Abstract
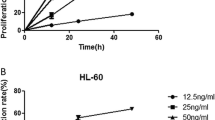
Ursolic acid (UA), a pentacyclic triterpenoid derived from a variety of medicinal plants, exhibits potent anticancer activity against many types of cancer cells. However, the anticancer mechanism of UA is not clearly understood. Suppression of phosphatase and a tensin homolog deleted on chromosome 10 (PTEN) gene expression leading to activation of the phosphatidylinositol-3-OH kinase (PI3K)/Akt pathway has been observed in many cancers including leukemia, making the PTEN gene and PI3K/Akt pathway a central target for cancer therapy. Here, we demonstrated that UA was able to inhibit growth, induce apoptosis in a human chronic myelogenous leukemia cell line (K562 cells) via upregulation of PTEN gene expression, inhibit Akt kinase activity, change mitochondrial transmembrane potential and reduce the release of cytochrome c and the activity of caspases. These results suggest that UA may elicit its strong antitumor effects via upregulation of the PTEN gene and inhibition of the PI3K/Akt pathway.
Similar content being viewed by others
References
Cheng, J. Q., Lindsley, C. W., Cheng, G. Z., Yang, H., and Nicosia, S. V., The Akt/PKB pathway: molecular target for cancer drug discovery. Oncogene, 24, 7482–7492 (2005).
Cohen, G. M., Caspases: the executioners of apoptosis. Biochem. J., 326, 1–16 (1997).
Gayathri, R., Priya, D. K., Gunassekaran, G. R., and Sakthisekaran, D., Ursolic acid attenuates oxidative stressmediated hepatocellular carcinoma induction by diethyl nitrosamine in male Wistar rats. Asian Pac. J. Cancer Prev., 10, 933–938 (2009).
Grandage, V. L., Gale, R. E., Linch, D. C., and Khwaja, A., PI3-kinase/Aktis consttutively activein primary acute myeloid leukaemia cells and regulates survival and chemoresistance via NFkappaB Mapkinase and p53 pathways. Leukemia, 19, 586–594 (2005).
Lin, J. Q., Zhang, Z. H., and Zeng, S. Q., TRAIL-induced apoptosis proceeding from caspase-3-dependent and -independent pathways in distinct HeLa cells. Biochem. Biophys. Res. Commun., 346, 1136–1141 (2006).
Liu, M. L. and Hong, S. T., Early phase of amyloid beta42-induced cytotoxicity in neuronal cells is associated with vacuole formation and enhancement of exocytosis. Exp. Mol. Med., 37, 559–566 (2005).
Liu, X. S. and Jiang, J., Induction of apoptosis and regulation of the MAPK pathway by ursolic acid in human leukemia K562 cells. Planta Med., 73, 1192–1194 (2007).
Maehama, T. and Dixon, J. E., The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J. Biol. Chem., 273, 13375–13378 (1998).
Parsons, D. W., Wang, T. L., Samuels, Y., Bardelli, A., Cummins, J. M., DeLong, L., Silliman, N., Ptak, J., Szabo, S., Willson, J. K., Markowitz, S., Kinzler, K. W., Vogelstein, B., Lengauer, C., and Velculescu, V. E., Colorectal cancer: mutations in a signalling pathway. Nature, 436, 792 (2005).
Qiang, Y. W., Kopantzev, E., and Rudikoff, S., Insulin like growth factor-I signaling in multiple myeloma: downstream elements, functional correlates, and pathway cross-talk. Blood, 99, 4138–4146 (2002).
Rehm, M., Düßmann, H., and Jänicke, R. U., Single-cell fluorescence resonance energy transfer analysis demonstrates that caspase activation during apoptosis is a rapid process. J. Biol. Chem., 277, 24506–24514 (2002).
Song, G., Ouyang, G., and Bao, S., The activation of Akt/PKB signaling pathway and cell survival. J. Cell. Mol. Med., 9, 59–71 (2005).
Tamura, M., Gu, J., Danen, E. H., Takino, T., Miyamoto, S., and Yamada, K. M., PTEN interactions with focal adhesion kinase and suppression of the extracellular matrixdependent phosphate-dylinositol 3-kinase/Akt cell survival pathway. J. Biol. Chem., 274, 20693–20703 (1999).
Tazzari, P. L., Cappellini, A., Grafone, T., Mantovani, I., Ricci, F., Billi, A. M., Ottaviani, E., Conte, R., Martinelli, G., and Martelli, A. M., Detection ofserine 473 phosphorylated Akt in acute myeloid leukaemia blasts by ow cytometry. Br. J. Haematol., 126, 675–681 (2004).
Vivanco, I. and Sawyers, C. L., The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat. Rev. Cancer, 2, 489–501 (2002).
Wang, X., Li, L., Wang, B., and Xiang, J., Effects of ursolic acid on the proliferation and apoptosis of human ovarian cancer cells. J. Huazhong Univ. Sci. Technolog. Med. Sci., 29, 761–764 (2009).
Wang, Z., Smith, K. S., Murphy, M., Piloto, O., Somervaille, T. C., and Cleary, M. L., Glycogensynthase kinase 3 in MLL leukaemia maintenance and targeted therapy. Nature, 455, 1205–1209 (2008).
Xu, Q., Simpson, S. E., Scialla, T. J., Bagg, A., and Carroll, M., Survival of acute myeloid leukemia cells requires PI3 kinase activation. Blood, 102, 972–980 (2003).
Zhang, Y. X., Kong, C. Z., Wang, L. H., Li, J. Y., Liu, X. K., Xu, B., Xu, C. L., and Sun, Y. H., Ursolic acid overcomes Bcl-2-mediated resistance to apoptosis in prostate cancer cells involving activation of JNK-induced Bcl-2 phosphorylation and degradation. J. Cell. Biochem., 109, 764–773 (2010).
Zhao, S., Konopleva, M., Cabreira-Hansen, M., Xie, Z., Hu, W., Milella, M., Estrov, Z., Mills, G. B., and Andreeff, M., Inhibition of phosphatidylinositol3-kinase dephosphorylates BAD and promotes apoptosis in myeloid leukemias. Leukemia, 18, 267–275 (2004).
Zou, H., Li, Y., Liu, X., and Wang, X., An APAF-1.cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J. Biol. Chem., 274, 11549–11556 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, B., Wang, X., Chi, Zf. et al. Ursolic acid-induced apoptosis in K562 cells involving upregulation of PTEN gene expression and inactivation of the PI3K/Akt pathway. Arch. Pharm. Res. 35, 543–548 (2012). https://doi.org/10.1007/s12272-012-0318-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0318-1