Abstract
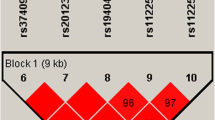
Breast cancer is a common disease in both developing and developed countries with early identification and treatment improving prognosis and survival. Heparan sulfate proteoglycans (HSPGs) are key components of the extracellular matrix (ECM) that mediate cell adhesion, motility, proliferation, invasion and cell signalling. Members of the syndecan family of HSPGs have been identified to be involved in breast cancer progression through their varied interactions with a number of growth factors, ligands and receptors. Specifically, high expression levels of syndecan-1 (SDC1) have been demonstrated in more invasive breast tumours while elevated syndecan-4 (SDC4) levels have been identified to correspond with improved prognosis. With genetic changes in the syndecans and their association with breast cancers plausible, we examined two single nucleotide polymorphisms in SDC1 (rs1131351) and SDC4 (rs67068737) within an Australian Caucasian breast cancer case/control population. No association was found with SDC4 and breast cancer in our population. However, a significant association between SDC1 and breast cancer was identified in both our case/control population and in a replication cohort. When both populations were combined for analysis, this association became more significant (genotype, p = 0.0003; allele, p = 0.0001). This data suggests an increased risk of developing breast cancer associated with the presence of the C allele of the SDC1 rs1131351 single nucleotide polymorphism (SNP) and may provide a marker toward early breast cancer detection.


Similar content being viewed by others
References
ABS. Leading causes of death by gender. In: 3303.0 Causes of Death, Australia, 2011. Australian Bureau of Statistics, Australia. 2011. http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/F25E446E1BE6C931CA257B2E000D729C?opendocument. Accessed 04/03/2014 2013.
Davies EL. Breast cancer. Medicine. 2012;40(1):5–9.
Kamangar F, Abnet CC, Hutchinson AA, Newschaffer CJ, Helzlsouer K, Shugart YY, et al. Polymorphisms in inflammation-related genes and risk of gastric cancer (Finland). Cancer Causes Control CCC. 2006;17(1):117–25. doi:10.1007/s10552-005-0439-7.
Haupt LM, Griffiths LR. Heparan sulfate proteoglycans, tumour progression and the cancer stem cell niche. Curr Cancer Ther Rev. 2009;5(4):256–60. doi:10.2174/157339409789712672.
Bernfield M, Gotte M, Park PW, Reizes O, Fitzgerald ML, Lincecum J, et al. Functions of cell surface heparan sulfate proteoglycans. Annu Rev Biochem. 1999;68:729–77. doi:10.1146/annurev.biochem.68.1.729.
Gallagher JT, Turnbull JE, Lyon M. Patterns of sulphation in heparan sulphate: polymorphism based on a common structural theme. Int J Biochem. 1992;24(4):553–60.
Park PW, Reizes O, Bernfield M. Cell surface heparan sulfate proteoglycans: selective regulators of ligand-receptor encounters. J Biol Chem. 2000;275(39):29923–6. doi:10.1074/jbc.R000008200.
Tumova S, Woods A, Couchman JR. Heparan sulfate proteoglycans on the cell surface: versatile coordinators of cellular functions. Int J Biochem Cell Biol. 2000;32(3):269–88.
Gallagher JT. Heparan sulfate: growth control with a restricted sequence menu. J Clin Invest. 2001;108(3):357–61. doi:10.1172/JCI13713.
Blackhall FH, Merry CL, Davies EJ, Jayson GC. Heparan sulfate proteoglycans and cancer. Br J Cancer. 2001;85(8):1094–8. doi:10.1054/bjoc.2001.2054.
Sugahara K, Kitagawa H. Heparin and heparan sulfate biosynthesis. IUBMB Life. 2002;54(4):163–75. doi:10.1080/15216540214928.
Grobe K, Ledin J, Ringvall M, Holmborn K, Forsberg E, Esko JD, et al. Heparan sulfate and development: differential roles of the N-acetylglucosamine N-deacetylase/N-sulfotransferase isozymes. Biochim Biophys Acta. 2002;1573(3):209–15.
Lander AD, Selleck SB. The elusive functions of proteoglycans: in vivo veritas. J Cell Biol. 2000;148(2):227–32.
Sasisekharan R, Shriver Z, Venkataraman G, Narayanasami U. Roles of heparan-sulphate glycosaminoglycans in cancer. Nat Rev Cancer. 2002;2(7):521–8. doi:10.1038/nrc842.
Sanderson B, Hohlweck F, McMilan B. 3 pillars of a successful denials management program. Healthc Financ Manag J Healthc Financ Manag Assoc. 2008;62(9):74–80.
Tkachenko E, Rhodes JM, Simons M. Syndecans: new kids on the signaling block. Circ Res. 2005;96(5):488–500. doi:10.1161/01.RES.0000159708.71142.c8.
Beauvais DM, Rapraeger AC. Syndecan-1-mediated cell spreading requires signaling by alphavbeta3 integrins in human breast carcinoma cells. Exp Cell Res. 2003;286(2):219–32.
Beauvais DM, Rapraeger AC. Syndecans in tumor cell adhesion and signaling. Reprod Biol Endocrinol RB&E. 2004;2:3. doi:10.1186/1477-7827-2-3.
Burbach BJ, Ji Y, Rapraeger AC. Syndecan-1 ectodomain regulates matrix-dependent signaling in human breast carcinoma cells. Exp Cell Res. 2004;300(1):234–47. doi:10.1016/j.yexcr.2004.07.001.
Maeda T, Alexander CM, Friedl A. Induction of syndecan-1 expression in stromal fibroblasts promotes proliferation of human breast cancer cells. Cancer Res. 2004;64(2):612–21.
Maeda N, Fukazawa N, Hata T. The binding of chondroitin sulfate to pleiotrophin/heparin-binding growth-associated molecule is regulated by chain length and oversulfated structures. J Biol Chem. 2006;281(8):4894–902. doi:10.1074/jbc.M507750200.
Zhang Y, McKown RL, Raab RW, Rapraeger AC, Laurie GW. Focus on molecules: syndecan-1. Exp Eye Res. 2011;93(4):329–30. doi:10.1016/j.exer.2010.06.008.
Okolicsanyi RK, van Wijnen AJ, Cool SM, Stein GS, Griffiths LR, Haupt LM. Heparan sulfate proteoglycans and human breast cancer epithelial cell tumorigenicity. J Cell Biochem. 2014;115(5):967–76. doi:10.1002/jcb.24746.
Elenius K, Jalkanen M. Function of the syndecans—a family of cell surface proteoglycans. J Cell Sci. 1994;107(Pt 11):2975–82.
Lendorf ME, Manon-Jensen T, Kronqvist P, Multhaupt HA, Couchman JR. Syndecan-1 and syndecan-4 are independent indicators in breast carcinoma. J Histochem Cytochem Off J Histochem Soc. 2011;59(6):615–29. doi:10.1369/0022155411405057.
Ha E, Kim MJ, Choi BK, Rho JJ, Oh DJ, Rho TH, et al. Positive association of obesity with single nucleotide polymorphisms of syndecan 3 in the Korean population. J Clin Endocrinol Metab. 2006;91(12):5095–9. doi:10.1210/jc.2005-2086.
Schuring AN, Lutz F, Tuttelmann F, Gromoll J, Kiesel L, Gotte M. Role of syndecan-3 polymorphisms in obesity and female hyperandrogenism. J Mol Med. 2009;87(12):1241–50. doi:10.1007/s00109-009-0529-1.
Menashe I, Maeder D, Garcia-Closas M, Figueroa JD, Bhattacharjee S, Rotunno M, et al. Pathway analysis of breast cancer genome-wide association study highlights three pathways and one canonical signaling cascade. Cancer Res. 2010;70(11):4453–9. doi:10.1158/0008-5472.CAN-09-4502.
Okolicsanyi RK, Faure M, Jacinto JM, Chacon-Cortes D, Chambers S, Youl PH, et al. Association of the SNP rs2623047 in the HSPG modification enzyme SULF1 with an Australian Caucasian Breast Cancer Cohort. Gene. 2014;547(1):50–4. doi:10.1016/j.gene.2014.06.009.
Chacon-Cortes D, Haupt LM, Lea RA, Griffiths LR. Comparison of genomic DNA extraction techniques from whole blood samples: a time, cost and quality evaluation study. Mol Biol Rep. 2012;39(5):5961–6. doi:10.1007/s11033-011-1408-8.
Nasiri H, Forouzandeh M, Rasaee MJ, Rahbarizadeh F. Modified salting-out method: high-yield, high-quality genomic DNA extraction from whole blood using laundry detergent. J Clin Lab Anal. 2005;19(6):229–32. doi:10.1002/jcla.20083.
Kato M, Saunders S, Nguyen H, Bernfield M. Loss of cell surface syndecan-1 causes epithelia to transform into anchorage-independent mesenchyme-like cells. Mol Biol Cell. 1995;6(5):559–76.
Leppa S, Harkonen P, Jalkanen M. Steroid-induced epithelial-fibroblastic conversion associated with syndecan suppression in S115 mouse mammary tumor cells. Cell Regul. 1991;2(1):1–11.
Leppa S, Mali M, Miettinen HM, Jalkanen M. Syndecan expression regulates cell morphology and growth of mouse mammary epithelial tumor cells. Proc Natl Acad Sci U S A. 1992;89(3):932–6.
Sanderson RD, Borset M. Syndecan-1 in B lymphoid malignancies. Ann Hematol. 2002;81(3):125–35. doi:10.1007/s00277-002-0437-8.
Barbareschi M, Maisonneuve P, Aldovini D, Cangi MG, Pecciarini L, Angelo Mauri F, et al. High syndecan-1 expression in breast carcinoma is related to an aggressive phenotype and to poorer prognosis. Cancer. 2003;98(3):474–83. doi:10.1002/cncr.11515.
Lundstrom E, Sahlin L, Skoog L, Hagerstrom T, Svane G, Azavedo E, et al. Expression of syndecan-1 in histologically normal breast tissue from postmenopausal women with breast cancer according to mammographic density. Climacteric J Int Menopause Soc. 2006;9(4):277–82. doi:10.1080/13697130600865741.
Alexander CM, Reichsman F, Hinkes MT, Lincecum J, Becker KA, Cumberledge S, et al. Syndecan-1 is required for Wnt-1-induced mammary tumorigenesis in mice. Nat Genet. 2000;25(3):329–32. doi:10.1038/77108.
Maeda T, Desouky J, Friedl A. Syndecan-1 expression by stromal fibroblasts promotes breast carcinoma growth in vivo and stimulates tumor angiogenesis. Oncogenesis. 2006;25(9):1408–12. doi:10.1038/sj.onc.1209168.
Baba F, Swartz K, van Buren R, Eickhoff J, Zhang Y, Wolberg W, et al. Syndecan-1 and syndecan-4 are overexpressed in an estrogen receptor-negative, highly proliferative breast carcinoma subtype. Breast Cancer Res Treat. 2006;98(1):91–8. doi:10.1007/s10549-005-9135-2.
Leivonen M, Lundin J, Nordling S, von Boguslawski K, Haglund C. Prognostic value of syndecan-1 expression in breast cancer. Oncology. 2004;67(1):11–8. doi:10.1159/000080280.
Burbach BJ, Friedl A, Mundhenke C, Rapraeger AC. Syndecan-1 accumulates in lysosomes of poorly differentiated breast carcinoma cells. Matrix Biol J Int Soc Matrix Biol. 2003;22(2):163–77.
Woods A, Couchman JR. Syndecan-4 and focal adhesion function. Curr Opin Cell Biol. 2001;13(5):578–83.
Langford JK, Yang Y, Kieber-Emmons T, Sanderson RD. Identification of an invasion regulatory domain within the core protein of syndecan-1. J Biol Chem. 2005;280(5):3467–73. doi:10.1074/jbc.M412451200.
Oh ES, Woods A, Couchman JR. Multimerization of the cytoplasmic domain of syndecan-4 is required for its ability to activate protein kinase C. J Biol Chem. 1997;272(18):11805–11.
Baciu PC, Goetinck PF. Protein kinase C regulates the recruitment of syndecan-4 into focal contacts. Mol Biol Cell. 1995;6(11):1503–13.
Longley RL, Woods A, Fleetwood A, Cowling GJ, Gallagher JT, Couchman JR. Control of morphology, cytoskeleton and migration by syndecan-4. J Cell Sci. 1999;112(Pt 20):3421–31.
Woods A, Couchman JR. Syndecan 4 heparan sulfate proteoglycan is a selectively enriched and widespread focal adhesion component. Mol Biol Cell. 1994;5(2):183–92.
Mundhenke C, Meyer K, Drew S, Friedl A. Heparan sulfate proteoglycans as regulators of fibroblast growth factor-2 receptor binding in breast carcinomas. Am J Pathol. 2002;160(1):185–94. doi:10.1016/S0002-9440(10)64362-3.
Financial support
RKO is supported by an Australian Postgraduate Award stipend, and the authors wish to acknowledge the support of the Estate of the late Clem Jones AO, Cancer Council Queensland, Cancer Australia Grant (100639) and the Griffith Health Institute, Griffith University. SKC is an Australian Research Council Future Fellow. PHY is an NHMRC Early Career Fellow.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Rachel K. Okolicsanyi and Anne Buffiere are co-first authors.
Rights and permissions
About this article
Cite this article
Okolicsanyi, R.K., Buffiere, A., Jacinto, J.M.E. et al. Association of heparan sulfate proteoglycans SDC1 and SDC4 polymorphisms with breast cancer in an Australian Caucasian population. Tumor Biol. 36, 1731–1738 (2015). https://doi.org/10.1007/s13277-014-2774-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2774-3