Abstract
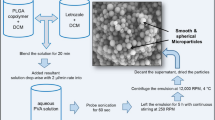
Production and evaluation of novel formulations of tizanidine and tramadol microparticles was the chief purpose of this project. Microparticles of both drugs were prepared separately via temperature change method. To extend the release of formulations, ethyl cellulose was employed. Higuchi, zero-order, first-order, and Korsmeyer–Peppas kinetic models were applied to appraise the mechanism and mode of drugs release. Higuichi model was found to be best for all release profiles. Stability of microparticles at 40°C/75% RH over a 3-month duration was determined by Fourier transform infrared (FTIR), X-ray diffractometry (XRD), and drugs assay. Microparticles were compatible and stable as no significant differences were observed when subjected to drug assay, FTIR, and XRD during accelerated stability studies.







Similar content being viewed by others
References
Meythaler JM, Guin-Renfroe S, Johnson A. Prospective assessment of tizanidine for spasticity due to acquired brain injury. Arch Phys Med Rehab. 2001;82:1155–63.
Saper JR, Lake AE, Cantrell DT. Chronic daily headache prophylaxis with tizanidine: a double-blind, placebo-controlled, multicenter outcome study. Headache. 2002;42:470–82.
Valle M, Garrido MJ, Pavon JM, Calvo R, Troconiz IF. Pharmacokinetic pharmacodynamic modeling of the antinociceptive effects of main active metabolites of tramadol, (þ)-O-desmethyltramadol and (−)-O-desmethyltramadol, in rats. J Pharma Exp Ther. 2000;293:646–53.
Altman RA, Hochberg MC, Moskowitz RW, Schnitzer TJ. Recommendations for the medical management of osteoarthritis of the hip and knee. Arthritis & Rheumatism. 2000;43:1905.
Ernesto R, Renata A, Giuseppe C. Production of cromolyn sodium microparticles for aerosol delivery by supercritical assisted atomization. AAPS PharmSciTech. 2007;8(4):Article 114.
Kyeo RL, Eun JK, Sang WS, Hoo KC. Effect of poloxamer on the dissolution of felodipine and preparation of controlled release matrix tablets containing felodipine. Arch Pharm Res. 2008;31:1023–8.
Ernesto R, Renata A, Giuseppe C. Production of cromolyn sodium microparticles for aerosol delivery by supercritical assisted atomization. AAPS PharmSciTech. 2007;8(4):Article 114.
Pimwipha P, Nalena P, Nuanphun C, Nongnuj M. Preparation and evaluation of chitosan/carrageenan beads for controlled release of sodium diclofenac. AAPS PharmSciTech. 2007;8(4):Article 97.
Sheena YM, Ushakumari L, Harikumar BH, Tresa V, Yohannan CP. FT-IR, FT-Raman and SERS spectra of l-proline. J Iran Chem Soc. 2009;6:138–44.
Li Z, Lu Y, Wu Z, Chen H. Preparation and characterization of coacervate microcapsules for the delivery of antimicrobial oyster peptides. Drug Delev Indus Pharm. 2009;35:369–78.
Khatun M, Islam SMA, Akter P, Quadir MA, Reza MS. Controlled release of naproxen sodium from eudragit RS100 transdermal film. Dhaka University J Pharm Sci. 2004;3:1–10.
Higuchi T. Mechanism of sustained action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52:1145–9.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15:25–35.
Peppas NA. Analysis of Fickian and non-Fickian drug release from polymers. Pharm Acta Helv. 1985;60:110–1.
Acknowledgments
We are grateful to the Higher Education Commission for providing financial support for the analytical analysis conducted in this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aamir, M.N., Ahmad, M. Production and Stability Evaluation of Modified-Release Microparticles for the Delivery of Drug Combinations. AAPS PharmSciTech 11, 351–355 (2010). https://doi.org/10.1208/s12249-010-9392-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-010-9392-1