Abstract
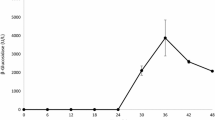
Tamarind kernel powder (TKP), a soluble agro-residue, was used to examine the production of both cellulolytic and xylanolytic enzymes in a submerged culture of Termitomyces clypeatus, an edible mushroom. Soluble TKP containing xyloglucan as the major polysaccharide induced all cellulolytic and xylanolytic enzymes, and enzyme production increased up to 3% (w/v) TKP with culture filtrate consisting of xylanase and CMCase at a ratio of 4: 1 app. Strong catabolic repression of enzyme production was also observed with the soluble substrate, although fed-batch addition of soluble substrate at late growth phase modified the enzyme kinetics by improving the yield by 30%. The results indicate that inducers were possibly released from TKP by cellulose and xylan fractions of the lignocellulosic polymer. Therefore, the present study reports the successful economic utilization of TKP, an abundantly available soluble agro-residue, for the production of both cellulolytic and xylanolytic enzymes in a single fermentation method.
Similar content being viewed by others
References
Damaso, C. T. M., M. M. C. A. Carolinam, and N. J. Pereira (2000) Use of corncob for endo-xylanase production by thermophilic fungus Thermomyces lanuginosus IOC-4145. Appl. Biochem. Biotechnol. 84: 821–834.
Shin, C. S., J. P. Lee, J. S. Lee, and S. C. Park (2000) Enzyme production of Trichoderma ressei Rut C-30 on various lignocellulosic substrates. Appl. Biochem. Biotechnol. 84: 237–245.
Purkarthofer, H., M. Sinner, and W. Steiner (1993) Cellulase free xylanase from Thermomyces lanuginosus: Optimization of production in submerged and solid state culture. Enz. Microbiol. Technol. 15: 677–682.
Sivori, A. S., O. A. Mercuri, and F. Farchiassin (1996) Kinetics of xylanase and cellulase production by Ascobolous gamundii (Fungi- Ascomycotina). Rev. Argent. Microbiol. 28: 9–15.
Gasper, A., T. Cosson, C. Roques, and P. Thronant (1997) Study on the production of a xylanolytic complex from Penicillum canescens 10-10c. Appl. Biochem. Biotechnol. 67: 45–58.
Srivastava, H. C. and P. P. Singh (1967) Structure of the polysaccharide from tamarind kernal. Carbohydr. Res. 4: 326–342.
Gidley, M. J., P. S. Lillford, D. W. Rowlands, P. Lang, M. Dentini, V. Crescenzi, S. M. Edward, C. Fanutti, and J. S. G. Reid (1991) Structure and solution properties of tamarind seed polysaccharides. Carbhydr. Res. 214: 299–314.
Marathe, R. M., U. S. Annapure, R. S. Singhal, and P. R. Kulkarni (2002) Gelling behaviour of polyose from tamarind kernel polysaccharides. Food Hydrocolloid 16: 423–426.
Ghosh, A. K., M. L. Jana, A. K. Naskar, R. Chatterjee, and S. Sengupta (2002) Removal of carboxymethyl cellulase activity from the culture filtrate of Termitomyces clypeatus producing xylanase. Biotechnol. Lett. 24: 1249–1252.
Sengupta, S., M. L. Jana, D. Sengupta, and A. K. Naskar (2000) A note on the estimation of microbial glycosidase activities by dinitrosalicylic acid reagent. Appl. Microbiol. Biotechnol. 53: 732–735.
Saha, R., S. Burman Roy, and S. Sengupta (2003) Stabilization and improvement of catalytic activity of a low molar mass cellobiase by cellobiase-sucrase aggregation in the culture filtrate of Termitomyces clypeatus. Biotechnol. Prog. 18: 1240–1248.
Sinha, N. and S. Sengupta (1995) Simultaneous production of α-arabinofuranosidase and xylanase by Termitomyces clypeatus. World J. Microbiol. Biotechnol. 11: 359–360.
Mandels, M., R. Andreotti, and C. Roche (1975) Measurement of saccharifying cellulase. Biotechnol. Bioeng. 6: 21–33.
Ashwell, G. (1957) Estimation of total carbohydrate in solution in suspension or in whole cell or tissue by anthrone reagent. pp. 73. In: S. P. Colock, and N.O. Kaplan (eds) Methods in Enzymology, vol 3, Academic Press, NY.
Hrmova, M., P. Biely, and M. Vrsanska (1986) Specificity of cellulase and β-xylanase induction in Trichoderma ressei QM9414. Arch. Microbiol. 144: 307–311.
Senior, D. J., P. R. Mayers, and J. N. Saddler (1989) Production and purification of xylanases. pp. 641–654. In: N. G. Lewis and M.G. Paice (eds.). Biogenesis and Biodegradation of Plant Cell Wall Polymers. American Chemical Society, Washington DC, USA.
Hrmova, M., P. Biely, and M. Vrsanska (1989) Cellulose- and xylan-degrading enzymes of Aspergillus terreus and Aspergillus niger. Enz. Microbiol. Technol. 11: 610–616.
Mishra, C., S. Keskar, and M. Rao (1984) Production and properties of extracellular endoxylanase from Neurospora crassa. Appl. Env. Microbiol. 48: 224–228.
Haltrich, D., B. Sebesta, and W. Steiner (1995) Induction of xylanase and cellulase in Schizophyllum commune. pp. 305–318. In: J. N. Saddler and M. H. Penner (eds.). Enzymatic Degradation of Insoluble Carbohydrates. ACS Symposium, Series 618, American Chemical Society, Washington DC, USA.
Brown, J. A., S. A. Collin, and T. M. Wood (1997) Development of a medium for high cellulase, xylanase and β-glucosidase production by a mutant strain (NTG III/6) of the cellulolytic fungus Penicillum pinophilum. Enz. Microbiol. Technol. 9: 355–360.
Meng, M., J. Mukherjee, and T. Scheper (2001) Application of oxygen vectors to Claviceps purpurea cultivation. Appl. Microbiol. Biotechnol. 55: 411–416.
Gomes, D. J., J. Gomes, and W. Steiner (1994) Production of highly thermostable xylanase by a wild strain of thermophilic fungus Thermoascus aurantiacus and partial characterization of the enzyme. J. Biotechnol. 37: 11–22.
Teunissen, M. J., G. V. M. de Kort, H. J. M. Opden Camp, and J. H. J. Huis in t’ Veld (1992) Production of cellulolytic and xylanolytic enzymes during growth of the anaerobic fungus Piromyces sp. on different substrates. J. Gen. Microbiol. 138: 1657–1664.
Warzywoda, M., V. Ferre, and J. Pourquie (1983) Development of a culture medium for large-scale production of cellulolytic enzymes by Trichoderma reesei. Biotechnol. Bioeng. 25: 3005–3010.
Saddler, J. N., C. M. Hogan, and G. Louis-Seize (1985) A comparison between the celluase systems of Trichoderma hazianum E58 and Trichoderma ressei C30. Appl. Microbiol. Biotechnol. 22: 139–145.
Pardo, A.G., M. Obertello, and F. Forchiassin (2000) Cellulose and xylan degrading enzymes in Thecotheus pellelier. Rev. Argent. Microbiol. 32: 190–195.
Haltrich, D. and W. Steiner (1994) Formation of xylanase by Schizophyllum commun: Effect of medium components. Enz. Microbiol. Technol. 16: 229–235.
Pinaga, F., M. T. Fernandez-Espinar, S. Valles, and D. Ramon (1994) Xylanase production in Aspergillus nidulans: Induction and carbon catabolite repression. FEMS Microbiol. Lett. 115: 319–324.
Bailey, M. J. and L. Viikari (1993) Production of xylanases by Aspergillus fumigatus and Aspergillus oryzae on xylan based media. World J. Microbiol. Biotechnol. 9: 80–84.
Ghosh, M. and G. Nanda (1994) Physiological studies on xylose induction and glucose repression of xylanolytic enzymes in Aspergillus sydowii MG 49. FEMS Microbiol. Lett. 117: 151–156.
Sabu, A., A. Pandey, M. Jaafor Daud, and G. Szakacs (2005) Tamarind seed powder and palm kernel cake: Two novel agro residues for the production of tannase under solid state fermentation of Aspergillus niger ATCC 16620. Bioresour. Technol. 96: 1223–1228.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chatterjee, R., Majumder, K. & Sengupta, S. Tamarind kernel powder co-induces xylanase and cellulase production during submerged fermentation of Termitomyces clypeatus . Biotechnol Bioproc E 15, 854–861 (2010). https://doi.org/10.1007/s12257-009-3042-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-009-3042-z