Abstract
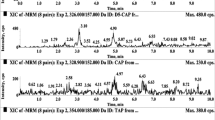
A liquid chromatography tandem-mass spectrometry (LC-MS/MS) method has been developed for the simultaneous determination of 22 veterinary drug residues, belonging to eight classes (coccidiostats, lincosamides, macrolides, tetracyclines, sulfonamides, benzimidazoles, diterpenes, and diaminopyrimidines), in eggs. Chromatographic separations were achieved on an XBridge BEH C18 column (150 × 2.1 mm, 3.5 μm, Waters, USA) maintained at 35 °C. The mobile phase was eluted at 400 μL min−1 in gradient mode between water and methanol/acetonitrile (20:80 v/v), both containing 0.1 % formic acid. The samples were prepared by protein precipitation with acetonitrile without additional cleanup steps. The method was successfully validated according to the Commission Decision 2002/657/EC and was demonstrated to be highly selective and free of matrix and residual effects. The method presented low limits of detection (0.37 to 7.5 μg kg−1) and quantification (1.25 to 20 μg kg−1). For banned substances, the decision limit values (CCα) and detection capability (CCβ) were 0.62–7.5 and 0.65–8.1 μg kg−1, respectively. For substances with a maximum residue limit, the CCα and CCβ values were 2.15–1061.5 and 2.3–1135.9 μg kg−1, respectively. All calibration curves showed excellent correlation (r ≥ 0.99). The recovery of the analytes and internal standards (49.0–103.7 %) was reached with high precision (RSD <8 %). At different concentration levels, the variations in precision and accuracy, in terms of repeatability and in-laboratory reproducibility, were <11 %. The new method is applicable to the routine analysis of commercial egg samples.



Similar content being viewed by others
References
Aerts MM, Hogenboom AC, Brinkman UA (1995) Analytical strategies for the screening of veterinary drugs and their residues in edible products. J Chromatogr B 667:1–40
Brasil, Agência Nacional de Vigilância Sanitária. Resolução n. 899, de 29 de maio de 2003. Determina a publicação do Guia para Validação de Métodos Analíticos e Bioanalíticos. Diário Oficial da República Federativa do Brasil, Brasília, DF, 02 fev. 2003.
Brasil, Agência Nacional de Vigilância Sanitária. Resolução RDC n. 27, de 17 de maio de 2012. Diário Oficial da União. Dispõe sobre os requisitos mínimos para a validação de métodos bioanalíticos empregados em estudos com fins de registro e pós-registro de medicamentos. Diário Oficial da República Federativa do Brasil, Brasília, DF, 22 maio 2012.
Capriotti AL, Cavaliere C, Piovesana S, Samperi R, Laganà A (2012) Multiclass screening method based on solvent extraction and liquid chromatography-tandem mass spectrometry for the determination of antimicrobials and mycotoxins in egg. J Chromatogr A 1268:84–90
Christodoulou EA, Samanidou VF, Papadoyannis LN (2007) Validation of an HPLC-UV method according to the European Union Decision 2002/657/EC for the simultaneous determination of 10 quinolones in chicken muscle and egg yolk. J Chromatogr B 859:246–255
Codex Alimentarius Commission (2012) Maximum residue limits for veterinary drugs in foods, updated as at the 35th Session of the Codex Alimentarius Commission, CAC/MRL 2-2012, July 2012, ftp://ftp.fao.org/codex/weblinks/MRL2_e_2012.pdf. Accessed 09 August 2015
Commission Decision 2002/657/EC implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results, 2002. Off J Eur Commun, No. L 221/8–36
Commission Regulation (EU) 124/2009 setting maximum levels for the presence of coccidiostats or histomonostats in food resulting from the unavoidable carry-over of these substances in non-target feed, 2009. Off J Eur Commun, No. L40/7–11
Commission Regulation (EU) 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin, 2010. Off J Eur Commun, No. L15/1–72
Croubels S, De Baere S, De Backer P (2003) Practical approach for the stability testing of veterinary drugs in solutions and in biological matrices during storage. Anal Chim Acta 483:419–427
Dasenaki ME, Thomaidis NS (2015) Multi-residue determination of 115 veterinary drugs and pharmaceutical residues in milk powder, butter, fish tissue and eggs using liquid chromatography-tandem mass spectrometry. Anal Chim Acta 880:103–121
de Paula FC, de Pietro AC, Cass QB (2008) Simultaneous quantification of sulfamethoxazole and trimethoprim in whole egg samples by column-switching high-performance liquid chromatography using restricted access media column for on-line sample clean-up. J Chromatogr A 1189:221–226
De Ruyck H, De Ridder H (2007) Determination of tetracycline antibiotics in cow’s milk by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectr 21:1511–1520
Deng XJ, Yang HQ, Li JZ, Song Y, Guo DH, Luo Y, Du XN, Bo T (2011) Multiclass residues screening of 105 veterinary drugs in meat, milk, and egg using ultra high performance liquid chromatography tandem quadrupole time-of-flight mass spectrometry. J Liq Chromatogr R T 34:2286–2303
Dubreil-Cheneau E, Bessiral M, Roudaut B, Verdon E, Sanders P (2009) Validation of a multi-residue liquid chromatography-tandem mass spectrometry confirmatory method for 10 anticoccidials in eggs according to Commission Decision 2002/657/EC. J Chromatogr A 1216:8149–8157
Food and Drug Administration (2015) Code of Federal Regulations, Title 21, Part 556, Section 500, U.S. Government Printing Office, Washington DC, (Chapter 1), http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/cfrsearch.cfm?cfrpart=556. Accessed 01 September 2015
Frenich AG, Aguilera-Luiz MD, Vidal JLM, Romero-Gonzalez R (2010) Comparison of several extraction techniques for multiclass analysis of veterinary drugs in eggs using ultra-high pressure liquid chromatography-tandem mass spectrometry. Anal Chim Acta 661:150–160
Furusawa N (2003) Rapid high-performance liquid chromatographic determining technique of sulfamonomethoxine, sulfadimethoxine, and sulfaquinoxaline in eggs without use of organic solvents. Anal Chim Acta 481:255–259
Gehring TA, Rushing LG, Churchwell MI, Doerge DR, McErlane KM, Thompson HC (1996) HPLC determination of sulfadiazine residues in coho salmon (Oncorhynchus kisutch) with confirmation by liquid chromatography with atmospheric pressure chemical ionization mass spectrometry. J Agric Food Chem 44:3164–3169
Heller DN, Nochetto CB (2004) Development of multiclass methods for drug residues in eggs: silica SPE cleanup and LC-MS/MS analysis of ionophore and macrolide residues. J Agric Food Chem 52:6848–6856
Heller DN, Nochetto CB, Rummel NG, Thomas MH (2006) Development of multiclass methods for drug residues in eggs: hydrophilic solid-phase extraction cleanup and liquid chromatography/tandem mass spectrometry analysis of tetracycline, fluoroquinolone, sulfonamide, and beta-lactam residues. J Agric Food Chem 54:5267–5278
Jia X, Shao B, Wu Y, Yang Y, Zhang J (2008) Simultaneous determination of tetracylines and quinolones antibiotics in egg by ultra-performance liquid chromatography-electrospray tandem mass spectrometry. J AOAC Int 91:461–468
Jimenez V, Rubies A, Centrich F, Companyo R, Guiteras J (2011) Development and validation of a multiclass method for the analysis of antibiotic residues in eggs by liquid chromatography-tandem mass spectrometry. J Chromatogr A 1218:1443–1451
Kan CA, Petz M (2000) Residues of veterinary drugs in eggs and their distribution between yolk and white. J Agric Food Chem 48:6397–6403
Kennedy DG, McCracken RJ, Cannavan A, Hewitt SA (1998) Use of liquid chromatography mass spectrometry in the analysis of residues of antibiotics in meat and milk. J Chromatogr A 812:77–98
Kim E, Bahn K, Kang E, Kim M (2012) Quantitative analysis of lincomycin and narasin in poultry, milk and eggs by liquid chromatography-tandem mass spectrometry. Food Chem 132:1063–1070
Mol HGJ, Plaza-Bolanos P, Zomer P, de Rijk TC, Stolker AAM, Mulder PPJ (2008) Toward a generic extraction method for simultaneous determination of pesticides, mycotoxins, plant toxins, and veterinary drugs in feed and food matrixes. Anal Chem 80:9450–9459
Nakajima T, Nagano C, Sasamoto T, Hayashi H, Kanda M, Kanai S, Takeba K, Matsushima Y, Takano I (2012) Development and validation of rapid analysis method for multi-class veterinary drugs in livestock products by LC-MS/MS. Shokuhin Eiseigaku Zasshi 53:243–253
Nakazawa H, Ino S, Kato K, Watanabe T, Ito Y, Oka H (1999) Simultaneous determination of residual tetracyclines in foods by high-performance liquid chromatography with atmospheric pressure chemical ionization tandem mass spectrometry. J Chromatogr B 732:55–64
Oka H, Ikai Y, Kawamura N, Yamada M, Harada K, Ito S, Suzuki M (1989) Photodecomposition products of tetracycline in aqueous-solution. J Agric Food Chem 37:226–231
Pereira AS, Santos TTD, Coelho AFS (2014) Quality of eggs sold in different commercial establishments and the study of the conditions of storage. Food Sci Technol Int 34:82–87
Peters RJB, Bolck YJC, Rutgers P, Stolker AAM, Nielen MWF (2009) Multi-residue screening of veterinary drugs in egg, fish and meat using high-resolution liquid chromatography accurate mass time-of-flight mass spectrometry. J Chromatogr A 1216:8206–8216
Robert C, Gillard N, Brasseur PY, Pierret G, Ralet N, Dubois M, Delahaut P (2013) Rapid multi-residue and multi-class qualitative screening for veterinary drugs in foods of animal origin by UHPLC-MS/MS. Food Addit Contam A: Chem Anal Control Expo Risk Assess 30:443–457
Schneider MJ, Donoghue DJ (2003) Multiresidue determination of fluoroquinolone antibiotics in eggs using liquid chromatography-fluorescence-mass spectrometry. Anal Chim Acta 483:39–49
Soeborg T, Ingerslev F, Halling-Sorensen B (2004) Chemical stability of chlortetracycline and chlortetracycline degradation products and epimers in soil interstitial water. Chemosphere 57:1515–1524
Spisso BF, Ferreira RG, Pereira MU, Monteiro MA, Cruz TA, da Costa RP, Lima AMB, da Nobrega AW (2010) Simultaneous determination of polyether ionophores, macrolides and lincosamides in hen eggs by liquid chromatography-electrospray ionization tandem mass spectrometry using a simple solvent extraction. Anal Chim Acta 682:82–92
Stolker AA, Brinkman UA (2005) Analytical strategies for residue analysis of veterinary drugs and growth-promoting agents in food-producing animals—a review. J Chromatogr A 1067:15–53
Summa S, Lo Magro S, Armentano A, Muscarella M (2015) Development and validation of an HPLC/DAD method for the determination of 13 sulphonamides in eggs. Food Chem 187:477–484
Tolika EP, Samanidou VF, Papadoyannis IN (2011) Development and validation of an HPLC method for the simultaneous determination of ten sulfonamide residues in whole egg according to 2002/657/EC. J Liq Chromatogr R T 34:2396–2410
World Health Organization (2011) Tackling antibiotic resistance from a food safety perspective in Europe http://www.euro.who.int/__data/assets/pdf_file/0005/136454/e94889.pdf. Accessed 09 August 2015
Xie KZ, Jia LF, Yao YL, Xu D, Chen SQ, Xie X, Pei Y, Bao WB, Dai GJ, Wang JY, Liu ZP (2011) Simultaneous determination of thiamphenicol, florfenicol and florfenicol amine in eggs by reversed-phase high-performance liquid chromatography with fluorescence detection. J Chromatogr B 879:2351–2354
Acknowledgments
The authors would like to thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the scholarship, and Fundação Araucária for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Flávia Lada Degaut Pontes declares that she has no conflict of interest. João Cleverson Gasparetto declares that he has no conflict of interest. Thais Martins Guimarães de Francisco declares that she has no conflict of interest. Heloise Calzado Goetzke declares that she has no conflict of interest. Letícia Paula Leonart declares that she has no conflict of interest. Daniel Altino de Jesus declares that he has no conflict of interest. Roberto Pontarolo declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Pontes, F.L.D., Gasparetto, J.C., de Francisco, T.M.G. et al. Development and Validation of a Multiclass Method for the Analysis of Veterinary Drug Residues in Eggs Using Liquid Chromatography-Tandem Mass Spectrometry. Food Anal. Methods 10, 1063–1077 (2017). https://doi.org/10.1007/s12161-016-0670-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-016-0670-8