Abstract
Background
Sentinel lymph node (SLN) biopsy for melanoma often detects minimal nodal tumor burden. Although all node-positive patients are considered stage III, there is controversy regarding the necessity of adjuvant therapy for all patients with tumor-positive SLN.
Methods
Post hoc analysis was performed of a prospective multi-institutional study of patients with melanoma ≥ 1.0 mm Breslow thickness. All patients underwent SLN biopsy; completion lymphadenectomy was performed for patients with SLN metastasis. Kaplan–Meier analysis of disease-free survival (DFS) and overall survival (OS) was performed. Univariate and multivariate Cox regression analyses were performed. Classification and regression tree (CART) analysis also was performed.
Results
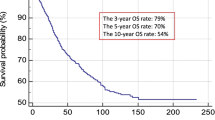
A total of 509 patients with tumor-positive SLN were evaluated. Independent risk factors for worse OS included thickness, age, gender, presence of ulceration, and tumor-positive non-SLN (nodal metastasis found on completion lymphadenectomy). As the number of tumor-positive SLN and the total number of tumor-positive nodes (SLN and non-SLN) increased, DFS and OS worsened on Kaplan–Meier analysis. On CART analysis, the 5-year OS rates ranged from 84.9 % (women with thickness < 2.1 mm, age < 59 years, no ulceration, and tumor-negative non-SLN) to 14.3 % (men with thickness ≥ 2.1 mm, age ≥ 59 years, ulceration present, and tumor-positive non-SLN). Six distinct subgroups were identified with 5-year OS in excess of 70 %.
Conclusions
Stage III melanoma in the era of SLN is associated with a very wide range of prognosis. CART analysis of prognostic factors allows discrimination of low-risk subgroups for which adjuvant therapy may not be warranted.



Similar content being viewed by others
References
Balch CM, Gershenwald JE, Soong SJ, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199–206.
Eggermont AM, Testori A, Marsden J, et al. Utility of adjuvant systemic therapy in melanoma. Ann Oncol. 2009;20 Suppl 6:vi30–4.
Bevilacqua RG, Coit DG, Rogatko A, Younes RN, Brennan MF. Axillary dissection in melanoma. Prognostic variables in node-positive patients. Ann Surg. 1990;212:125–31.
Morton DL, Wanek L, Nizze JA, Elashoff RM, Wong JH. Improved long-term survival after lymphadenectomy of melanoma metastatic to regional nodes. Analysis of prognostic factors in 1134 patients from the John Wayne Cancer Clinic. Ann Surg. 1991;214:491–9.
Drepper H, Biess B, Hofherr B, et al. The prognosis of patients with stage III melanoma. Prospective long-term study of 286 patients of the Fachklinik Hornheide. Cancer. 1993;71:1239–46.
White RR, Stanley WE, Johnson JL, Tyler DS, Seigler HF. Long-term survival in 2,505 patients with melanoma with regional lymph node metastasis. Ann Surg. 2002;235:879–87.
Wiener M, Acland KM, Shaw HM, et al. Sentinel node positive melanoma patients: prediction and prognostic significance of nonsentinel node metastases and development of a survival tree model. Ann Surg Oncol. 2010;17:1995–2005.
Carlson GW, Murray DR, Lyles RH, Staley CA, Hestley A, Cohen C. The amount of metastatic melanoma in a sentinel lymph node: does it have prognostic significance? Ann Surg Oncol. 2003;10:575–81.
Ariyan C, Brady MS, Gonen M, Busam K, Coit D. Positive nonsentinel node status predicts mortality in patients with cutaneous melanoma. Ann Surg Oncol. 2009;16:186–90.
Jakub JW, Huebner M, Shivers S, et al. The number of lymph nodes involved with metastatic disease does not affect outcome in melanoma patients as long as all disease is confined to the sentinel lymph node. Ann Surg Oncol. 2009;16:2245–51.
Ghaferi AA, Wong SL, Johnson TM, et al. Prognostic significance of a positive nonsentinel lymph node in cutaneous melanoma. Ann Surg Oncol. 2009;16:2978–84.
Murali R, Desilva C, Thompson JF, Scolyer RA. Factors predicting recurrence and survival in sentinel lymph node-positive melanoma patients. Ann Surg. 2011;253:1155–64.
McMasters KM, Noyes RD, Reintgen DS, et al. Lessons learned from the Sunbelt Melanoma Trial. J Surg Oncol. 2004;86:212–23.
McMasters KM, Ross MI, Reintgen DS, et al. Final results of the Sunbelt Melanoma Trial. J Clin Oncol. 26: 2008 (May 20 Suppl; abstr 9003).
Balch CM, Gershenwald JE, Soong SJ, et al. Multivariate analysis of prognostic factors among 2,313 patients with stage III melanoma: comparison of nodal micrometastases versus macrometastases. J Clin Oncol. 2010;28:2452–9.
Balch CM, Buzaid AC, Soong SJ, et al. Final version of the American Joint Committee on Cancer staging system for cutaneous melanoma. J Clin Oncol. 2001;19:3635–48.
Roka F, Mastan P, Binder M, et al. Prediction of non-sentinel node status and outcome in sentinel node-positive melanoma patients. Eur J Surg Oncol. 2008;34:82–8.
Lasithiotakis K, Leiter U, Meier F, et al. Age and gender are significant independent predictors of survival in primary cutaneous melanoma. Cancer. 2008;112:1795–804.
Chao C, Martin RC II, Ross MI, et al. Correlation between prognostic factors and increasing age in melanoma. Ann Surg Oncol. 2004;11:259–64.
Scoggins CR, Ross MI, Reintgen DS, et al. Gender-related differences in outcome for melanoma patients. Ann Surg. 2006;243:693–8.
Bowles TL, Xing Y, Hu CY, et al. Conditional survival estimates improve over 5 years for melanoma survivors with node-positive disease. Ann Surg Oncol. 2010;17:2015–23.
Rossi CR, De Salvo GL, Bonandini E, et al. Factors predictive of nonsentinel lymph node involvement and clinical outcome in melanoma patients with metastatic sentinel lymph node. Ann Surg Oncol. 2008;15:1202–10.
Schmidt CR, Panageas KS, Coit DG, Patel A, Brady MS. An increased number of sentinel lymph nodes is associated with advanced Breslow depth and lymphovascular invasion in patients with primary melanoma. Ann Surg Oncol. 2009;16:948–52.
Cadili A, Scolyer RA, Brown PT, Dabbs K, Thompson JF. Total sentinel lymph node tumor size predicts nonsentinel node metastasis and survival in patients with melanoma. Ann Surg Oncol. 2010;17:3015–20.
Ranieri JM, Wagner JD, Azuaje R, et al. Prognostic importance of lymph node tumor burden in melanoma patients staged by sentinel node biopsy. Ann Surg Oncol. 2002;9:975–81.
Conflict of interest
This is a study of the Sunbelt Melanoma Trial, which was sponsored by a grant from Schering Oncology Biotech. All data management and subsequent analysis has been performed independently at the University of Louisville. Schering Oncology Biotech was not involved in conduct of the trial, data analysis, or production of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Egger, M.E., Callender, G.G., McMasters, K.M. et al. Diversity of Stage III Melanoma in the Era of Sentinel Lymph Node Biopsy. Ann Surg Oncol 20, 956–963 (2013). https://doi.org/10.1245/s10434-012-2701-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2701-z