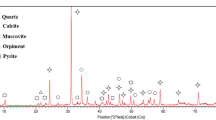
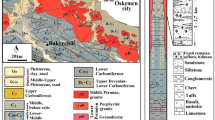
Abstract
In this study, we assess the link between morphologies and composition of gold grains and supergene processes of Au migration and redeposition by aqueous solutions in the aggressive environment of cyanidation tailings. High concentrations of both dissolved (10 μg/L) and solid (0.97 ppm) native gold in the upper horizons of the Komsomolsk tailings profile formed as the result of the long-term oxidation of the cyanidation waste. These horizons stand out due to contrasting changes in pH, Eh and the concentration of the elements in solutions (from 0.8 × 10−4 to 72 g/L iron, from 1.5 × 10−5 to 30 g/L arsenic and from 0 to 37 g/L aluminum). One of the main findings of this study is that the Au and Ag contained in tailings as impurities in the residual sulfide minerals are almost completely redeposited in the top few centimeters of the cross-section. Gold dissolves into pore solution in thiosulfate complexes in the reducing environment of the lower part of tailings and mainly in hydroxide complexes in the acidic environment of the upper horizons. Native gold is found only in the upper few centimeters and presents as irregular grains and crystals of an Au–Ag alloy and as microcrystalline and flat growths of pure Au. The surface morphologies of gold grains are the result of its supergene dissolution, redeposition and crystallization in tailings.








Similar content being viewed by others
References
Alakangas L, Ohlander B (2006) Formation and composition of cemented layers in low-sulphide mine tailings, Laver, northern Sweden. Environ Geol 50:809–819
Andreu E, Torró L, Proenza JA, Domenech C, García-Casco A, Villanova de Benavent C, Chavez C, Espaillat J, Lewis JF (2015) Weathering profile of the Cerro de Maimón VMS deposit (Dominican Republic): textures, mineralogy, gossan evolution and mobility of gold and silver. Ore Geol Rev 65:165–179
Aylmore MG, Muir DM (2001) Thiosulfate leaching of gold. Miner Eng 14(2):1–46
Azizi A, Olsen C, Larachi F (2014) Efficient strategies to enhance gold leaching during cyanidation of multisulfidic ores. Can J Chem Eng 92:1687–1692
Berrodier I, Farges F, Benedetti M, Winterer M, BE Brown Jr, Deveughèle M (2004) Adsorption mechanisms of trivalent gold on iron- and aluminum-(oxy)hydroxides. Part 1: X-ray absorption and Raman scattering spectroscopic studies of Au(III) adsorbed on ferrihydrite, goethite, and boehmite. Geochim Cosmochim Acta 68(14):3019–3042
Blowes DW, Jambor JL (1990) The pore-water geochemistry and the mineralogy of the vadose zone of sulfide tailings, Waite Amulet, Quebec, Canada. Appl Geochem 5:327–346
Blowes DW, Reardon EJ, Jambor JL, Cherry JA (1991) The formation and potential importance of cemented layers in inactive sulfide mine tailings. Geochim Cosmochim Acta 55:965–978
De Oliveira SMB, Campos EG (1991) Gold-bearing iron duricrust in Central Brazil. J Geochem Explor 41(3):309–323
Dutova EM, Bukaty MB, Nevolko AI, Pokrovcky DS, Shvartsev SL (2006) Hydrogenous concentration of gold in alluvial placers of Egorevsky district (Salair). Russ Geol Geophys 47(3):364–376
Freyssinet P, Butt CRM, Morris RC, Piantone P (2005) Ore-forming processes related to lateritic weathering. Econ Geol 100:681–722
Gaskova OL, Bortnikova SB, Airiyants AA, Kolmogorov YuP, Pashkov MV (2000) Geochemical Features of an anthropogenic impoundment with cyanidation wastes of gold–arsenopyrite–quartz ores. Geokhimiya 38(3):281–291
Getsina ML, Toropchenova ES, Nabiullina SN, Koshcheeva IY, Kubrakova IV (2012) Modeling of colloidal gold migration in surface natural waters. B ESD RAS 4:NZ9001. doi:10.2205/2012NZ_ASEMPG
Graupner T, Kassahun A, Rammlmair D, Meima JA, Kock D, Furche M, Fiege A, Schippers A, Melcher F (2007) Formation of sequences of cemented layers and hardpans within sulfide-bearing mine tailings (mine district Freiberg, Germany). Appl Geochem 22:2486–2508
Greffié C, Benedetti MF, Parron C, Amouric M (1996) Gold and iron oxide associations under supergene conditions: an experimental approach. Geochim Cosmochim Acta 60(9):1531–1542
Johnson CA (2015) The fate of cyanide in leach wastes at gold mines: an environmental perspective. Appl Geochem 57:194–205
Kubrakova IV, Fortygin AV, Lobov SG, Koshcheeva IJ, Tyutyunnik OA, Mironenko MV (2011) Migration of platinum, palladium and gold in the water systems of platinum deposits. Geochem Int 49(11):1138–1152
Leybourne MI, Goodfellow WD, Boyle DR, Hall GEM (2000) Form and distribution of gold mobilized into surface waters and sediments from a gossan tailings pile, Murray Brook massive sulphide deposit, New Brunswick, Canada. Appl Geochem 15(5):629–646
Liu W, Etschmann B, Testemale D, Hazemann J-L, Rempel K, Müller H, Brugger J (2014) Gold transport in hydrothermal fluids: competition among the Cl−, Br−, HS− and NH3(aq) ligands. Chem Geol 376:11–19
Myagkaya IN, Lazareva EV, Gustaytis MA, Zhmodik SM (2016) Gold and silver in a system of sulfide tailings. Part 1: migration in water flow. J Geochem Explor 160(1):16–30
Nesterenko GV (1991) The forecast of gold mineralization by placers (on the example of areas of southern Siberia). Nauka Siberian Branch, Novosibirsk, p 190
Plyusnin AM, Pogrebnyak YuF, Tatyankina EM (1980) Proportion between gold species in the water of gold deposits. Dok Akad Nauk SSSR 251(4):971–973
Ran Y, Fu J, Rate AW, Gilkes RJ (2002) Adsorption of Au(I, III) complexes on Fe, Mn oxides and humic acid. Chem Geol 185(1–2):33–49
Seeley JB, Senden TJ (1994) Alluvial gold in Kalimantan, Indonesia: a colloidal origin? J Geochem Explor 50(1–3):457–478
Smith ACS, Dehrmann A, Pullen R (1984) The effect of cyanide-bearing gold tailings disposal on water quality in Witwatersrand, South Africa. Cyanide and the Environment. In: DJ van Zyl (ed) Colorado State University 1:221–229
Stoffregen R (1986) Observations on the behavior of gold during supergene oxidation at Summitville, Colorado, U.S.A., and implications for electrum stability in the weathering environment. Appl Geochem 1(5):549–558
Ta C, Reith F, Brugger J, Pring A, Lenehan CE (2014) Analysis of gold(I/III)-complexes by HPLC-ICP-MS demonstrates gold(III) stability in surface waters. Environ Sci Technol 48(10):5737–5744
Usher A, McPhail DC, Brugger J (2009) A spectrophotometric study of aqueous Au(III) halide–hydroxide complexes at 25–80 °C. Geochim Cosmochim Acta 73(11):3359–3380
Vilor NV, Kazmin LA, Goryachev NA (2014) Sulphide–arsenide gold complexes in hydrothermal solutions at formation of gold deposits (thermodynamic modeling). Dokl Earth Sci 458(1):61–66
Webster JG (1986) The solubility of gold and silver in the system AuAgSO2H2O at 25 °C and 1 atm. Geochim Cosmochim Acta 50(9):1837–1845
Webster JG, Mann AW (1984) The influence of climate, geomorphology and primary geology on the supergene migration of gold and silver. J Geochem Explor 22(1–3):21–42
Acknowledgments
The reported study was funded by the Russian Foundation for Basic Research (research projects Nos. 14-05-31005 mol_a; 14-05-00293). Authors thank employees IGM SB RAS and IPGG SB RAS: A. Ya. Shevko, V. V. Olenchenko, E. P. Shevko, N. A. Abrosimova, D. O. Kucher for participation in field work in 2014 and analysts N. V. Androsova, N. F. Nechepurenko, Yu. P. Kolmogorov, O. S. Hmelnikova, and N. G. Karmanova for analytical works.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Kirillov M. V. has received research grant from the Russian Foundation for Basic Research (Research Project No. 14-05-31005 mol_a), and Bortnikova S. B. has received research grants from the Russian Foundation for Basic Research (Research Project No. 14-05-00293).
Rights and permissions
About this article
Cite this article
Kirillov, M.V., Bortnikova, S.B. & Gaskova, O.L. Authigenic gold formation in the cyanidation tailings of gold–arsenopyrite–quartz ore of Komsomolsk deposit (Kuznetski Alatau, Russia). Environ Earth Sci 75, 1050 (2016). https://doi.org/10.1007/s12665-016-5852-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-016-5852-6