Abstract
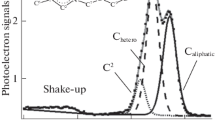
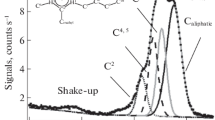
We investigate a typical trihalide ionic liquid, 1-octyl-3-methylimidazolium tribromide, using X‑ray photoelectron spectroscopy. The electronic environment of the anion is analyzed in detail. The Br 3d binding energies of the two components are both higher than that measured for 1-octyl-3-methylimidazolium bromide, due to the delocalization of the negative charge spreading over three atoms within the anion. The basicity of [Br3]– is estimated based upon N 1s binding energy. It is found similar to that of [BF4]–.



Similar content being viewed by others
REFERENCES
A. Ejigu, K. R. J. Lovelock, P. Licence, and D. A. Walsh, Electrochim. Acta 56, 10313 (2011).
M. P. Kaushik and V. Polshettiwar, Indian J. Chem., Sect. B 45, 2542 (2006).
E. F. Smith, I. J. Villar-Garcia, D. Briggs, and P. Licence, Chem. Commun., No. 45, 5633 (2005).
S. Caporali, U. Bardi, and A. Lavacchi, J. Electron Spectrosc. Relat. Phenom. 151, 4 (2006).
T. Cremer, C. Kolbeck, K. R. J. Lovelock, N. Paape, R. Wölfel, P. S. Schulz, P. Wasserscheid, H. Weber, J. Thar, B. Kirchner, F. Maier, and H.-P. Steinrück, Chem.-Eur. J. 16, 9018 (2010).
I. J. Villar-Garcia, E. F. Smith, A. W. Taylor, F. Qiu, K. R. J. Lovelock, R. G. Jones, and P. Licence, Phys. Chem. Chem. Phys. 13, 2797 (2011).
J. S. Zhu, Y. F. Liu, and Q. L. Shen, Angew. Chem., Int. Ed. 55, 9050 (2016).
P. J. Carvalho, S. P. M. Ventura, M. L. S. Batista, B. Schroder, F. Goncalves, J. Esperanca, F. Mutelet, and J. A. P. Coutinho, J. Chem. Phys. 140, 64505 (2014).
D. Gerhard, S. C. Alpaslan, H. J. Gores, M. Uerdingen, and P. Wasserscheid, Chem. Commun., No. 40, 5080 (2005).
S. Men, K. R. J. Lovelock, and P. Licence, Chem. Phys. Lett. 679, 207 (2017).
J. G. Huddleston, A. E. Visser, W. M. Reichert, H. D. Willauer, G. A. Broker, and R. D. Rogers, Green Chem. 3, 156 (2001).
P. A. Z. Suarez, J. E. L. Dullius, S. Einloft, R. F. DeSouza, and J. Dupont, Polyhedron 15, 1217 (1996).
A. W. Taylor, K. R. J. Lovelock, A. Deyko, P. Licence, and R. G. Jones, Phys. Chem. Chem. Phys. 12, 1772 (2010).
C. D. Wagner, L. E. Davis, M. V. Zeller, J. A. Taylor, R. H. Raymond, and L. H. Gale, Surf. Interface Anal. 3, 211 (1981).
D. Briggs and G. Beamson, Anal. Chem. 64, 1729 (1992).
S. Men, J. Jiang, and P. Licence, Chem. Phys. Lett. 674, 86 (2017).
S. Men, K. R. J. Lovelock, and P. Licence, RSC Adv. 5, 35958 (2015).
P. A. Frey and O. T. Magnusson, Chem. Rev. 103, 2129 (2003).
J. F. Moulder, W. F. Stickle, P. E. Sobol and K. D. Bomben, Handbook of X-ray Photoelectron Spectroscopy: A Reference Book of Standard Spectra for Identification and Interpretation of XPS Data (Physical Electronics, Chanhassen, 1995).
S. Spange, R. Lungwitz, and A. Schade, J. Mol. Liq. 192, 137 (2014).
ACKNOWLEDGMENTS
We thank Liaoning Provincial Foundation of Science and Technology (20180550482) for financial support. SM acknowledges Beijing Key Laboratory of Quality Evaluation Technology for Hygiene and Safety of Plastics (Beijing Technology and Business University, Beijing 100048, China) for the award of an Open Fund (QETHSP2019004).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rui Mu, Deng, A. & Men, S. Tribromide Ionic Liquids: Probing the Charge Distribution of the Anion by XPS. Russ. J. Phys. Chem. 94, 1053–1056 (2020). https://doi.org/10.1134/S0036024420050167
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024420050167