Abstract
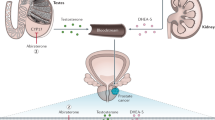
Secondary hormonal therapy is a treatment option in patients with castration-resistant prostate cancer (CRPC); however, it is underutilized and is room for optimization and improvement. In this context, androgen receptor (AR) is the Achilles’ heel, being critically important and various mechanisms ranging from receptor mutations to secondary signaling pathways are responsible for some of the biological heterogeneity, demanding a multimodal approach. A comprehensive review of the peer-reviewed literature is performed on the topic of molecular mechanisms supporting secondary hormonal therapies, including expanded alternative hormonal therapies for CRPC. Essential concepts in clinical treatment of patients with progression on primary hormonal therapy are maintaining the castrate state, accounting for the intermittency phenomenon and sequentially using oral antiandrogens and adrenolytics heading to androgen depletion microenvironment. Survival prolongation, pain relief or measurable improvement in tumor-related symptoms should be persecuted and are considered to be a tangible benefit of obvious worth to the patient. Understanding the underlying molecular substratum is of paramount importance to hormonal therapy optimization in this context once current androgen-depletion strategies are incomplete, and residual androgens as well as alternative routes contribute to sustained AR activity and disease progression to a lethal phenotype. One or many mechanisms may be playing a role, even within the same patient and lastly are potential targets for treatment. Five fundamental mechanisms mediated through the AR to promote tumor growth (three of which depend on ligand signaling) added to the stem cell pathway must be recognized in CRPC. They are persistence of intratumoral androgens as a result of in situ steroidogenesis or adrenal source; AR mutations that allow promiscuous activation by otherwise nonsignaling ligands; wild-type AR gene amplification; alterations in AR coactivator-to-corepressor ratio that impact transcription; outlaw AR pathways that bypass the need for androgens by signaling through crosstalk with other ligand-bound receptors, cytokines, or transactivation of activated tyrosine kinase receptors in the cytosol.


Similar content being viewed by others
Abbreviations
- PCa:
-
Prostate cancer
- SAP:
-
Serum acid phosphatase
- PSA:
-
Prostate-specific antigen
- CRPC:
-
Castration-resistant prostate cancer
- AAWD:
-
Antiandrogen withdrawal phenomenon
- AR:
-
Androgen receptor
- ER:
-
Estrogen receptor
- NTD:
-
Amino-terminal transactivation domain
- DBD:
-
DNA-binding domain
- LBD:
-
Ligand-binding domain
- DHT:
-
Dihydrotestosterone
- AREs:
-
Androgen response elements
- DHEA:
-
Dihydroepiandrostenedione
- DHEAS:
-
DHEA-sulfate
- DES:
-
Diethylstilbestrol
- RANKL:
-
Receptor activator of nuclear factor kappa B ligand
- TNF:
-
Tumor necrosis factor
- COX-2:
-
Cyclooxygenase-2
- CoAD:
-
Collateral androgen depletion
- ET-1:
-
Endothelin-1
- NE:
-
Neuroendocrine
- CGA:
-
Chromogranin A
References
Huggins C, Hodges CV. Studies of prostate cancer I. The effect of castration, of oestrogen and of androgen injection on serum phosphatases in metastatic carcinoma f the prostate. Cancer Res. 1941;1:293–301.
Scher HI, Steineck G, Kelly WK. Hormone-refractory (D3) prostate cancer: refining the concept. Urology. 1995;46:142–8.
Petrylak DP, Tangen CM, Hussain MHA, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20.
Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12.
Fleming MT, Morris MJ, Heller G, et al. Post-therapy changes in PSA as an outcome measure in prostate cancer clinical trials. Nat Clin Pract Oncol. 2006;3:658–67.
Scher HI, Halabi S, Tannock I, Morris M, Sternberg CN, Carducci MA, et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol. 2008;26:1148–59.
Reis LO, Sasse AD, Matheus WE, Denardi F, Stopiglia RM, da Silva MM, Ferreira U. Prostate cancer: evidence based clinical practice. Actas Urol Esp. 2009;33:344–50.
Claessens F, Denayer S, Van Tilborgh N, Kerkhofs S, Helsen C, Haelens A. Diverse roles of androgen receptor (AR) domains in AR-mediated signaling. Nucl Recept Signal. 2008;6:e008.
Denmeade SR, Lin XS, Isaacs JT. Role of programmed (apoptotic) cell death during the progression and therapy for prostate cancer. Prostate. 1996;28:251–65.
Chmelar R, Buchanan G, Need EF, et al. Androgen receptor coregulators and their involvement in the development and progression of prostate cancer. Int J Cancer. 2007;120:719–33.
Hsieh AC, Small EJ, Ryan CJ. Androgen-response elements in hormone refractory prostate cancer: implications for treatment development. Lancet Oncol. 2007;8:933–9.
Hellerstedt B. Hormonal therapy options for patients with a rising prostate specific antigen level after primary treatment for prostate cancer. Urology. 2003;62:79–86.
Culig Z, Stober J, Gast A, et al. Activation of two mutant androgen receptors from human prostatic carcinoma by adrenal androgens and metabolic derivatives of testosterone. Cancer Detect Prev. 1996;20:68–75.
Tan J, Sharief Y, Hamil KG, et al. Dehydroepiandrosterone activates mutant androgen receptors expressed in the androgen-dependent human prostate cancer xenograft CWR22 and LNCaP cells. Mol Endocrinol. 1997;11:450–9.
Serrate C, Loriot Y, De La Motte Rouge T, et al. Diethylstilbestrol (DES) retains activity, is a reasonable option in patients previously treated with docetaxel for castration-resistant prostate cancer. Ann Oncol. 2009;20:965.
Cleffi S, Neto AS, Reis LO, Maia P, Fonseca F, Wroclawski ML, Neves M, Pompeo AC, Del Giglio A, Faria EF, Tobias-Machado M. Androgen deprivation therapy and morbid obesity: do they share cardiovascular risk through metabolic syndrome? Actas Urol Esp. 2011;35(5):259–65.
Mellado B, Codony J, Ribal MJ, Visa L, Gascon P. Molecular biology of androgen-independent prostate cancer: the role of the androgen receptor pathway. Clin Trans Oncol. 2009;11:5–10.
Freeman MR, Cinar B, Lu ML. Membrane rafts as potential sites of nongenomic hormonal signaling in prostate cancer. Trends Endocrinol Metab. 2005;16:273–9.
Scher HI, Sawyers CL. Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol. 2005;23:8253–61.
Eder IE, Culig Z, Putz T, Nessler-Menardi C, Bartsch G, Klocker H. Molecular biology of the androgen receptor: from molecular understanding to the clinic. Eur Urol. 2001;40:241–51.
Feldman BJ, Feldman D. The development of androgen-independent prostate cancer. Nat Rev Cancer. 2001;1:34–45.
Buchanan G, Yang M, Cheong A, Harris JM, Irvine RA, Lambert PF, et al. Structural and functional consequences of glutamine tract variation in the androgen receptor. Hum Mol Genet. 2004;13:1677–92.
Veldscholte J, Risstalpers C, Kuiper G, Jenster G, Berrevoets C, Claassen E, et al. A mutation in the ligand-binding domain of the androgen receptor of human Lncap cells affects steroid binding characteristics and response to anti-androgens. Biochem Biophys Res Commu. 1990;173:534–40.
Taplin ME, Bubley GJ, Shuster TD, et al. Mutations of the androgen-receptor gene in metastatic androgen independent prostate cancer. N Engl J Med. 1995;332:1393–8.
Dehm SM, Schmidt LJ, Heemers HV, et al. Splicing of a novel androgen receptor exon generates a constitutively active androgen receptor that mediates prostate cancer therapy resistance. Cancer Res. 2008;68:5469–77.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vessella R, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33–9.
Dehm SM, Tindall DJ. Androgen receptor structural and functional elements: role and regulation in prostate cancer. Mol Endocrinol. 2007;21:2855–63.
Holzbeierlein J, Lal P, LaTulippe E, et al. Gene expression analysis of human prostate carcinoma during hormonal therapy identifies androgen-responsive genes and mechanisms of therapy resistance. Am J Pathol. 2004;164:217–27.
Cohen BL, Gomez P, Omori Y, Duncan RC, Civantos F, Soloway MS, Lokeshwar VB, Lokeshwar BL. Cyclooxygenase-2 (cox-2) expression is an independent predictor of prostate cancer recurrence. Int J Cancer. 2006;119:1082–7.
Quinn DI, Henshall SM, Sutherland RL. Molecular markers of prostate cancer outcome. Eur J Cancer. 2005;41:858–87.
Need EF, Scher HI, Peters AA, Moore NL, Cheong A, Ryan CJ, et al. A Novel androgen receptor amino terminal region reveals two classes of amino/carboxyl interaction-deficient variants with divergent capacity to activate responsive sites in chromatin. Endocrinology. 2009;150:2674–82.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vessella R, Rosenfeld MG, Sawyers CL. Molecular determinants of resistance to anti-androgen therapy. Nature Med. 2004;10:33–9.
Baron S, Manin M, Beaudoin C, Leotoing L, Communal Y, Veyssiere G, et al. Androgen receptor mediates non-genomic activation of phosphatidylinositol 3-oh kinase in androgen-sensitive epithelial cells. J Biol Chem. 2004;279:14579–86.
Migliaccio A, Castoria G, Di Domenico M, De Falco A, Bilancio A, Lombardi M, et al. Steroid-induced androgen receptor-oestradiol receptor beta-src complex triggers prostate cancer cell proliferation. EMBO J. 2000;19:5406–17.
Peterziel H, Mink S, Schonert A, Becker M, Klocker H, Cato ACB. Rapid signalling by androgen receptor in prostate cancer cells. Oncogene. 1999;18:6322–9.
Malinowska K, Neuwirt H, Cavarretta IT, Bektic J, Steiner H, Dietrich H, et al. Interleukin-6 stimulation of growth of prostate cancer in vitro and in vivo through activation of the androgen receptor. Endocr Relat Cancer. 2009;16:155–69.
Robinson DR, Zylstra CR, Williams BO. Wnt signaling and prostate cancer. Curr Drug Targets. 2008;9:571–80.
Seaton A, Scullin P, Maxwell PJ, Wilson C, Pettigrew J, Gallagher R, et al. Interleukin-8 signaling promotes androgen-independent proliferation of prostate cancer cells via induction of androgen receptor expression and activation. Carcinogenesis. 2008;29:1148–56.
Dagvadorj A, Collins S, Jomain JB, Abdulghani J, Karras J, Zellweger T, Li H, Nurmi M, Alanen K, Mirtti T, Visakorpi T, Bubendorf L, Goffin V, Nevalainen MT. Autocrine prolactin promotes prostate cancer cell growth via Janus kinase-2-signal transducer and activator of transcription-5a/b signaling pathway. Endocrinology. 2007;148:3089–101.
Montgomery RB, Mostaghel EA, Vessella R, Hess DL, Kalhorn TF, Higano CS, True LD, Nelson PS. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 2008;68:4447–54.
Geller J, Albert J, Nachtsheim D, Loza D, Lippman S. Steroid levels in cancer of the prostate–markers of tumor differentiation and adequacy of antiandrogen therapy. Prog Clin Biol Res. 1979;33:103–11.
Geller J, de la Vega DJ, Albert JD, Nachtsheim DA. Tissue dihydrotestosterone levels and clinical response to hormonal therapy in patients with advanced prostate cancer. J Clin Endocrinol Metab. 1984;58:36–40.
Mohler JL, Gregory CW, Ford OH 3rd, Kim D, Weaver CM, Petrusz P, Wilson EM, French FS. The androgen axis in recurrent prostate cancer. Clin Cancer Res. 2004;10:440–8.
Mostaghel EA, Page ST, Lin DW, Fazli L, Coleman IM, True LD, Knudsen B, Hess DL, Nelson CC, Matsumoto AM, et al. Intraprostatic androgens and androgen-regulated gene expression persist after testosterone suppression: therapeutic implications for castration-resistant prostate cancer. Cancer Res. 2007;67:5033–41.
Nishiyama T, Hashimoto Y, Takahashi K. The influence of androgen deprivation therapy on dihydrotestosterone levels in the prostatic tissue of patients with prostate cancer. Clin Cancer Res. 2004;10:7121–6.
Labrie F, Belanger A, Dupont A, Luu-The V, Simard J, Labrie C. Science behind total androgen blockade: from gene to combination therapy. Clin Invest Med. 1993;16:475–92.
Palapattu GS, Wu C, Silvers CR, Martin HB, Williams K, Salamone L, Bushnell T, Huang LS, Yang Q, Huang J. Selective expression of CD44, a putative prostate cancer stem cell marker, in neuroendocrine tumor cells of human prostate cancer. Prostate. 2009;69:787–98.
Reis LO, Vieira LF, Zani EL, Denardi F, de Oliveira LC, Ferreira U. Assessment of serum chromogranin-A as prognostic factor in high-risk prostate cancer. J Investig Med. 2010;58:957–60.
Gugliotta A, Ferreira U, Reis LO, Matheus WE, Denardi F, Mamprim Stopiglia R, Piccolotto Naccarato AM. Satisfaction analysis in men presenting with localized prostate cancer treated with radical prostatectomy or radiotherapy: psychological and social aspects. Actas Urol Esp. 2008;32:411–6.
Andriole GL, Humphrey P, Ray P, et al. Effect of the dual 5alpha-reductase inhibitor dutasteride on markers of tumor regression in prostate cancer. J Urol. 2004;172:915–9.
Reis LO, Pereira TC, Lopes-Cendes I, Ferreira U. MicroRNAs: a new paradigm on molecular urological oncology. Urology. 2010;76:521–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reis, L.O. Old issues and new perspectives on prostate cancer hormonal therapy: the molecular substratum. Med Oncol 29, 1948–1955 (2012). https://doi.org/10.1007/s12032-011-9991-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-9991-z