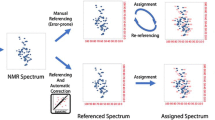
Abstract
RefDB is a secondary database of reference-corrected protein chemical shifts derived from the BioMagResBank (BMRB). The database was assembled by using a recently developed program (SHIFTX) to predict protein 1H, 13C and 15N chemical shifts from X-ray or NMR coordinate data of previously assigned proteins. The predicted shifts were then compared with the corresponding observed shifts and a variety of statistical evaluations performed. In this way, potential mis-assignments, typographical errors and chemical referencing errors could be identified and, in many cases, corrected. This approach allows for an unbiased, instrument-independent solution to the problem of retrospectively re-referencing published protein chemical shifts. Results from this study indicate that nearly 25% of BMRB entries with 13C protein assignments and 27% of BMRB entries with 15N protein assignments required significant chemical shift reference readjustments. Additionally, nearly 40% of protein entries deposited in the BioMagResBank appear to have at least one assignment error. From this study it evident that protein NMR spectroscopists are increasingly adhering to recommended IUPAC 13C and 15N chemical shift referencing conventions, however, approximately 20% of newly deposited protein entries in the BMRB are still being incorrectly referenced. This is cause for some concern. However, the utilization of RefDB and its companion programs may help mitigate this ongoing problem. RefDB is updated weekly and the database, along with its associated software, is freely available at http://redpoll.pharmacy.ualberta.ca and the BMRB website.
Similar content being viewed by others
References
Altschul, S.F., Madden, T.L., Schaffer, A.A., Zhang, J., Zhang, Z., Miller, W. and Lipman, D.J. (1997) Nucl. Acids Res., 25, 3389-3402.
Archer, S.J., Vinson, V.K., Pollard, T.D. and Torchia, D.A. (1994) FEBS Lett., 37, 145-151.
Beger, R.D. and Bolton, P.H. (1997) J. Biomol. NMR, 10, 129-142.
Berman, H.M., Bhat, T.N., Bourne, P.E., Feng, Z., Gillicand, G., Weissig, H. and Westbrook, J. (2000) Nat. Struct. Biol., 7(Suppl.), 957-959.
Bewley, C.A., Gustafson, K.R., Boyd, R.M., Covell, D.G., Bax, A., Clore G.M. and Gronenborn, A.M. (1998) Nat. Struct. Biol., 5, 571-578.
Bjorndhal, T.C., Watson, M.S., Slupsky, C.M., Spyracopoulos, L., Sykes, B.D. and Wishart, D.S. (2001) J. Biomol. NMR, 19, 187-188.
Campbell-Burk, S., Domaille, P.J., Starovas, M.A., Boucher, W. and Laue, E.D. (1992) J. Biomol. NMR, 2, 639-646.
Case, D.A. (2000) Curr. Opin. Struct. Biol., 10, 197-203.
Chandrasekhar, K., Campbell, A.P., Jeng, M.F., Holmgren, A. and Dyson, H.J. (1994) J. Biomol. NMR, 4, 411-432.
Copie, V., Battles, J.A., Schwab, J.M. and Torchia, D.A. (1996) J. Biomol. NMR, 7, 335-340.
Cornilescu, G., Delaglio, F. and Bax, A. (1999) J. Biomol. NMR, 13, 289-302.
Davis, J.H., Agard, D.A., Handel, T.M. and Basus, V.J. (1997) J. Biomol. NMR, 10, 21-27.
Fogh, R.H., Schipper, D., Boelens, R. and Kaptein, R. (1995) J. Biomol. NMR, 5, 259-270.
Gardner, K.H., Rosen, M.K. and Kay, L.E. (1997) Biochemistry, 36, 1389-1401.
Gronenborn, A.M. and Clore, G.M. (1994) J. Biomol. NMR, 4, 455-458.
Haigh, C.W. and Mallion, R.B. (1979) Prog. Nucl. Magn. Reson. Spectrosc., 13, 303.
Hodsdon, M.E., Toner J.J., Cistola D.P. (1995) J. Biomol. NMR, 6, 198-210
Iwadate, M., Asakura, T. and Williamson, M.P. (1999) J. Biomol. NMR" 13, 199-211.
Lam, Y.F. and Kotowycz G. (1977) FEBS Lett. 78, 181-183.
Laskowski, R.A., MacArthur, M.W., Moss, D.S. and Thornton, J.M. (1993) J. Appl. Cryst., 26, 283-291.
Le, H. and Oldfield, E. (1994) J. Biomol. NMR, 4, 341-348.
Manber, U. and Bigot, P. (1997) In USENIX Symposium on Internet Technologies and Systems (NSITS’97), Monterey, California, pp. 231-239.
Manber, U. and Wu, S. (1994) In Usenix Winter 1994 Technical Conference(best paper award), San Francisco (January 1994), pp. 23-32.
Markley, J.L., Bax, A., Arata, Y., Hilbers, C.W., Kaptein, R., Sykes, B.D., Wright, P.E. and Wüthrich, K. (1998) J. Biomol. NMR, 12, 1-23.
Maurer, T. and Kalbitzer, H.R. (1996) J. Magn. Reson., B113, 177-178.
Metzler, W.J., Constantine, K.L., Friedrichs, M.S., Bell, A.J., Ernst, E.G., Lavoie, T.B. and Mueller, L. (1993) Biochemistry, 32, 13818-13829.
Moseley, H.N. and Montelione, G.T. (1999) Curr. Opin. Struct. Biol., 9, 635-642.
Osapay, K. and Case, D.A. (1991) J. Am. Chem. Soc., 113, 9436-9444.
Osapay, K. and Case, D.A. (1994) J. Biomol. NMR, 4, 215-320.
Ottinger, M., Zerbe, O., Guntert, P. and Wuthrich, K. (1997) J. Mol. Biol., 272, 64-81.
Pelton, J.G. Torchia, D.A., Meadow, N.D., Wong, C. and Roseman, S. (1991) Biochemistry, 30, 10043-10057.
Potts, B.C. and Chazin, W.J. (1998) J. Biomol. NMR, 11, 45-57.
Qin, J., Clore, G.C. and Gronenborn, A.M. (1996) Biochemistry, 35, 7-13.
Schwarzinger, S., Kroon, G.J., Foss, T.R., Chung, J., Wright, P.E. and Dyson, H.J. (2001) J. Am. Chem. Soc., 123, 2970-2978.
Seavey, B.R., Farr, E.A., Westler, W.M. and Markley, J.L. (1991) J. Biomol. NMR, 1, 217-236.
Spera, S. and Bax, A. (1991) J. Am. Chem. Soc., 113, 5490-5492.
Szilagyi, L. (1995) Prog. Nucl. Magn. Reson. Spectrosc., 27, 325.
Vaguine, A.A., Richelle, J. and Wodak, S.J. (1999) Acta Cryst., D55, 191-205.
Vriend, G. (1990) J. Mol. Graph., 8, 52-56.
Wagner, G., Pardi, A. and Wüthrich, K. (1983) J. Am, Chem. Soc., 105, 5948.
Wang, A.C., Grzesiek, S., Tschudin, R., Lodi, P.J. and Bax, A. (1995) J. Biomol. NMR, 5, 376-382.
Weisemann, R., Rüterjans, H. and Bermel, W. (1993) J. Biomol. NMR, 3, 113-120
Williamson, M. P. and Asakura, T. (1997) Meth. Mol. Biol., 60, 53-69.
Williamson, M.P., Kikuchi, J. and Asakura, T. (1995) J. Mol. Biol., 247, 541-546.
Wishart, D.S. and Case, D.A. (2001) Meth. Enzymol., 338, 3-34.
Wishart, D.S. and Nip, A.M. (1998) Biochem. Cell Biol., 76, 153-163.
Wishart, D.S. and Sykes, B.D. (1994a) Meth. Enzymol., 239, 363-392.
Wishart, D.S. and Sykes, B.D. (1994b) J. Biomol. NMR, 4, 171-180.
Wishart, D.S., Bigam, C.G., Yao, J., Abildgaard, F., Dyson, H.J., Oldfield, E., Markley, J.L. and Sykes, B.D. (1995) J. Biomol. NMR, 6, 135-140.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991) J. Mol. Biol., 222, 311-333.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1992) Biochemistry, 31, 1647-1651.
Wishart, D.S., Watson, M.S., Boyko, R.F. and Sykes, B.D. (1997) J. Biomol. NMR, 10, 329-336.
Wishart D.S., Willard L., Richards F.M. and Sykes B.D. (1994) VADAR: A comprehensive program for protein structure evaluation. Version 1.2. Edmonton, Alberta, Canada.
Xu, X.P. and Case, D.A. (2001) J. Biomol. NMR, 21, 321-333.
Yamazaki, T., Hinck, A.P., Wang, Y.-X., Nicholson, L.K., Trchia, D.A., Wingfield, P.T., Stahl, S.J., Kaufman J.D., Chan, C.-H., Domaile, P.J. and Lam, P.Y.S. (1996) Protein Sci., 5, 495-506.
Yamazaki, T., Yoshida, M., Kanaya, S., Nakamura, H. and Nagayama, K. (1991) Biochemistry, 30,6036-6047.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Neal, S. & Wishart, D.S. RefDB: A database of uniformly referenced protein chemical shifts. J Biomol NMR 25, 173–195 (2003). https://doi.org/10.1023/A:1022836027055
Issue Date:
DOI: https://doi.org/10.1023/A:1022836027055