Abstract
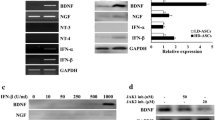
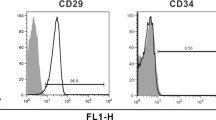
Parkinson’s disease (PD) is a neurodegenerative disorder with its motor phenomena due mostly to loss of dopamine-producing neurons in the substantia nigra. Pharmacological treatments aimed to increase the deficient dopaminergic neurotransmission are effective in ameliorating the cardinal symptoms, but none of these therapies is curative. It has been suggested that treatment with neurotrophic factors (NTFs) might protect and prevent death of the surviving dopaminergic neurons and induce proliferation of their axonal nerve terminals with reinnervations of the deafferented striatum. However, long-term delivery of such proteins into the CNS is problematic. We therefore aimed to differentiate ex vivo human bone marrow-derived mesenchymal stromal cells into astrocyte-like cells, capable of generating NTFs for future transplantation into basal ganglia of PD patients. Indeed, mesenchymal stromal cells treated with our novel astrocyte differentiation medium, present astrocyte-like morphology and express the astrocyte markers S100β, glutamine synthetase and glial fibrillary acidic protein. Moreover, these astrocyte-like cells produce and secrete significant amounts of glial cell line-derived neurotrophic factor (GDNF), nerve growth factor (NGF), and brain-derived neurotrophic factor as indicated by messenger RNA, real-time polymerase chain reaction, ELISA, and Western blot analyses. Such NTF-producing cells transplanted into the striatum of 6-hydroxydopamine-lesioned rats, a model of PD, produced a progressive reduction in the apomorphine-induced contralateral rotations as well as behavioral improvement in rotor-rod and the “sunflower seeds” eating motor tests. Histological assessments revealed that the engrafted cells survived and expressed astrocyte and human markers and acted to regenerate the damaged dopaminergic nerve terminal system. Findings indicate that our novel procedure to induce NTF-producing astrocyte-like cells derived from human bone marrow stromal cells might become a promising and feasible autologous transplantation strategy for PD.






Similar content being viewed by others
References
Arnhold, S., Klein, H., Klinz, F. J., et al. (2006). Human bone marrow stroma cells display certain neural characteristics and integrate in the subventricular compartment after injection into the liquor system. European Journal of Cell Biology, 85, 551–565. doi:10.1016/j.ejcb.2006.01.015.
Bahat-Stroomza, M., Gilgun-Sherki, Y., Offen, D., et al. (2005). A novel thiol antioxidant that crosses the blood brain barrier protects dopaminergic neurons in experimental models of Parkinson’s disease. The European Journal of Neuroscience, 21, 637–646. doi:10.1111/j.1460-9568.2005.03889.x.
Bjorklund, A., Kirik, D., Rosenblad, C., et al. (2000). Towards a neuroprotective gene therapy for Parkinson’s disease: Use of adenovirus, AAV and lentivirus vectors for gene transfer of GDNF to the nigrostriatal system in the rat Parkinson model. Brain Research, 886, 82–98. doi:10.1016/S0006-8993(00)02915-2.
Blondheim, N. R., Levy, Y. S., Ben-Zur, T., et al. (2006). Human mesenchymal stem cells express neural genes, suggesting a neural predisposition. Stem Cells and Development, 15, 141–164. doi:10.1089/scd.2006.15.141.
Chauhan, N. B., Siegel, G. J., & Lee, J. M. (2001). Depletion of glial cell line-derived neurotrophic factor in substantia nigra neurons of Parkinson’s disease brain. Journal of Chemical Neuroanatomy, 21, 277–288. doi:10.1016/S0891-0618(01)00115-6.
Chen, L. W., Yung, K. L., & Chan, Y. S. (2005a). Reactive astrocytes as potential manipulation targets in novel cell replacement therapy of Parkinson’s disease. Current Drug Targets, 6, 821–833. doi:10.2174/138945005774574506.
Chen, Q., He, Y., & Yang, K. (2005b). Gene therapy for Parkinson’s disease: Progress and challenges. Current Gene Therapy, 5, 71–80.
Collier, T. J., & Sortwell, C. E. (1999). Therapeutic potential of nerve growth factors in Parkinson’s disease. Drugs & Aging, 4, 261–287. doi:10.2165/00002512-199914040-00003.
Crigler, L., Robey, R. C., Asawachaicharn, A., et al. (2005). Human mesenchymal stem cell subpopulations express a variety of neuro-regulatory molecules and promote neuronal cell survival and neuritogenesis. Experimental Neurology, 1, 54–64.
Dass, B., Olanow, C. W., & Kordower, J. H. (2006). Gene transfer of trophic factors and stem cell grafting as treatments for Parkinson’s disease. Neurology, 66, 89–103.
Deng, J., Petersen, B. E., Steindler, D. A., et al. (2006). Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells (Dayton, Ohio), 24, 1054–1064. doi:10.1634/stemcells.2005-0370.
Freed, C. R., Greene, P. E., Breeze, R. E., et al. (2001). Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. The New England Journal of Medicine, 344, 710–719. doi:10.1056/NEJM200103083441002.
Gill, S. S., Patel, N. K., Hotton, G. R., et al. (2003). Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nature Medicine, 9, 589–595. doi:10.1038/nm850.
Gonzalez, C., & Kolb, B. (2003). A comparison of different models of stroke on behaviour and brain morphology. The European Journal of Neuroscience, 18, 1950–1962. doi:10.1046/j.1460-9568.2003.02928.x.
Gouhier, C., Chalon, S., Aubert-Pouessel, A., et al. (2002). Protection of dopaminergic nigrostriatal afferents by GDNF delivered by microspheres in a rodent model of Parkinson’s disease. Synapse (New York, N.Y.), 44, 124–131. doi:10.1002/syn.10063.
Hagell, P., Piccini, P., Bjorklund, A., et al. (2002). Dyskinesias following neural transplantation in Parkinson’s disease. Nature Neuroscience, 5, 627–628.
Herzog, E. L., Chai, L., & Krause, D. S. (2003). Plasticity of marrow-derived stem cells. Blood, 102, 3483–3493. doi:10.1182/blood-2003-05-1664.
Kirik, D., Winkler, C., & Bjorklund, A. (2001). Growth and functional efficacy of intrastriatal nigral transplants depend on the extent of nigrostriatal degeneration. The Journal of Neuroscience, 21, 2889–2896.
Kordower, J. H., Palfi, S., Chen, E. Y., et al. (1999). Clinicopathological findings following intraventricular glial derived neurotriphic factor treatment in a patient with Parkinson’s disease. Annals of Neurology, 46, 419–424. doi:10.1002/1531-8249(199909)46:3<419::AID-ANA21>3.0.CO;2-Q.
Levy, Y. S., Stroomza, M., Melamed, E., & Offen, D. (2004). Embryonic and adult stem cells as a source for cell therapy in Parkinson’s disease. Journal of Molecular Neuroscience, 24, 353–386. doi:10.1385/JMN:24:3:353.
Levy, Y. S., Gilgun-Sherki, Y., Melamed, E., & Offen, D. (2005). Therapeutic potential of neurotrophic factors in neurodegenerative diseases. BioDrugs, 19, 97–127. doi:10.2165/00063030-200519020-00003.
Levy, Y. S., Bahat-Stroomza, M., Barzilay, R., Burshtein, A., Bulvik, S., Barhum, Y., et al. (2008). Regenerative effect of neural-induced human mesenchymal stromal cells in rat models of Parkinson’s disease. Cytotherapy, 10, 340–352. doi:10.1080/14653240802021330.
Lin, L. F. H., Doherty, D. H., Lile, J. D., et al. (1993). A glial cell-derived neurotrophic factor for midbrain dopaminergic neurons. Science, 260, 1130–1132. doi:10.1126/science.8493557.
Lu, L., Zhao, C., Liu, Y., et al. (2005). Therapeutic benefit of TH-engineered mesenchymal stem cells for Parkinson’s disease. Brain Research Protocols, 15, 46–51. doi:10.1016/j.brainresprot.2005.03.002.
Müller, H. W., Junghans, U., & Kappler, J. (1995). Astroglial meurotrophic and neurite-promoting factors. Pharmacology & Therapeutics, 65, 1–18. doi:10.1016/0163-7258(94)00047-7.
Nagatsu, T., Mogi, M., Ichinose, H., & Togari, A. (2000). Changes in cytokines and neurotrophins in Parkinson disease. Journal of Neural Transmission, 60, 277–290.
Nakagawa, T., & Schwartz, J. P. (2004). Gene expression patterns in in-vivo normal adult astrocytes compared with cultured neonatal and normal adult astrocytes. Neurochemistry International, 45, 203–242.
Nakajima, K., Hida, H., Shimano, Y., Fujimoto, I., Hashitani, T., Kumazaki, M., et al. (2001). GDNF is a major component of trophic activity in DA-depleted striatum for survival and neurite extension of DAergic neurons. Brain Research, 916, 76–84. doi:10.1016/S0006-8993(01)02866-9.
Nutt, J. G., Burchiel, K. J., Comella, C. L., et al. (2003). Randomized, double blind trial of glial cell line-derived neurotrophic factor (GDNF) in PD. Neurology, 60, 69–73.
Park, K. W., Eglitis, M. A., & Mouradian, M. M. (2001). Protection of nigral neurons by GDNF-engineered marrow cell transplantation. Neuroscience Research, 40, 315–323. doi:10.1016/S0168-0102(01)00242-5.
Paxinos, G., Watson, C., Pennisi, M., et al (1985) Bregma, lambda and the interaural midpoint in stereotaxic surgery with rats of different sex, strain and weight. Journal of Neuroscience Methods, 13, 139–143.
Pekny, M., & Nilsson, M. (2005). Astrocyte activation and reactive gliosis. Glia, 50, 427–434. doi:10.1002/glia.20207.
Pinkas-Kramarski, R., Eilam, R., Spiegler, O., et al. (1994). Brain neurons and glial cells express Neu differentiation factor/heregulin: A survival factor for astrocytes. Proceedings of the National Academy of Sciences of the United States of America, 91, 9387–9391. doi:10.1073/pnas.91.20.9387.
Ransom, B. R. (1991). Vertebrate glial classification, lineage, and heterogeneity. Annals of the New York Academy of Sciences, 633, 19–26. doi:10.1111/j.1749-6632.1991.tb15591.x.
Rosenblad, C., Martinez-Serrano, A., & Bjorklund, A. (1998). Intrastriatal glial cell line-derived neurotrophic factor promotes sprouting of spared nigrostriatal dopaminergic afferents and induces recovery of function in a rat model of Parkinson’s disease. Neuroscience, 82, 129–137. doi:10.1016/S0306-4522(97)00269-8.
Rosenblad, C., Kirik, D., Devaux, B., et al (1999). Protection and regeneration of nigral dopaminergic neurons by neurturin or GDNF in a partial lesion model of Parkinson’s disease after administration into the striatum or the lateral ventricle. European Journal of Neuroscience, 11, 1554–1566.
Rudge, J. S., Alderson, R. F., Pasnikowski, E., et al. (1992). Expression of ciliary neurotrophic factor and the neurotrophins—nerve growth factor, brain-derived neurotrophic factor and neurotrophin 3—in cultured rat hippocampal astrocytes. The European Journal of Neuroscience, 4, 459–471. doi:10.1111/j.1460-9568.1992.tb00896.x.
Ryu, M. Y., Lee, M. A., Ahn, Y. H., et al. (2005). Brain transplantation of neural stem cells cotransduced with tyrosine hydroxylase and GTP cyclohydrolase 1 in Parkinsonian rats. Cell Transplantation, 14, 193–202. doi:10.3727/000000005783983133.
Salinas, M., Diaz, R., Abraham, N. G., et al. (2003). Nerve growth factor protects against 6-hydroxydopamine-induced oxidative stress by increasing expression of heme oxygenase-1 in a phosphatidylinositol 3-kinase-dependent manner. The Journal of Biological Chemistry, 278, 13898–13904. doi:10.1074/jbc.M209164200.
Shimoke, K., & Chiba, H. (2001). Nerve growth factor prevents 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced cell death via the Akt pathway by suppressing caspase-3-like activity using PC12 cells: Relevance to therapeutical application for Parkinson’s disease. The Journal of Neuroscience, 63, 402–409. doi:10.1002/1097-4547(20010301)63:5<402::AID-JNR1035>3.0.CO;2-F.
Teismann, P., Tieu, K., Cohen, O., et al. (2003). Pathogenic role of glial cells in Parkinson’s disease. Movement Disorders, 18, 121–129. doi:10.1002/mds.10332.
Tondreau, T., Lagneaux, L., Dejeneffe, M., et al. (2004). Bone marrow-derived mesenchymal stem cells already express specific neural proteins before any differentiation. Differentiation, 72, 319–326. doi:10.1111/j.1432-0436.2004.07207003.x.
Yurek, D. M., & Fletcher-Turner, A. (2001). Differential expression of GDNF, BDNF, and NT-3 in the aging nigrostriatal system following a neurotoxic lesion. Brain Research, 891, 228–235. doi:10.1016/S0006-8993(00)03217-0.
Acknowledgment
The authors thank Moshe Mizrachy, Alejandro Aran, and Debi Iskovich for their technical assistance. The work was performed in partial fulfillment of the requirements for a Ph.D. degree of Merav Bahat-Stromza, Sackler Faculty of Medicine, Tel Aviv University, Israel. The study was partially supported by The National Parkinson Foundation, USA, The Norma and Alan Aufzien Chair for Research in Parkinson’s Disease, Tel Aviv University, Israel (E.M.), Brainstorm Cell Therapeutics Ltd, and the Ted Arison Family Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahat-Stroomza, M., Barhum, Y., Levy, Y.S. et al. Induction of Adult Human Bone Marrow Mesenchymal Stromal Cells into Functional Astrocyte-Like Cells: Potential for Restorative Treatment in Parkinson’s Disease. J Mol Neurosci 39, 199–210 (2009). https://doi.org/10.1007/s12031-008-9166-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-008-9166-3