ueda R, et al. Effective immunotherapy against murine gliomas using type 1 polarizing dendritic cells—significant roles of CXCL10. Cancer Res. 2009;69:1587–95.
Jiang XB, Lu XL, Hu P, et al. Improved therapeutic efficacy using vaccination with glioma lysate-pulsed dendritic cells combined with IP-10 in murine glioma. Vaccine. 2009;27:6210–6.
Rainczuk A, Rao JR, Gathercole JL, Fairweather NJ, et al. Evidence for the antagonistic form of CXC-motif chemokine CXCL10 in serous epithelial ovarian tumours. Int J Cancer. 2014;1;134:530–41.
Furuya M, Suyama T, Usui H, et al. Up-regulation of CXC chemokines and their receptors: implications for proinflammatory microenvironments of ovarian carcinomas and endometriosis. Hum Pathol. 2007;38:1676–87.
Datta D, Flaxenburg JA, Laxmanan S, et al. Ras-induced modulation of CXCL10 and its receptor splice variant CXCR3-B in MDA-MB-435 and MCF-7 cells: relevance for the development of human breast cancer. Cancer Res. 2006;66:9509–18.
Kawada K, Hosogi H, Sonoshita M, et al. Chemokine receptor CXCR3 promotes colon cancer metastasis to lymph nodes. Oncogene. 2007;26:4679–88.
Lo BK, Yu M, Zloty D, et al. CXCR3/ligands are significantly involved in the tumorigenesis of basal cell carcinomas. Am J Pathol. 2010;176:2435–46.
Maru SV, Holloway KA, Flynn G, et al. Chemokine production and chemokine receptor expression by human glioma cells: role of CXCL10 in tumour cell proliferation. J Neuroimmunol. 2008;199:35–45.
Acknowledgment
Supported in part by JSPS KAKENHI (Grant 25870072).
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sato, Y., Motoyama, S., Nanjo, H. et al. CXCL10 Expression Status is Prognostic in Patients with Advanced Thoracic Esophageal Squamous Cell Carcinoma. Ann Surg Oncol 23, 936–942 (2016). https://doi.org/10.1245/s10434-015-4909-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4909-1